The effect of transportation stress on tissue ascorbic acid levels of
Mexican silverside (Chirostoma estor estor Jordan, 1979)
M. G. Ríos-Durán*, A. R. Hernández-Téllez**, C. A. Martínez-Palacios** and L. G. Ross***.
* Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Circuito exterior s/n, Ciudad
Universitaria, México, D.F.
** Universidad Michoacana de San Nicolás de Hidalgo. Av. San Juanito Itzícuaro s/n, Col. San Juanito Itzícuaro,
Morelia, Michoacán, México.
*** Institute of Aquaculture, University of Stirling, FK9 4LA, Scotland, UK.
Address correspondence to: M.G. Ríos-Durán. Instituto de
Investigaciones sobre los Recursos Naturales, Universidad
Michoacana de San Nicolás de Hidalgo. Av. San Juanito Itzícuaro
s/n, Col. San Juanito Itzícuaro, Morelia, Michoacán, CP 58330,
MÉXICO. E-mail: riosd@icmyl.unam.mx
Key words: Ascorbic acid tissue levels; Mexican silverside; Transport stress
The Atherinopsid silverside (Chirostoma estor estor Jordan, 1879) is a native freshwater species from the
Central Mexican Plate. It is the principal species in an
artisanal fishery in Lake Pátzcuaro, one of the altiplano
lakes of Central Mexico, where for many years it has
been the basis of the subsistence of the riparian
P'urépecha Indian people. It has a high market demand
and a high commercial value and so is an important income
source in the region. However, problems like pollution,
introduction of exotic species and overfishing
have caused it to become an endangered species.
C. estor estor has a great potential for aquaculture,
and although there have been several attempts at culture
on experimental scales (Lara, 1974; Armijo and
Sasso, 1976), research on the basic requirements for its
culture began only recently. Martínez-Palacios et al. (2002a) described techniques for measuring and handling
C. estor estor larvae and determined the optimum
temperature for growth and survival. Other studies have
been conducted on physiology, feeding habits, anatomical
feeding structures and activity of digestive enzymes
of the species (Martínez-Palacios et al., 2002b) and the
effects of saline environments on growth and survival
of eggs and larvae of whitefish are now well known
(Martínez-Palacios et al., 2004). Currently, several studies
are in progress on feeding requirements, in vivo and
in vitro digestibility of different food sources, molecular
biology and stress responses in order to establish the
optimal conditions for aquaculture.
The species is very easily stressed by handling and
changes in environmental conditions such as salinity
and dissolved oxygen, result in high mortality, low
growth, lack of appetite, etc. According to Pankhurst
and van der Kraak (1997), stress can inhibit growth because
of its effect on metabolism and the endocrine pathways
that regulate this process.
It has been shown that vitamin C (ascorbic acid)
plays an important role in resistance to environmental
stress in fish (Wee, 1996; Papp et al., 1997; Li et al.,
1998; Henrique et al., 1998; Montero et al., 1999), as
well as acting on numerous physiological processes including
growth, bone formation, reproduction, wound
healing, and immune responses, amongst others (Lygren et al., 1999; Lim et al., 2000; Lee and Dabrowski, 2004).
One of the most widely accepted functions of vitamin
C is that it acts as an antioxidant as it interrupts free
radical chain reactions.
Vitamin C occurs at high levels in many vital organs
with an active metabolism and its concentration in
various tissues is related to the dietary intake of this
vitamin. In some tissues, such as the brain, it is accumulated
in high concentrations and, compared with other
storage organs such as liver and kidney, its levels can
be retained for longer. It seems that Vitamin C has a
great importance in protecting vital tissues against oxidation
processes, and this may explain the high levels
found in brain and other organs such as thymus in fish
(Gabaudan and Verlhac, 2001). According to Robinson
(1989), the best tissue for evaluating ascorbic acid status
in fish is debatable. Blood ascorbate levels are rapidly
affected by dietary vitamin C intake, thus these levels
not appear to be a good indicator of the vitamin C
status. Anterior kidney ascorbate levels have been suggested
as a good indicator of ascorbic acid reserves in
fish, but this tissue is small, which causes problems for
vitamin C assays. Some authors consider liver ascorbate
levels to be a better indicator of ascorbic acid status
than anterior kidney vitamin C levels. Lim and Lovell
(1978) found highly variable ascorbate concentrations
in anterior kidney of catfish, while concentrations of
this vitamin in liver were consistent. In other species
like Rainbow trout (Onchorhynchus mykiss) the liver
ascorbic acid concentration can be used as an index of
the vitamin C status (Hilton et al., 1977).
We evaluated the ascorbic acid concentration in
brain, liver, gonad and muscle of several wild
Chirostoma estor estor1 . Total ascorbic acid (TAA), reduced
ascorbic acid (AA) and Dehydro-ascorbic acid
(DHAA) concentrations were evaluated by HPLC
techniques2 (Fig. 1). Tissue ascorbate (TAA, AA and DHAA) levels were different with brain having the highest
values, followed by gonad, liver and muscle in both
sexes. No significant difference (p>0.05) exists in TAA
concentrations of the evaluated tissues between females
and males (Table 1), nor are there significant differences
between females and males in AA and DHAA concentrations,
except in brain, which has higher levels of AA
in males. As shown in Figure 2, both in females and
males, vitamin C in the evaluated tissues is found in
higher proportion as AA (reduced form) than DHAA
(oxidized form), with the exception of female brain although
this difference was not significant (p>0.05).
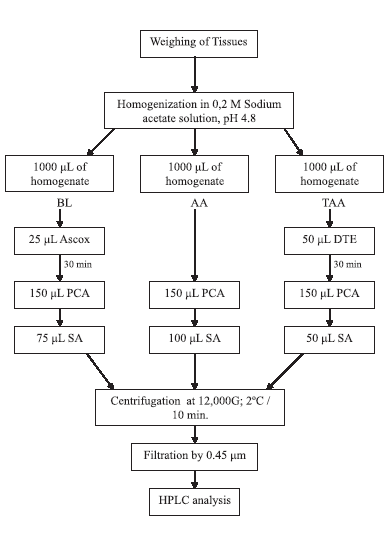
FIGURE 1. Sample preparation for the determination of ascorbic acid by HPLC
method. (BL: Base line determination, AA: reduced ascorbic acid quantification,
TAA: Total ascorbic acid quantification, Ascox: Ascorbate oxidase, DTE:
4% Dithioerythritol solution, PCA: 10% Perchloric acid solution, SA: 0.2 M
Sodium acetate Buffer).
TABLE 1.
Total Ascorbic Acid tissue levels (mg/g) found in wild
Chirostoma estor estor females and males. Data are
expressed as mean ± S.E.M.

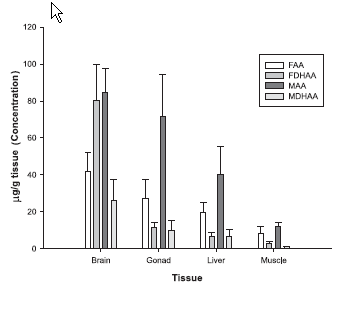
FIGURE 2. Reduced ascorbic acid (AA) and
Dehydro-ascorbic acid (DHAA) levels in both
females and males of wild Chirostoma estor
estor (FAA: Females reduced ascorbic acid;
FDHAA: Females Dehydro-ascorbic acid;
MAA: Males reduced ascorbic acid; MDHAA:
Males Dehydro-ascorbic acid).
During changes in environmental conditions ascorbic
acid fluctuations can occur in fish tissues which
suggests that vitamin C is involved in certain physiological
adaptation mechanisms. In some species like mullet
(Mugil cephalus L.), during periods of stress, tissue
ascorbate concentrations can be significantly altered
(decrease or increase) (Thomas, 1984). However, there
is no data on vitamin C status in C.estor estor and the
effects of stress on AA concentrations in tissues.
We monitored variations in ascorbic acid concentrations
after transport stress in brain, liver, gonad and
muscle of several C. estor estor adults. We took these
fish as the stressed group and the fish sampled previously,
without transport stress, as the control group3 .
After transport stress, both females and males show the
same pattern of tissue ascorbic acid (TAA, AA and
DHAA) as the controls, with the highest values in brain
and the lowest in muscle. In comparison with pre-stress
vitamin C levels there are not significant changes
(p>0.05) in tissue ascorbate concentrations (Fig. 3).

FIGURE 3. Total Ascorbic Acid (TAA) levels
found in the evaluated tissues of Chirostoma
estor estor females and males before (control
group) and after transport stress (stressed
group) (FBT: Females before transport; MBT:
Males before transport, FAT: Females after
transport, MAT: Males after transport).
No significant difference was found between females
and males in tissue TAA concentrations after
stress, with the exception of liver which had lower levels
in females than males. As in the control group, poststress
vitamin C was higher in both sexes as AA than
DHAA. However, there was no significant difference
between these two forms in the brain of females. There
is a trend of an increase of AA levels and a decrease of
DHAA levels in all tissues of both sexes after the transport
stress (Fig. 4), although the change in their proportion
is only significant (p<0.05) in female gonads. We
can conclude that the stress caused by three hours transportation
is not enough to cause significant changes in
tissue TAA concentrations, but it is sufficient to change
significantly the proportion of AA and DHAA in gonad
of females.
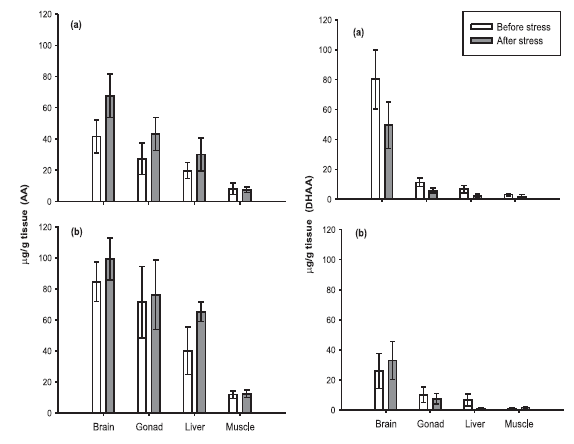
FIGURE 4. Reduced Ascorbic Acid (AA) and Dehydro-ascorbic Acid (DHAA)
tissue levels found in Chirostoma estor estor (a) females and (b) males
before (control group) and after transport stress (stressed group).
Context and perspectives
Because the evaluated fishes were taken from the
wild, it was inevitable that physical stress was caused
during capture; however, it is not known if such stress
caused significant changes in the normal tissue ascorbate levels. Thomas (1984) found that liver and brain
ascorbate contents were relatively unaffected by capture
stress. It was assumed that capture stress does not
alter the vitamin C stores in these tissues in C. estor
estor and that fish sampled without transport stress were
a valid control group.
As has been found in several fish species, highest
concentrations of TAA were found in brain tissue in C.
estor estor and these levels were maintained after the
transport stress, confirming that in brain tissue the protection
against oxidative damage is very important with
vitamin C acting as an electron receptor (Gabaudan and
Verlhac, 2001). According to Patro and Patnaik (1979)
in Ophiocephalus punctatus the ascorbic acid concentrations
in brain is unaffected by short-term exposure
to physical stress, which agrees with the results of this
study. Thomas (1984) also found this pattern in the brain
and liver of mullet Mugil cephalus L. after short-term
capture stress. In C. estor estor transport stress of three
hours did not alter the ascorbate content of this tissue.
Hilton et al. (1977) have proposed liver ascorbic
acid content as a reliable indicator of vitamin C status
in fish, and considered liver ascorbate concentrations
of 20 mg/g or below to be an indicator of poor vitamin
C status in rainbow trout. Lim and Lovell (1978) considered
levels of liver ascorbate below 30 mg/g as indicative
of vitamin C deficiency in catfish. Robinson
(1989) found that levels of ascorbic acid below 30mg/g
did not reflect a vitamin C deficiency in channel catfish
and that levels of 16.5 and 18.2 mg/g did not show
signs of vitamin C deficiency. As shown in Table 1, adult
C. estor estor had liver ascorbate levels of 25.9 mg/g
(females) and 46.2 mg/g (males), which most probably
indicates a good vitamin C status. Because the concentration
of vitamin C in tissues is related to the dietary
intake of the vitamin, the liver ascorbate concentrations
found may reflect its natural feeding based on zooplankton,
a natural supply of this vitamin. Nonetheless,
further data are needed to establish hepatic ascorbic acid
levels, which could be used as indicators of vitamin C
deficiency in this species. Liver TAA levels in silverside
were not affected by transport, which suggests that
this physical stress may not significantly influence the
vitamin C status of this fish.
As has been reported in other scurvy-prone fish
species (Table 2), C. estor estor has significant amounts
of ascorbic acid in the gonads, to guarantee the high
quality of gametes. For the control group, there were no
differences of TAA concentrations in gonads between
males and females. Most of the ascorbic acid found in
testis was in the reduced form, while in ovaries there
were no significant differences between reduced and
oxidized forms. Although TAA level was not influenced
by sex in C. estor estor, the proportion of reduced and
oxidized forms did vary in gonads. In North Sea dab
(Limanda limanda) Saborowski et al. (1997) found differences
in AA concentrations between male and female
gonads and, as was found in other species (Carassius
carassius, Gadus morhua, Perca flavescens), these levels
varied with the reproductive cycle (Seymour, 1981;
Sandnes and Braekkan, 1981; Dabrowski and Ciereszko,
1996). In C. estor estor, although the evaluated gonads
were immature (gonadosomatic indices between 0.11
and 0.25% for males, and between 0.27 to 0.47% for
females), these tissues had higher values than liver and
muscle. Different concentrations of vitamin C may be
expected in mature individuals, depending on the stage
of the reproductive cycle. Transportation stress did not
affect the TAA levels in either males or females gonads,
but the proportion of AA and DHAA changed significantly
in ovaries with AA (reduced form) increasing and
DHAA (oxidized form) decreasing. This suggests that
vitamin C would have acted as an electron receptor, effectively
protecting against oxidative damage in ovaries
in a stress situation.
TABLE 2.
Ascorbic acid (AA) concentrations in gonads of different wild scurvy-prone fishes
The lowest concentrations of TAA were found in
muscle of C. estor estor, indicating that this tissue is
not an important storage place for vitamin C. Low
muscle TAA levels have been observed in other fish
such as european sea bass (Dicentrarchus labrax),
gilthead sea bream (Sparus aurata L.) and rainbow trout
(Alexis et al., 1999; Gabaudan and Verlhac, 2001). The
stress caused by transport for three hours did not affect
muscle ascorbate levels of silverside.
Although the changes in tissue TAA levels after the
stress are not significant, low variations did exist and it
may be that longer periods of transport stress could result
in more significant changes.
Notes
1 Chirostoma estor estor, (Standard length = 14.4 ± 2.03 cm) were collected in lake Pátzcuaro, Michoacán, México in September 2003. Six males
and six females were sacrificed, in order to extract the brain, liver and gonad and take samples of muscle, which were frozen in liquid nitrogen
within 1 minute. Frozen tissues were then stored at -80ºC until ascorbic acid analysis.
2 Frozen tissues were weighed and immediately homogenized in ice-cold buffer (0.2 M Sodium acetate solution, pH 4.8). Samples of each homogenate
(100 mL) were added to three tubes, one for the base line (BL), one for reduced ascorbic acid (AA) and the last for total ascorbic acid (TAA)
quantification. The scheme of sample preparation is shown in Figure 1. Quantification of ascorbic acid concentration in the prepared samples was
carried out by high performance liquid chromatography (HPLC), using the method modified by Papp et al. (1998). Then, dehydro-ascorbic acid
(DHAA) concentrations were determined by the difference between TAA and AA. Ultrapure chemicals used in the analysis were obtained from J.T.
Baker (USA), Fluka (Switzerland), SIGMA (Germany) and Aldrich (USA), and Durapore (Millipore) membrane filters (0.45 mm) were used for the
filtration of buffers and homogenates. A Liquid chromatograph (isocratic single column system, Agillent 1100 series) was used with UV-visible
detector and a Lichrosorb RP-18 column (4.6 x 200mm, Agillent). The mobile phase was 0.04 M Sodium acetate solution, with EDTA (0.05 mM)
and Tetrabutylamonium dihydrogen phosphate (0.5 mM), pH 3.76. The chromatographic conditions were: Flow rate= 0.5 ml/min; Pressure= 65 bar;
Wave length (UV detector)= 254 nm; Temperature= 23ºC; Injection volume= 2 mL. The chromatograms were evaluated with ChemStation Plus
software (Agillent).
3 A further six males and six females (Standard length = 17.7 ± 1.7 cm)
were packed alive in plastic bags with oxygenated freshwater and then
transported for 3 hours at 22-24ºC to the laboratory. This group of fish was
captured at the same time as the control group and so capture stress was the
same for the two groups. After transport, fish were sacrificed and samples
of brain, liver, gonad and muscle were taken and frozen in liquid nitrogen
within 1 minute. Frozen tissues were then stored at -80ºC until ascorbic
acid analysis.
For each tissue, the mean and standard error (S.E.) were calculated
separately for males and females, before and after transport stress. For each
tissue, differences in AA, TAA and DHAA before and after transport and
differences between sexes were analysed using Student's t-test and the
Minitab Statistical software, version 13.32. The accepted level of
significance was 0.05. When data did not have a normal distribution,
statistical analysis were performed using the Mann-Whitney test with a
confidence level of 95%. Because the fish were captured from the wild,
differences in sizes was unavaiodable; in this case the obtained data were
reported as micrograms of ascorbate per gram of tissue (mg/g).
References
Agrawal NK, Mahajan CL (1980). Comparative tissue ascorbic acid
studies in fishes. J Fish Biol 17: 135-141.
Alexis MN, Nengas I, Fountoulaki E, Papoutsi E, Andriopoulou A,
Koutsodimou M, Gabaudan J (1999). Tissue ascorbic acid
levels in European sea bass (Dicentrarchus labrax) and
gilthead sea bream (Sparus aurata L.) fingerlings fed diets
containing different forms of ascorbic acid. Aquaculture, 179:
447-456.
Armijo OA, Sasso YL (1976). Observaciones preliminaries en
acuarios sobre incubación y alevinaje de aterínidos
(Chirostoma spp.) del lago de Pátzcuaro, Michoacán.
Fideicomiso para el desarrollo de la Fauna Acuática (3). 13pp.
Dabrowski K (1991). Ascorbic acid status in high-mountain charr,
Salvelinus alpinus, in relation to the reproductive cycle. Environmental
Biology of Fishes, 31: 213-217.
Dabrowski K, Ciereszko A (1996). The dynamics of gonad growth
and ascorbate status in yellow perch. Aquaculture Research,
27: 539-542.
Gabaudan J, Verlhac V (2001). Critical review of the requirements
of ascorbic acid in cold and cool water fishes (salmonid,
percids, plecoglossids and flatfishes). In: Dabrowski K
(2001). Ascorbic Acid in aquatic organisms. Status and perspectives.
CRC Press. U.S.A. 33-48 pp.
Henrique M, Gomes E, Gouilloou-Coustans M, Oliva-Tele A,
Davies S (1998). Influence of supplementation of practical
diets with vitamin C on growth and response to hypoxic stress
of seabream, Sparus aurata. Aquaculture, 161(1-4): 415-426.
Hilton JW, Cho CY, Slinger SJ (1977). Evaluation of the ascorbic
acid status of rainbow trout (Salmo gairdneri). J Fish Res
Board Can 34: 2207-2210.
Lara VA (1974). Aspectos del cultivo extensivo e intensivo del
pescado blanco de Pátzcuaro Chirostoma estor estor, Jordan
1879. Actas del Simposio sobre Acuicultura en América
Latina. Documentos de Investigación. Montevideo, Uruguay.
Informes de Pesca, FAO, Roma. 159(1): 113-116.
Lee KJ, Dabrowski K (2004). Long-term effects and interactions
of dietary vitamins C and E on growth and reproduction of
yellow perch, Perca flavescens. Aquaculture, 230: 377-389.
Li MH, Wise DJ, Robinson EH (1998). Effect of dietary vitamin C
on weight gain, tissue ascorbate concentration, stress response
and disease resistance of channel catf ish Ictalurus punctatus.
J World Aquacult Soc 29(1): 1-8.
Lim C, Lovell RT (1978). Pathology of the vitamin C deficiency
syndrome in channel catfish (Ictalurus punctatus). J Nutr 108:
1137-1146.
Lim C, Klesius PH, Li MH, Robinson E (2000). Interaction between
dietary levels of iron and vitamin C on growth, immune
response and resistance of channel catf ish (Ictalurus
punctatus) to Edwardsiella ictaluri challenge. Aquaculture,
185: 313-327.
Lygren B, Sveier H, Hjeltnes B, Waagbo R (1999). Examination of
the immunomodulatory properties and the effect on disease
resistance of dietary bovine lactoferrin an vitamin C fed to
Atlantic salmon (Salmo salar) for a short-term period. Fish
Shellfish Immunology, 9: 95-107.
Martínez-Palacios CA, Barriga Tovar E, Taylor JF, Ríos Durán G,
Ross LG (2002a). Effect of temperature on growth and survival
of Chirostoma estor estor, Jordan 1879, monitored using
a simple video technique for remote measurement of length
and mass of larval and juvenile fishes. Aquaculture, 209: 369-377.
Martínez-Palacios CA, Ríos-Durán MG, Campos Memdoza A,
Toledo Cuevas M, Aguilar-Valdez MC, Ross LG (2002b).
Progresos en el cultivo del pescado blanco de Pátzcuaro Chirostoma estor estor. Ciencia Nicolaita, 32: 73-90.
Martínez-Palacios CA, Comas Morte J, Tello-Ballinas JA, Toledo-Cuevas M, Ross LG (2004). The effects of saline environments
on survival and growth of eggs and larvae of Chirostoma
estor estor Jordan 1880 (Pisces: Atherinidae). Aquaculture,
238: 509-522.
Montero D, Marrero M, Izquierdo MS, Robaina L, Vergara JM,
Tort DL (1999). Effect of vitamin E and C dietary supplementation
on some immune parameters of gilthead seabream
(Sparus aurata) juveniles subjected to crowding stress. Aquaculture,
171: 269-278.
Pankhurst N, Van der Kraak G (1997). Effects of stress on reproduction
and growth of fish. Fish Stress and Health in Aquaculture.
Iwama, G., Pickering, A., Sumpter, J., Schreck, C.
(eds.). Cambridge UK. Cambridge University Press. 62: 73-93.
Papp ZS GY, Saroglia M, Jeney ZS, Terova-Saroglia G (1997).
Methodological study on the determination of vitamin C requirements
of different fish species. Reports of intersessional
working parties meetings, 1997. N° 541, suppl., p123.
Papp ZS GY, Saroglia M, Terova G (1998). An improved method
for assay of vitamin C in sample series of fish feed and tissues.
Chromatographia, 48(1/2): 43-47.
Patro UKN, Patnaik BK (1979). Change in ascorbic acid, glycogen
and protein of muscle and brain of Ophiocephalus punctatus Bloch following short-term cold stress. Indian J exp Biol 17:
(521-522).
Robinson EH (1989). Vitamin C in catfish nutrition. Delta Branch
Experiment Station, Mississipi State University. Takeda, USA.
Saborowski R, Koprivnjak JF, Sisak MM, Sahling G, Buchholz F,
Lum KR, Schneider R (1997). Ascorbic acid in the gonads of
North Sea dab (Limanda limanda) during the reproductive
cycle. Canadian Journal of Fisheries and Aquatic Science, 54:
2847-2852.
Sandnes K, Braekkan OR (1981). Ascorbic acid and the reproductive
cycle of ovaries in cod (Gadus morhua). Comparative
Biochemistry and Physiology, 70A: 545-546.
Seymour EA (1981). The effects of powdered carp pituitary on
ovarian development, ovarian ascorbic acid and ovulation in
Carassius carassius L. exposed to various photoperiod and
temperature regimes. J Fish Biol, 19: 675-682.
Thomas P (1984). Influence of some environmental variables on
the ascorbic acid status of mullet, Mugil cephalus L., tissues.
I. Effect of salinity, capture-stress and temperature. J Fish Biol,
25: 711-720.
Wee KL (1996). The role of vitamin C in aquaculture. Proceedings
of the seminar on fisheries multibillion dollar industry held
at Madras, India. August 17-19, 1995. Krishnamoorthi, B.,
Krishnamoorthi, K. Meenakshisundaram, P., K. Nayar (eds.).
Madras India Aquaculture Foundation of India. 50-62pp.
Received on January 29, 2005.
Accepted on September 15, 2005.




