Silversides in South Brazil: Morphological and ecological aspects
M. A. Bemvenuti
Laboratório de Ictiologia, Departamento de Oceanografia, Fundação Universidade Federal do Rio Grande, C. Postal 474, Rio Grande, 96201-900, RS, Brazil.
Address correspondence to: Dr. M.A. Bemvenuti. Laboratório de Ictiologia, Departamento de Oceanografia, FURG, C.Postal 474, Rio Grande, CEP 96201-900, RS, BRAZIL. E-mail: docmab@furg.br
Key words: Silversides; Odontesthes; Brazil; Morphology; Multivariate.
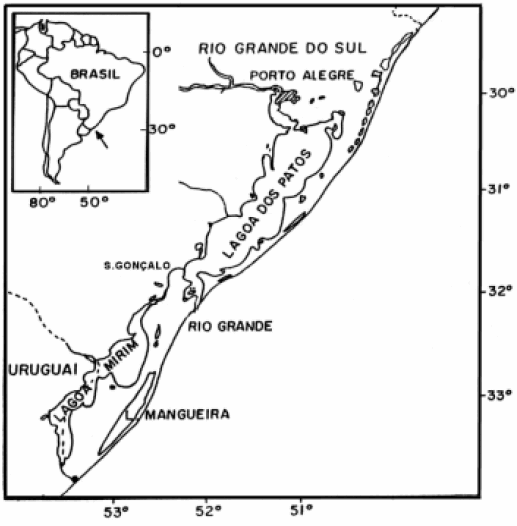
The coastal plain of the Rio Grande do Sul state
(RS) in southern Brazil, has an enormous lagoon complex
in its southern portion formed by Patos Lagoon,
Mirim Lagoon, Mangueira Lagoon and several other
smaller lakes. This system was originated from successive
transgression and regression cycles, which took
place since the upper Pleistocene (Vilwoock, 1984).
Patos Lagoon is a huge chocked lagoon (10.360 area
km2), with an estuarine area comprising 10% of the total
area in its southern portion, where one can find small,
oligohaline shallow water embayment less than 3m
maximum depth (Delaney, 1965).
Mirim Lagoon has an area of 3.749 km2, but only
2.382 km2 is located on Brazilian's territory because the
remaining belongs to Uruguay. Mirim Lagoon communicates
with Patos Lagoon through the São Gonçalo
channel (Delaney, 1965).
Other small coastal lagoons are located along the
coastal line, and are locally known as "lakes in rosary".
The larger one is the Mangueira Lake, a closed lake
with an area of approximately 800 km2. Its northern
portion is connected with the Taim wetland, which is
formed by small lakes (Jacaré, Nicola, Flores). The other
lakes are shallow (from 2 to 3 m) and are situated in the
north portion of the Rio Grande do Sul state (Fig. 1).

FIGURE 1. Area of distribution of silverside species in southern Brazil.
Ten species of silversides, comprising the genus Odontesthes, occur along the RS coastal plain (Bemvenuti, 2002; Malabarba and Dyer, 2002). In Patos Lagoon estuary and its adjacent marine coastal area occurs O. argentinensis (Valenciennes) and O. incisa (Jenyns), whereas in the freshwater habitats of Patos- Mirim lagoon system can be found O. bonariensis (Valenciennes), O. humensis De Buen, O. retropinnis (De Buen), O. aff. perugiae Evermann and Kendall and O. mirinensis Bemvenuti. In contrast, O. bicudo Malabarba and Dyer, O. piquava Malabarba and Dyer and O. ledae Malabarba and Dyer are endemic of a chain of small shallow lakes spread along the RS north coastline. Most of Odontesthes species co-occur in the same habitats and they are characterized by morphological similarities, which hinder their taxonomic identification.
Comparisons among multivariate morphometric techniques
There has been a lot of controversy regarding morphological
differences among organisms due to the lack
of agreement about which morphometric variables are
more adequate and which kind of mathematic transformations of these variables are better to distinguish different
forms.
Morphometric variables obtained by traditional
measurement techniques do not take into account allometric
variation or the different growth stages, which
occur naturally in the organisms. Consequently, these
data usually bring error to the analyses, leading to misleading
interpretation and, ultimately, do not represent
the real form of the organisms Bookstein et al. (1985).
According to these authors, the use of traditional morphometric
measurements can lead to more difficulties
when differentiating among forms due to: (1) longitudinal
measurements along the main body axis produces
repetitive information in the same direction, which results
in a unequal coverage of the body form, (2) utilization
of maximum and minimum distances, which are
not homologous from one individual to another, (3) utilization
of long measurements, crossing several growth
units (e.g., bones) and short ones, which contain local
information.
Currently, the selection of characters that represent
the whole body utilize short measure distances that connect
anatomical points (homologous among individuals)
(e.g., truss network) (Strauss and Bookstein, 1982).
The different forms are distinguished in all directions,
which compensate for random errors of measurements.
Beside the description of growth and allometric patterns
inside populations, the technique can discriminate
among groups of organism that change in size and shape.
A comparison between traditional measurements
and truss network (Fig. 2A, B) was investigated between
two silverside species Odontesthes bonariensis and O.
humensis through a principal component analysis. The
first main component have been interpreted as variation
in size, which results from the different growth
stages in fishes, whereas the second component represents
changes in shape of the organisms (Bookstein et al., 1985).

FIGURE 2. (A) Conventional set of distances measures used in fish morphometrics of O. humensis; (B) a truss networks of distances measures connect ten homologous landmarks of O. bonariensis (for explanation of this data set see Bemvenuti and Rodrigues, 2002).
In the comparative study of the different forms between two species it is interesting to separate the information obtained with size due to the differential growth showed by the organisms. Therefore, the data were log transform and also adjusted to remove the size effect factor (Burnaby, 1966). The two series of measurements used during adjust and no-adjust analyzes showed the formation of two groups. However, truss network analysis results in a better differentiation between O. bonariensis and O. humensis, probably because the method enhances the discrimination of the forms with the utilization of cross measurements not considered in the traditional method (Fig. 3A, 3B). The differential pattern between both groups was also observed by the position of values in the graph, which was located nearer one another in the truss network when compared with the traditional method (Fig. 3B; Bemvenuti and Rodrigues, 2002).

FIGURE 3. Principal components analysis of O. bonariensis (n=26) and O. humensis (n=25), in the space of the first two adjusted principal components (PC1, PC2), for (A) 25 traditional measures and (B) 21 distances measures with truss network.
Morphological differentiation among species of the perugiae complex1
Due to the large variation in the abiotic factors in the estuarine area of the Patos Lagoon it is possible to find O. argentinensis and O. mirinensis in the same location inside the estuary, but in distinct conditions of salinity. Odontesthes argentinensis shows preference for the lower reaches of the estuary, whereas O. mirinensis can be found in the northern reaches of the estuary, near the São Gonçalo Canal and Mirim Lagoon. This pattern brings difficult to the taxonomic identification of these species in the estuarine region. With the principal component analysis it was possible to distinguish O. argentinensis and O. mirinensis groups, but differences were not observed in body proportion between both species (Fig. 4). Only the meristic character of number of lower gill rakers shows differentiation: O. argentinensis 20 and 24 gill rakers, and O. mirinensis - 24 and 29 gill rakers (Bemvenuti, 1995, 2000, 2002).

FIGURE 4. Principal components analysis of O. mirinensis (n=41) and O. argentinensis (n=35), in the space of the first two adjusted main components (PC1, PC2), for 21 measures with truss network.
In the freshwater sites, the silversides O. aff. perugiae e O. mirinensis2 also were discriminated through the form (Fig. 5), being possible to observe a superficial variation in snout length, head and jaw (Fig. 6). In O. perugiae, the head is prominent (24.3% cp), the snout is salient (9,1% cp) and the maxilla is located ahead of the mandible. These proportions are relatively larger in O. peruagiae when compared with O. mirinensis, where the head is smaller (22.5% cp), the snout is shorter (6.9% cp) and the maxilla and mandible are in the same anterior projection (Bemvenuti, 1995, 1997, 2002).
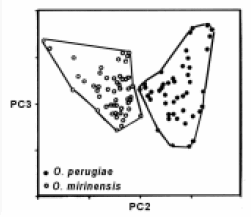
FIGURE 5. Principal components analysis of O. aff. perugiae (n=52) and O. mirinensis (n=44), in the space of the second and third components (PC2, PC3), for 25 traditional measures.

FIGURE 6. Lateral view of the head of O. perugiae (A) e O. mirinensis (B).
Description of the bone structure for the perugiae complex
A compared description of some bone structures
was investigated to incorporate new data for the differentiation
of the following species O. mirinensis, O.
perugiae and O. argentinensis3. The teeth in the vomer
are placed in three large plates in O. perugiae, in a medium
size plate in O. argentinensis, and absents in O.
mirinensis. The accessory canal of the infraorbital 1
(IO1) has two external pores in O. mirinensis, one anterior
and intern pore and other posterior and external pore
in O. perugiae, and it is absent in O. argentinensis. The
infraorbital 2 (IO2) has a main dorsal canal and four
long accessory canals with six pores in O. argentinensis,
three long canals with five pores in O. mirinensis, and
two short canals in O. perugiae.
The premaxilla shows posterior process upward,
lengthier than higher in O. mirinensis, slim, large and
straight in O. perugiae positioned upward in O.
argentinensis. The mandibular sensory canal of the
dentary shows seven pores in O. mirinensis and O.
perugiae, and six in O. argentinensis. The tooth plate
of the endopterygoid is numerous in O. mirinensis and O. argentinensis, whereas it is reduced a few and scattered
tooth in O. perugiae. The preopercle sensory canal
of the horizontal shaft has four large pores in O.
mirinensis, three large pores in O. argentinensis, and
no pore in O. perugiae due to the absence of bridges
among them. The vertical shaft is totally open in O.
mirinensis, with a wide opening in O. perugiae and with
two openings in O. argentinensis. The hemal funnel,
expansion of the hemal arch, start at the 24a vertebra in O. argentinensis, whereas in O. mirinensis, O. perugiae,
and in the other freshwater silversides, the hemal funnel
start at the 27a vertebra. The estuarine species has
the same number of precaudal and caudal vertebrae,
whereas the freshwater species has a larger number of
pre-caudal vertebrates (Dyer, 1997; Bemvenuti, 2005).
Geographical differentiation in O. argentinensis
Odontesthes argentinensis is an estuarine-resident (Chao et al., 1985) showing a complete life-cycle inside the estuary. Morphological variations were observed among specimens occurring in the estuarine area and in the marine adjacent region (Fig. 7; Bemvenuti, 1995; 1997).
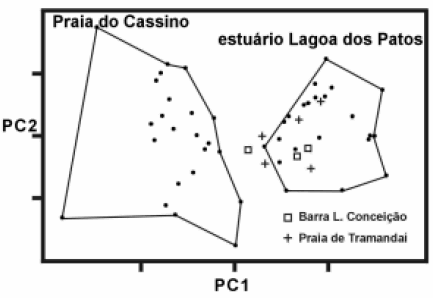
FIGURE 7. Principal components analysis of O. argentinensis, obtained in the marine coastal (n=26 - Praia do Cassino) and estuarine area (n=26 - Lagoa dos Patos; n=5 - Tramandai; n=3 - Lagoa da Concei‹o), in the space of the first two adjusted components (PC1, PC2), for 25 traditional measures.
Based on isoenzyme electrophoresis analysis, Beheregaray and Levy (2000) also observed similar patterns of genetic divergence between both populations. These authors suggested that the colonization of the estuary occurred when a segment of the marine silverside group managed to stay in the estuarine area, leading to the generation of differences among these groups. Based on molecular analysis, Beheregaray and Sunnucks (2001) suggested that all species of the genus Odontesthes are derived from the marine group. Therefore, this geographical area supports two resident silverside populations: one inside the estuarine area of Patos Lagoon and other in the adjacent marine area (Bemvenuti, 2000; Beheregaray, 2000; Beheregaray and Levy, 2000).
Bio-ecological aspects of the silversides
Feeding
The occurrence of several silverside species along
the coastal lagoons of southern Brazil suggest changes
in the feeding modes and prey selection to avoid diet
overlap and, therefore, allowing their co-existence in the
same system. The feeding behaviour is a fundamental
step in understanding the ecology of one species and its
place in the food web.
Species occurring in the same location probably feed
upon different kinds of food, or occupy different habitats
or utilize distinct resources in different time frames. Such
species usually show diets with low overlap, which can
be attributed to differences in mouth morphology and/or
feeding behaviour. Diet composition must be related with
morphological structures linked to feeding, such as the
form, position and mouth size, and form and number of
gill rakers (Wootton, 1990).
The feeding habit of O. humensis is benthos carnivore,
preying upon mainly on mollusks Heleobia sp.
(61.4% frequency of occurrence FO), Corbicula
fluminea (58%), Neocorbicula limosa (17.4%) and coleoptera
insects (18,8%). In lower frequency can be
found diptera insects, crustacean Paleomonetes
argentinus and fishes (Rodrigues and Bemvenuti, 2001).
In spite of being a pelagic organism, the species browse
for food in the bottom as well, making it different from
other silverside species. The predominance of Heleobia sp, in comparison of other mollusks found in the diet,
can be related with the high abundance of this prey in
the study area and, probably, due to its low size. The
majority of the shells of adults from C. fluminea and N.
limosa were found crushed, whereas the shells of the
young individuals were found as whole shells.
The form and number of the short and thick gill
rakers (20-24= 4-6+15-19), as well the form and type
of the tooth in the pharyngeal plates4 (molariforms in
the center and conic in the edges), can be related with
the zoo-benthos diet. The pharyngeal plates play a role
in crushing mollusk shells and/or arthropod carapaces
when they are too large. Shells of young C. fluminea,
N. limosa and, in part Heleobia sp, are not crushed by
the pharyngeal plates but are swallowed as entire pieces. The occurrence of coleopteran insects, as a prey item,
can be explained by the high temperatures during the
sample collection. This item also showed larger size
them mollusk preys.
In the same environment, O. bonariensis shows zoobenthos
habit preying upon mollusks, isopods, shrimps,
tanaidacea, insects and small fishes. Among mollusk
items, showed preference in frequency of occurrence
by the gastropod Heleobia sp. (31%), bivalves Corbicula fluminea (16.7%) and Neocorbicula limosa (7.1%).
Odonthestes aff. perugiae shows preference by mollusks,
isopods, insect larvae, followed by tanaidacea and
copepods. The food items with higher weight in the
stomachs were the mollusk Corbicula fluminea (48%
of the total weight) and isopods (31%), followed by insect
larvae (8%), Hymenoptera insects (2%) and fishes
remaining (5%) showing lower weight.
In the estuarine region, O. argentinensis's juvenile
shows preference for zooplankton food items, whereas
adults show preference for benthic preys. Juveniles prey
upon copepods (63,8%) and diptera insects (35.5%),
whereas adults feed on tanaidacea Kalliapseudes schubartii (35.6%), amphipod (22%) and polychaete
Laeonereis acuta (25.7%) (Bemvenuti, 1990).
Reproduction
The estuarine specie O. argentinensis shows spawning
season between late winter and early spring (June
and November with a peak in September). The size of
first maturation (L50) is 230mm TL (Bemvenuti, 1987).
Recent information suggests a reduction of the maturation
size in females (162mm TL in the estuary and
179mm TL in the marine area).
The freshwater species O. humensis and O.
bonariensis spawn in the coastal lagoons during the early
winter and spring. Odontesthes bonariensis spawns between
May and July, whereas O. humensis between July
and September (Kleerekoper, 1945; Moraes, 1991). The
overlap in the spawning season increases the chances
of combination among theses species, which favors hybridization.
Preliminary karyotype studies5 suggest there
is chromosomal affinity between both species, with
chromosomes showing high similarity in number and
morphology.
In spite of the extent literature about O. bonariensis and, in a less extent, about O. humensis, there is no record
regarding hybridization with these species or others
South American silversides. Recently it was developed
its hybridization in laboratory with the help of natural
populations of O. humensis e O. bonariensis6. The
sampled individuals were analyzed by principal component.
Two groups were observed (Fig. 8); one group
showing a relationship between the natural population
of O. humensis with hybrids which male progenitors
were O. humensis, and the other group showed a relationship
between the natural populations of O.
bonariensis with hybrids which male progenitors were
O. bonariensis. Such affinity shows the morphological
similarity among the hybrids and the species of the male
progenitor.

FIGURE 8. Principal components analysis of O. bonariensis (n=6 - bm x bf), O. humensis (n=5 - hm x hf), hybrids of O. bonariensis male and O. humensis female (n=8 - bm x hf), hybrids of O. humensis male and O. bonariensis female (n=14 - hm x bf) in the space of the second and third components (PC2, PC3), for 21 distances measures with truss network.
1. In the lagoons located in the south portion of the Rio Grande do Sul, the perugiae complex is comprised by
the freshwater silversides O. perugiae, O. mirinensis, and the estuarine O. argentinensis. These three species
are morphologically much closed and the morphometric and meristic characters are no sufficient tools to
distinguish among them (Bemvenuti, 2002). According to Beheregaray et al. (2002), these silverside species
were originated from the marine ancestor O. argentinensis.
2. Odontesthes perugiae is a specie restrict to rivers. Its occurrence is cited
for Rio da Prata and Rio Paraná floodplain, Argentina (Marrero 1950), and
including Uruguai and Negro rivers. Beheregaray et al. (2002) believed
that the individuals from Uruguai river, RS are O. perugiae, and the individuals
from the coastal lagoons as a new specie, O. aff. perugiae.
Odontesthes mirinensis is restricted to coastal lagoons in the southern RS,
Mirim, Mangueira e Lagoa dos Patos (Tapes, Camaquã, Guaíba river). Its
high abundance is recorded in the Mirim Lagoon, with many juvenile individuals
being captured in the Canal São Gonçalo Channel, Pelotas, which
makes the connection between Mirim lagoon and Lagoa dos Patos
(Bemvenuti, 1995).
3. The complete bone description for all Odontesthes species can be found
in Bemvenuti (2005).
4. The pharyngeal plates are distributed in four pairs, being three located in the upper portion of the mouth and
the other in the lower portion. The three plates of the upper portion have different size; one is large and oval
shape with molariform tooth showing different size and the other two are smaller, external with small, slim and
conic tooth. The lower plate is large, has a triangular shape, showing in the center several tooth of medium and
small size, with a molariform shape, and in its edges show few small, slim and conic teeth.
5. Personal communication of G. Born (DCMB-FURG, Rio Grande, Rio
Grande do Sul, Brasil).
6. Hybridization developed in laboratory in partnership with J. Pouey (UFPel,
Pelotas, Rio Grande do Sul, Brazil).
References
Beheregaray LB (2000). Molecular evolution, biogeography and
speciation of the Neotropical fish genus Odontesthes. PhD
thesis, Macquarie Univ., Sydney, 211p.
Beheregaray LB, Levy JA (2000). Population genetics of the silverside Odontesthes argentinensis (Teleostei, Atherinopsidae):
evidence for speciation in an estuary of southern Brazil.
Copeia, Lawrence, pp. 441-447.
Beheregaray LB, Sunnucks P (2001). Fine-scale genetic structure,
estuarine colonization and incipient speciation in the marine
silverside fish Odontesthes argentinensis. Molecular Ecology,
Oxford, 10: 2849-2866.
Beheregaray LB, Sunnucks P, Briscoe DA (2002). A rapid f ish radiation
associated with the last sea-level changes in southern
Brazil: the silverside Odontesthes perugiae complex. Proc R
Soc London B 269: 65-73.
Bemvenuti MA (1987). Abundância, distribuição e reprodução de
peixes-rei (Atherinidae) na região estuarina da Lagoa dos
Patos, RS, Brasil. Atlântica, Rio Grande, 9(1): 5-32.
Bemvenuti MA (1990). Hábitos alimentares de peixes-rei
(Atherinidae) na região estuarina da Lagoa dos Patos, Rs,
Brasil. Atlântica, Rio Grande, 12: 79-102.
Bemvenuti MA (1995). Odontesthes mirinensis, um novo peixerei
(Pisces, Atherinidae, Atherinopsinae), para o extremo sul
do Brasil. Revta Bras Zool Curitiba, 12(4): 881-903.
Bemvenuti MA (1997). Relações morfológicas e osteológicas entre
Odontesthes perugiae e O. mirinensis (Pisces: Atherinidae,
Atherinopsinae). Atlântica, Rio Grande, 19: 113-131.
Bemvenuti MA (2000). Diferenciação geográfica do peixe-rei Odontesthes argentinensis (Atherinopsidae), no extremo sul
do Brasil, através da morfometria multivariada. Atlântica, Rio
Grande, 22: 71-79.
Bemvenuti MA (2002). Diferenciação morfológica das espécies de
peixes-rei Odontesthes Evermann and Kendall (Osteichthyes,
Atherinopsidae) no extremo sul do Brasil: morfometria
multivariada. Revta bras Zool, Curitiba, 19(1): 239-249.
Bemvenuti MA (2005). Osteologia comparada entre as espécies de
peixes-rei Odontesthes Evermann and Kendall (Osteichthyes,
Atherinopsidae), do extremo sul do Brasil. Revta bras Zool,
Curitiba, (in press).
Bemvenuti MA, Rodrigues FL (2002). Análise comparativa entre
técnicas morfométricas aplicadas a Odontesthes bonariensis (Valenciennes) e Odontesthes humensis De Buen (Osteichthyes,
Atherinopsidae). Revta bras Zool, Curitiba, 19(3): 789-796.
Bookstein FL, Chernoff B, Elder R, Humphries J, Smith G, Strauss
R (1985). Morphometrics in evolutionary biology. Acad Nat
Sci Philad, Philadelphia, (15): 1-277.
Burnaby TP (1966). Growth invariant discriminant functions and
generalized distances. Biometrics, Arlington, 22: 96-110.
Chao LB, Pereira LE, Vieira JP (1985). Estuarine fish community
of the Patos Lagoon (Lagoa dos Patos, RS, Brazil) - A baseline
study. Ch.20, 26p. In: Yánez-Arancibia (ed) Fish community
ecology in estuaries and coastal lagoons. Towards na ecosystem
integration. UNANN, México 900p.
Delaney PJV (1965). Fisiografia e geologia da superfície da Planície
Costeira do Rio Grande do Sul. UFRGS, Porto Alegre, 6, 195p.
Dyer BS (1997). Phylogenetic revision of Atherinopsinae (Teleostei,
Atherinopsidae), with comments on the systematics of the
South American freshwater fish genus Basilichthys Girard.
Miscellaneous Publications, Museum of Zoology, University
of Michigan, Ann Arbor, USA (185): 1-64.
Kleerekoper H (1945). O peixe-rei. Serv Infor Min Agric Rio de
Janeiro, 98p.
Malabarba LR, Dyer BS (2002). Description of three new species
of the genus Odontesthes from the rio Tramandai drainage,
Brazil (Atheriniformes: Atherinopsidae). Ichthyol Explor
Freshwaters, München, 13(3): 257-272.
Marrero A (1950). Flechas de Plata. Atherinidos argentinos
pejerreys y laterinos. Ed. Breitman Buenos Aires, 175p.
Moraes PRR (1991). Peixe-rei: maturação e reprodução em
laboratório. Revta da UCPel, Pelotas 1(1): 60-73.
Rodrigues FL, Bemvenuti MA (2001). Hábito alimentar e osteologia
da boca do peixe-rei, Odontesthes humensis de Buen
(Atheriniformes, Atherinopsidae) na Lagoa Mirim, Rio
Grande do Sul, Brasil. Revta bras. Zool 18(3): 793-802.
Strauss RE, Bookstein, FL (1982). The truss: body form reconstructions
in morphometrics. Syst. Zool. Washington, 31(2):
113-135.
Vilwoock JA (1984). Geology of the coastal province of Rio Grande
do Sul, southern Brazil: a syntesis. Pesquisas, UFRGS, Porto
Alegre, 16: 5-50.
Wootton RJ (1990). Ecology of Teleost Fishes. New York, Chapman
and Hall, 404 p.
Received on February 3, 2005.
Accepted on May 11, 2005.