[article pii="nd"
doctopic="oa" language="es" ccode="FVET"
status="1" version="4.0" type="ilus tab"
order="02" seccode="invet060" sponsor="nd"
stitle="InVet" volid="18" issueno="2"
dateiso="20161200" fpage="307" lpage="316"
issn="1668-3498"][ign]ARTÍCULO
DE INVESTIGACIÓN
[/ign][front][titlegrp][title
language="en"]Effect of xanthine-xanthine oxidase-catalase
system on bovine sperm oxidative metabolism during capacitation induction[/title][/titlegrp][ign]
[/ign][authgrp][author role="nd" rid="a01
a03"][surname]Casas[/surname][ign], [/ign][fname]E.[/fname][ign]1,3[/ign][/author][ign]; [/ign][author
role="nd" rid="a01 a03"][surname]Marquínez[/surname][ign], [/ign][fname]A.[/fname][ign]1,3[/ign][/author][ign]; [/ign][author
role="nd" rid="a01 a02 a03"][surname]Córdoba[/surname][ign], [/ign][fname]M.[/fname][ign]1,2,3[/ign][/author][/authgrp][ign]
1[/ign][aff
id="a01" orgname="UBA" orgdiv1="Facultad de Ciencias
Veterinarias" orgdiv2="Instituto de Investigación y Tecnología en
Reproducción Animal"]Instituto de Investigación y Tecnología en Reproducción
Animal (FVET-UBA)[/aff][ign]
2[/ign][aff id="a02" orgname="UBA" orgdiv1="CONICET"]Unidad ejecutora de
Investigaciones en Producción Animal UBA-CONICET.[/aff][ign]
3[/ign][aff id="a03" orgname="Universidad de Buenos
Aires" orgdiv1="Facultad de Ciencias Veterinarias"
orgdiv2="Cátedra de Química Biológica"][ign]Universidad de [/ign][city]Buenos Aires[/city][ign], Facultad de
Ciencias Veterinarias, Cátedra de Química Biológica.[/ign][/aff][ign]
Correspondencia
e-mail: Mariana Córdoba mcordoba@fvet.uba.ar
[/ign][bibcom][hist][ign]Recibido: [/ign][received
dateiso="20160920"]20/09/2016[/received][ign]
Aceptado: [/ign][accepted dateiso="20170107"]01/07/2017[/accepted][/hist][ign]
Summary
[/ign][abstract language="en"]The aim of this work
was to study the effect of heparin or xanthine-xanthine oxidase-catalase system
(superoxide anion generating system) on the induction of capacitation, sperm
oxygen uptake, the variation in the activity of creatine kinase-B in bovine
spermatozoa and tyrosine kinase and protein kinase C regulation with heparin.
Genistein and GF 109203X were used as specific inhibitors of tyrosine kinase
and protein kinase C, respectively. Creatine kinase-B activity and lipid
peroxidation were registered spectrophotometrically. Oxygen uptake was measured
polarographically. Capacitation was evaluated by chlortetracycline technique
and viability by trypan blue stain. Data were analyzed by ANOVA and Tukey test.
Lipid peroxidation was modified by treatments (P<0.05). In sperm capacitated
with heparin the addition of genistein or GF 109203X, provoked a decrease in
creatine kinase-B activity, oxygen uptake and capacitation rates (P<0.05).
In the presence of xanthine-xanthine oxidase/catalase system, capacitation and
oxygen uptake presented a significant decrease at 15 and 45 min compared to
heparin treatment but creatine kinase-B activity only presented a decrease at
45 min (P<0.05). To conclude, in cryopreserved bovine spermatozoa heparin
capacitation is related to a respiratory burst and a decrease in creatine
kinase-B activity, both processes dependent of tyrosine kinase and protein
kinase C regulation. Superoxide anion induces sperm capacitation and lipid
peroxidation, which can provoke mitochondrial membrane alteration, depending on
the incubation time. This deleterious effect may modify the respiratory chain
function with a concomitant reduction in creatine kinase-B activity, an enzyme
of the cytosol- mitochondria shuttle, not allowing the sperm to reach the
energetic state necessary to achieve fertilizing capability.[/abstract][ign]
Key
words:
[/ign][keygrp scheme="nd"][keyword
type="m" language="en"]Capacitation[/keyword][ign]; [/ign][keyword
type="m" language="en"]Metabolism[/keyword][ign]; [/ign][keyword
type="m" language="en"]Bovine spermatozoa;[/keyword][ign] [/ign][keyword
type="m" language="en"]Superoxide anion[/keyword][/keygrp][ign].
[/ign][title
language="es"]Efecto del sistema xanthine-xanthine oxidase-catalasa en
el metabolismo oxidante de esperma bovina durante la inducción de la
capacitación[/title][ign]
Resumen
[/ign][abstract
language="es"]El
objetivo de este trabajo fue estudiar el efecto de heparina o el sistema
xantina-xantino oxidase catalase (sistema generador de anión superóxido) en la
inducción de la capacitación, consumo de oxígeno, la variación de la actividad
de creatina quinasa B, y su regulación por tirosina quinasa y proteína quinasa
C en presencia de heparina. Genisteina y GF 109203X fueron usados como
inhibidores específicos de la tirosina quinasa y proteína quinasa C
respectivamente. La actividad de creatina quinasa-B y la lipoperoxidación fueron
registradas espectrofotométricamente. El consumo de oxígeno fue
polarográficamente. Se evaluó viabilidad e integridad acrosomal por la
coloración de azul tripán y microscopia óptica de Contraste Diferencial
Interferencial y la capacitación por la coloración epifluorescente de
Clorotetraciclina (CTC). Los datos fueron analizados por ANOVA y test de Tukey
(P<0,05). La lipoperoxidación se modificó debido a los tratamientos
(P<0.05). En la capacitación espermática con heparina la adición de
genisteina o GF 109203X provocó una disminución de la actividad creatina
quinasa B, toma de oxígeno y el porcentaje de capacitación (P<0.05). En la
presencia del sistema xantina-xantino oxidase catalase, la capacitación y el
consumo de oxígeno presentaron una disminución significativa a los 15 y 45
minutos, comparados con el valor del tratamiento con heparina, pero la
actividad de la creatina quinasa B sólo disminuyó a los 45 minutos (P<0.05).
En conclusión, la capacitación con heparina está vinculada con burst
respiratorio y con la disminución de la creatina quinasa B, ambos procesos
dependientes de la proteína quinasa C. El anión superóxido induce la
capacitación espermática y la lipoperoxidación lo cual provoca una alteración
dependiente del tiempo de incubación. Este efecto deletéreo puede modificar el
funcionamiento de la cadena respiratoria con la concomitante reducción de la
actividad de la creatina quinasa B, enzima de la lanzadera
citosólica-mitocondrial, impidiendo que el espermatozoide tenga la carga
energética necesaria para adquirir su capacidad fertilizante.[/abstract][ign]
Palabras
clave:
[/ign][keygrp scheme="nd"][keyword type="m"
language="es"]Capacitación[/keyword][ign]; [/ign][keyword
type="m" language="es"]Metabolismo[/keyword][ign]; [/ign][keyword
type="m" language="es"]Espermatozoide bovino[/keyword][ign]; [/ign][keyword
type="m" language="es"]Anión superóxido[/keyword][/keygrp][/bibcom][ign].[/ign][/front][ign]
[/ign][body]Introduction
Capacitation is a
process that prepares spermatozoa for acquiring fertilizing capability.
Extensive research has been developed in order to elucidate the protein
phosphorylation event during sperm capacitation and acrosome reaction wherein
three pathways are mainly involved: cAMP/Protein kinase A (PKA), receptor tyrosine
kinases, and non-receptor protein tyrosine kinases 1,2,3,4. It is known that the
phosphorylation of sperm proteins is an important aspect of capacitation and has
been associated with sperm hypermotility, ZP binding and acrosome reaction 5,6. In mammalian spermatozoa,
capacitation is dependent on tyrosine kinase and protein kinase C activities
and the variation of intracellular calcium concentration 7,8,9. During the course of
capacitation and fertilization, the main tyrosine- phosphorylated proteins are
located in the flagellum, while they are less abundant in the sperm head10.
Protein kinase C (PKC) roles in spermatozoa have been widely investigated in
many species.
This enzyme participates in many sperm functions, i.e. capacitation, acrosome
reaction, and sperm motility11. It has been demonstrated that PKA activation
leads to PKC inhibition11 and phosphatidylinositol 3-kinase
(PI3K) activation, allowing actin polymerization12. Moreover, a role for PI3K
in sperm capacitation and acrosome reaction has been described13.
Many of the intracelullar signals that occur during capacitation and acrosome
reaction are regulated by the redox state of the sperm14. Futhermore, it was
demonstrated that the induction of capacitation, acrosome reaction and
hyperactivation are associated with superoxide anion and hydrogen peroxide 15,16,17. Xanthinexanthine oxidase
/ catalase system may be used to generate both types of ROS in bovine
cryopreserved spermatozoa in vitro18.
The excessive production of superoxide and hydrogen peroxide in the spermatozoa
can initiate peroxidation. As a consequence, spermatozoa that have suffered
oxidative stress would be characterized by the accumulation of lipid
hydroperoxides in their plasma membranes, which would be relatively stable
until induced to decompose and liberate malondialdehyde upon addition of the
ferrous ion promoter used in the TBA assay. The build-up of lipid
hydroperoxides in the sperm plasma membrane clearly had a profound negative
impact on sperm function 19.
There is evidence that capacitation is part of an oxidative process. In bovine
spermatozoa, heparin induces capacitation and this process involves NADPH
oxidase activity, intracellular calcium increase and a respiratory burst 17,
20. It was also suggested that a certain ROS level and high respiratory
activity in heparin-treated spermatozoa may be the factors associated with the
alteration of the phosphocreatine/creatine shuttle function8.
It has been demonstrated that in cryopreserved bovine spermatozoa, mitochondria
preserve respiratory coupling for ATP synthesis for sperm function. Creatine kinase
isoenzymes have been involved in the metabolism of bovine spermatozoa21 and have been found to be
specifically located at sites which have a high demand and production of energy22.
In order to contribute to increase the knowledge of the different mechanisms
involved in capacitation, the aim of the present work was to determine the
influence of xanthine-xanthine oxidase -catalase system in this process,
studying tyrosine kinase and PKC regulation, sperm oxygen uptake and creatine
kinase-B activity variation, as an enzyme of the phosphocreatine/ creatine
shuttle, in cryopreserved bovine spermatozoa.
Materials and
methods
Semen
collection and freezing
Semen was collected by an artificial vagina from four Holstein bulls (4 to 5
years old) of proven fertility. These bulls were routinely used to provide
semen for artificial insemination and they were maintained under uniform
nutritional and management conditions throughout the study. The ejaculates were
pooled and diluted in a buffer containing 0.20 M Tris, 0.06 M citrate, 0.13 M
glycine, 0.06 M fructose, 20% egg yolk and 7% glycerol at a 2:1 ratio. A slow
cooling curve at 5şC (1şC/min) was performed, and semen was then equilibrated
at 5şC for a further 90 min and then preserved at -196şC in liquid nitrogen21.
Sperm
suspension
Pooled frozen semen samples collected from the four bulls were thawed for 10
minutes at 37şC in TALP medium (99 mM NaCl, 3.1 mM KCl, 0.35 mM NaH2PO4.H2O,
1.1 mM MgCl2. H2O, 25 mM NaHCO3, 1 mM sodium pyruvate, 21.6 mM sodium lactate,
and 10 mM HEPES) without bovine serum albumin (BSA) or calcium. The vigor score
and the percentage of cells with progressive motility were evaluated at 38şC
using light microscopy. Samples with 60% average progressive motility and a 3
to 4 (scale 0 to 5) vigor score of were considered suitable for the
experiments. Sperm concentration was determined by hematocytometry in a
Neubauer chamber. Subsequent to the evaluation of motility, the samples were
centrifuged (600 x g for 5 minutes) and resuspended in TALP with 2.1 mM calcium
chloride and 6 mg/mL BSA. In order to induce capacitation, this sample
suspension was incubated at 38şC in the presence of no additional compounds
(control), 60 Ľg/mL heparin 15 min 20,23 or 0.5 mM xanthine ; 0,05
mUI/mL, xanthine oxidase 100 mg/mL, catalase24 at 15 and 45 min
incubation.
Sperm viability
and acrosome integrity
An aliquot of the sperm suspension taken from each different treatment was
incubated with an equal volume of 0.25 % (w/v) Trypan blue in TALP for 15
minutes at 38şC, centrifuged at 600 g for 10 minutes to remove excess stain and
then fixed with 5% formaldehyde in PBS. Acrosomes obtained from the different
sperm samples stained with Trypan blue were evaluated by differential
interference contrast (DIC) microscopy (200 spermatozoa per sample) in order to
assess acrosome integrity in live and dead spermatozoa. To account for
spontaneous damage, the value obtained at time zero was subtracted from the
values obtained after each treatment17.
Determination
of sperm capacitation using chlortetracycline technique
The percentages of capacitation for the different treatments were determined by
the epifluorescence chlortetracycline technique. To account for the percentage
of spermatozoa with the capacitated pattern induced by freezing and thawing,
the percentage of capacitated spermatozoa obtained at zero time was subtracted
from results after incubation (for each treatment group) 25.
Preparation of
extracts for the measurement of enzymatic activity
Suspensions of capacitated and control spermatozoa were centrifuged at 2000 rpm
for 5 min. The pellets were resuspended in distilled water and an aliquot (20
µL) was used to determine sperm concentration. The final sperm concentration
was adjusted to 1.0 x 108 spermatozoa/mL. The samples were frozen for 2 h at
-20şC, thawed at room temperature, refrozen (45 min) and thawed again at room
temperature. Samples were then centrifuged (17000 rpm for 20 min at 4şC).
Supernatants were used to determine the enzymatic activity of creatine
kinase-B. All replicates were processed by the same standard operating
procedure to obtain enzymatic extracts; therefore, protein recovery was
equivalent in all cases8.
Determination
of creatine kinase-B activity
Suspensions of capacitated spermatozoa and controls were centrifuged at 2000 rpm
for 5 min. The pellets were resuspended in distilled water, and then treated as
described in the section above. Supernatants were used to determine the
enzymatic activities of creatine kinase-B at a final sperm concentration of 1.0
x 108 spermatozoa/ mL in a measuring cuvette. Enzymatic activity was measured
using 30 mM creatine phosphate as substrate, 2 mM ADP, 20 mM glucose, 2 mM
NADP, 2500 U/L hexoquinase, 2000 U/L Glucose-6-phosphate-dehydrogenase, 10 mM
Magnesium acetate, 5 mM AMP, 10 mM di-(adenosine 5 )-phentaphophate and 20
mmol/L N-acetilcysteine, 200 U/L 6-phosphoglucolactonase, 400 U/L
6-phosphogluconate-dehydrogenase and 100 mM imidazol buffer and concentrations
of monoclonal antibody capable of inhibiting 1000 U/L CKM or 2000U/L CKM. The activity
was determined spectrophotometrically at 340 nm for 5 min by measuring the
reduction of NADP. Enzyme activity was expressed in U/108 spermatozoa. One
enzymatic unit of creatine kinase-B was defined as the enzyme quantity which
catalyses the transfer of 1.0 mmol of phosphate from phosphocreatine to ADP per
minute at pH 7.4 and 37şC26,27.
Oxygen
consumption
Respiration of the sperm suspension was measured polarographically at 38°C with
an oxygenelectrode modified Clark type and an Instech Laboratories
(Philadelphia, PA, USA) oxygraph. The reaction cell had a 0.6 mL capacity and
measured rapid changes in the oxygen consumption rate by the cell. The
measurement cuvette was kept at 38°C so that the diffusion of atmospheric oxygen
toward the solution could be negligible compared with the rate of oxygen uptake
recorded with constant stirring. For evaluation of sperm respiration during
capacitation, the final concentration in the cuvette was 1.0 x 108 spermatozoa/mL.
Carbanylcyanide- m-chloro phenylhydrazone (CCCP; 0.42 mM) was used as a
specific uncoupler of the respiratory chain and oxygen uptake was expressed as
mLO2/h/108
spermatozoa17.
Protein kinase
C inhibition
The GF 109203X, a specific inhibitor of protein kinase C was used at a 100 nM
concentration28. The inhibitor and heparin or
xanthine/xanthine-oxidase/catalase system were simultaneously added to the
sperm suspension.
Tyrosine kinase
inhibition Genistein (150 mM) was used as specific tyrosine kinase inhibitor29. It was added along with
heparin or xanthine/xanthine-oxidase/ catalase system to the sperm suspensions
and then they were incubated at 38şC during 15 min.
Determination
of Lipid peroxidation
Samples were incubated to enhanced lipid peroxidation at 37°C in the presence
of 0.5 mM sodium ascorbate and 0.11 mM ferrorus sulfate for 2 h. Lipid
peroxidation was evaluated as the mean of 2-thiobarbituric acid (TBARS) assay16.
Statistical
analysis
Differences of percentages of capacitated spermatozoa, sperm viability, CPK-B
activities, oxygen consumption and lipid peroxidation levels between treatments
were determined by ANOVA; a Tukey test was used as a post-ANOVA analysis to
compare means (STATISTIX 7. 2000, Analytical Software for Windows, Version 7.0;
Analytical Software, Tallahassee, Florida, United States). For all analyses
P<0.05 was regarded as significant.
Results
In the present
study, the influence of tyrosine kinase (genistein) or protein kinase C (GF
109203X) inhibitors on heparin capacitation induction as well as their
respective oxygen uptake rate were evaluated on bovine spermatozoa. The results
indicate that in spermatozoa treated with heparin, the presence of genistein or
GF 109203X provoked a significant decrease in the capacitation percentage and
sperm oxygen uptake compared to the values obtained with heparin treated
samples (P < 0.05) Table 1. No differences were observed in the
sperm viability with all treatments (P>0.05)
Table 1.
Capacitation and oxygen uptake in bovine spermatozoa

Bovine
spermatozoa were capacitated with heparin 60 Ľg/mL heparin or 0,5
mM xanthine -0,05 mUI/mL- xanthine oxidase (X-XO/Catalase)- 100 mg/mL catalase.
GF 109203X (100 nM) (GF) or Genistein (150 mM) inhibitors of protein kinase C
and tyrosine kinase respectively, were added simultaneosly with heparin. Data
are expressed as means ± SD, n=7. Different superscripts and symbols indicate
significant differences between treatments (P < 0.05).
In sperm
suspensions treated with xanthine -xanthine oxidase- catalase system, in both
incubation times, spermatozoa were capacitated showing a lower respiration rate
respect to heparin treated samples. Furthermore, in these conditions the capacitation
decreased (P < 0.05) Table 1.
A decrease in
creatine kinase-B activity level was observed in the presence of heparin or
xanthine -xanthine oxidase - catalase system in both incubation times respect
to sperm control (P < 0.05) Figure 1.
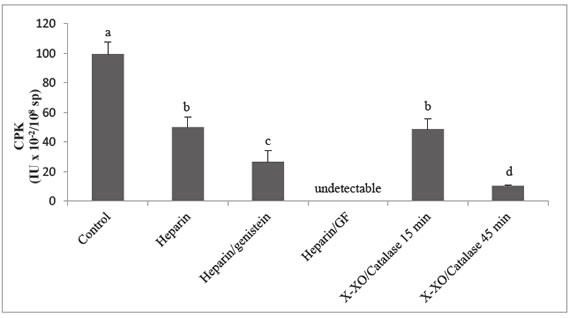
Figure
1. Creatine
kinase-B activity variation
Data are expressed as means ± SD, n=7.
Different superscripts and symbols indicate significant differences between
treatments (P<0.05). GF 109203X (100 nM) (GF) or Genistein (150 mM) inhibitors
of protein kinase C and tyrosine kinase respectively, were added simultaneosly
with heparin. Xanthine-xanthine oxidase-catalase system (X-XO/Catalase) or
heparin was used as capacitation inductors. The activity was determined
spectrophotometrically at 340 nm over 5 min by measuring the reduction of NADP.
Enzyme activity was expressed in International units per 108
spermatozoa (IU x 10-2/108 sp).
The addition of
genistein or GF 109203X, in order to demonstrate tyrosine kinase and protein
kinase C modulation respectively on creatine kinase-B activity during heparin
treatment, caused a significant inhibition and an undetectable level of
creatine kinase-B activity respectively (P < 0.05) Figure 1.
A significant increase in lipid peroxidation was detected by the addition of
xanthine xanthine oxidase/catalase at 45 min respect to controls, heparin
treatments and xanthine-xanthine oxidase/catalase addition at 15 min (P <
0.05) Figure 2.
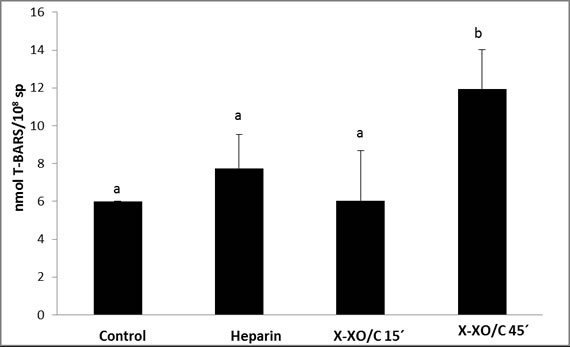
Figure
2. Lipid
peroxidation variations in the presence of heparin or xanthine-xanthine
oxidase-catalase system
Sperm samples were incubated to
enhance lipid peroxidation at 37°C. Lipid peroxidation level was determined as
means of 2-thiobarbituric acid (TBARS) assay. Data are expressed as means ± SD,
n=7. Different superscripts and symbols indicate significant differences
between treatments (P<0.05). Xanthine-xanthine oxidase-catalase system
(X-XO/C).
Discussion
Extensive research
has started to elucidate the protein phosphorylation event during sperm
capacitation and acrosome reaction, wherein three pathways are mainly involved:
cAMP/ PKA, receptor tyrosine kinases and non-receptor protein tyrosine kinases 1,2 which finally produce an
increase in the phosphorylation of Tyr residues 3,4.
In cryopreserved bovine spermatozoa, pyruvate and lactate are sources of
oxidative energy. Oxidative respiration provides the most efficient ATP
generation pathway, while the major site of ATP consumption in the spermatozoa
is the dynein ATPase, which is associated with the energy consumption in the
flagellum 30.
It is known that the phosphorylation of sperm proteins is an important aspect of
capacitation and has been associated with sperm hypermotility, ZP binding and
acrosome reaction5,6. In several mammalian species, all
the regulatory mechanisms of this event have not been fully elucidated yet.
Therefore, it is important to investigate how tyrosine kinase and protein
kinase C (PKC) would be involved in heparin capacitation induction through
sperm oxygen uptake and enzymatic activity to know the energy sources related
to intracellular signals activation.
In this study, GF 109203X or genistein (PKC and tyrosine kinase inhibitors
respectively) provoked a decrease in capacitation induction and in sperm oxygen
uptake in heparin treated spermatozoa, so we infer that both kinases may
modulate gamete oxidative metabolism related to mitochondrial function, which
may be involved in heparin sperm capacitation signals. Identified
Tyr-phosphorylated proteins in human sperm include ion channels, metabolic
enzymes and structural proteins 31,32. In cryopreserved bovine
spermatozoa was studied that tyrosine kinases including SRC-isoform modulate
capacitation, where the intracellular calcium variation may be a crucial point
related to tyrosine phosphorylation 33.
Genistein inhibition of intracellular heparin mechanisms provoke metabolic
changes that result in an energetic charge which is not enough to supply the
conditions for glycosaminoglycan induction, inferred by a low oxygen
consumption which indicates a decrease in oxidative phosphorylation as a
potential source of ATP. This finding agrees with our previous research, which
proposes that the heparin induced respiratory burst was produced mainly by
mitochondrial activity, the main energy source for the sperm 17. Another energy source for
the sperm, the phosphocreatine/creatine shuttle, has been vinculated with
oxidative phosphorylation and its regulation through cellular redox state
variation has been proposed 34,8. Creatine kinase (CPK) isoenzymes,
specifically located at sites of energy demand and production, are linked by a
phosphocreatine/creatine circuit 22. Our data suggest that the heparin
metabolic pathways including CPK shuttle are modulated by PKC and tyrosine
kinase because of the significant CPK-B activity decrease, an enzyme involved
in supporting cytosolic ATP, when both regulatory kinases were blocked. So
CPK-B activity is required to supply energy for heparin induced capacitation in
bovine spermatozoa.
Furthermore, CPK-B activity in heparin induced capacitation depends mainly on
PKC modulation confirmed by the complete inhibition by GF 109203X, but CPK-B
activity is also modulated (50%) by the activation of tyrosine kinase.
As explained above, the major energy requirement in the sperm is related to
hypermotility. Preliminary studies on sperm PKC subspecies distribution
revealed that they are involved in sperm flagellar motility in human 35 and in bovine spermatozoa 36. It is also noteworthy
that during the course of capacitation and fertilization, the main
tyrosine-phosphorylated proteins are located in the flagellum 10 and it has been related to
mammalian hyperactivated sperm motility 37.
CPK-B activity and oxygen uptake variations in heparin capacitated spermatozoa,
due to the localization and compartmentalization of tyrosine kinase and PKC
isoforms, may be related to the phosphorylation of substrates in different
sperm regions to regulate physiological functions connected with energy
sources. So these results suggest that PKC and tyrosine kinase would be related
with signaling pathways that maintain both CPK activity level and an active mitochondrial
oxidative phosphorylation as energy sources for heparin induced capacitation in
the bovine sperm.
The xanthine-xanthine oxidase-catalase system generates superoxide anion that
can capacitate bovine sperm 18 and it has been demonstrated that it
may improve sperm metabolism and oocyte fertilization in the mouse 38. Data suggest that a lower
sperm oxygen consumption induced by xanthine-xanthine oxidase-catalase system
compared to respiratory burst produced by heparin, allowing a lower
capacitation rate due to the energetic state generated by superoxide anion. As
regards creatine kinase-B in the presence of xanthinexanthine oxidase-catalase
system, the increase in the incubation time had a negative effect in this
enzyme activity, although the capacitation was similar with both incubation
times. These results may be due to the deleterious effect of reactive oxygen
species (ROS). It is known that despite their potential deleterious effect, ROS
at low and controlled levels participate in cell signaling events in sperm
physiology, allowing them to acquire fertilizing capability. In fact, in human
and bovine sperm, hyperactivation and capacitation are triggered by ROS 18,8,39.
According to Koufen the decrease in mitochondrial CPK activity is caused by an
increase in ROS 34, which was detected in the midpiece
of sea urchin40 and human spermatozoa 41. The increase in ROS
levels produces capacitation decrease in bovine sperm 8. In relation with these
observations, our data suggest that more incubation time (45 min) with
xanthine-xanthine oxidase-catalase system produces a toxic effect on sperm,
confirmed by the increase in lipid peroxidation that caused an alteration in
oxygen consumption and CPK-B activity respect to heparin induction. Lipid
peroxidation can provoke alterations in the plasma and mitochondrial membranes
that may modify the respiratory chain function, with the concomitant reduction
in CPK activity, not allowing the bovine sperm to capacitate because of a low
energy charge for the process. In accordance with these changes in sperm
metabolism, it has been demonstrated that heparin treatment is better than
xanthinexanthine oxidase-catalase system to induce highest rates early cleavage
in bovine oocytes 42,43.
In conclusion, in cryopreserved bovine spermatozoa heparin capacitation is
related to a respiratory burst and a decrease in CPK-B activity, both processes
dependent of tyrosine kinase and protein kinase C regulation. In the conditions
of this study, sperm capacitation and lipid peroxidation depending on the
incubation time can provoke mitochondrial membrane alteration. This deleterious
effect may modify the respiratory chain function, with the concomitant
reduction in creatine kinase shuttle, not allowing the sperm to reach the
energetic state required to achieve fertilizing capability.[/body][ign]
[/ign][back][ign]References[/ign][other
standard="other" count="43"][ign]
[/ign][ocitat][no]1[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Grasa [/surname][fname]P[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Colas [/surname][fname]C[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Gallego [/surname][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Monteagudo [/surname][fname]L[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Muino- Blanco[/surname][ign] [/ign][fname]T[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Cebrian-Perez[/surname][ign] [/ign][fname]J[/fname][/oauthor][ign].[/ign][title
language="en"]Changes in content and localization of
proteins phosphorylated at tyrosine, serine and threonine residues during ram
sperm capacitation and acrosome reaction[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Reproduction[/stitle][ign] ([/ign][city]Cambridge[/city][ign], [/ign][country]England[/country][ign]). [/ign][date
dateiso="20090000"]2009[/date][ign]; [/ign][volid]137[/volid][ign]: [/ign][pages]655-67[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]2[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Garbi [/surname][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Rubinstein [/surname][fname]S[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Lax [/surname][fname]Y[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Breitbart [/surname][fname]H[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Activation of protein kinase C in the
lysophosphatidic acid-induced bovine sperm acrosome reaction and phospholipase
D1 regulation[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Biology of
reproduction[/stitle][ign]. [/ign][date
dateiso="20000000"]2000[/date][ign]; [/ign][volid]63[/volid][ign]: [/ign][pages]1271-7[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]3[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Salicioni [/surname][fname]AM[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Platt [/surname][fname]MD[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Wertheimer [/surname][fname]EV[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Arcelay [/surname][fname]E[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Allaire [/surname][fname]A[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Sosnik [/surname][fname]J[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Visconti[/surname][ign] [/ign][fname]PE[/fname][/oauthor][ign]. [/ign][title
language="en"]Signalling pathways involved in sperm
capacitation[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Soc Reprod
Fertil
Suppl.[/stitle][ign] [/ign][date
dateiso="20070000"]2007[/date][ign]; [/ign][volid]65[/volid][ign]:[/ign][pages]245-59[/pages][/oiserial][/ocitat][ign]
[/ign][ocitat][no]4[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Visconti [/surname][fname]PE[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Understanding the molecular basis of sperm
capacitation through kinase design[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Proc Natl
Acad Sci [/stitle][country]USA[/country][ign]. [/ign][date
dateiso="20090000"]2009[/date][ign]; [/ign][volid]106[/volid][ign]:[/ign][pages]667-8[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]5[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Mor [/surname][fname]V[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Das [/surname][fname]T[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Bhattacharjee[/surname][ign] [/ign][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Chatterjee [/surname][fname]T[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Protein tyrosine phosphorylation of a
heparin-binding sperm membrane mitogen (HBSM) is associated with capacitation
and acrosome reaction[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Biochemical
and Biophysical Research Communication[/stitle][ign]. [/ign][date
dateiso="20070000"]2007[/date][ign]; [/ign][volid]352[/volid][ign]: [/ign][pages]404-9[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]6[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Urner [/surname][fname]F[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Sakkas [/surname][fname]D[/fname][/oauthor][ign]. [/ign][title
language="en"]Protein phosphorylation in mammalian
spermatozoa[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Reproduction
[/stitle][ign]([/ign][city]Cambridge[/city][ign], [/ign][country]England[/country][ign]). [/ign][date
dateiso="20030000"]2003[/date][ign]; [/ign][volid]125[/volid][ign]: [/ign][pages]17-26[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]7[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Naz [/surname][fname]RK[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Rajesh[/surname][ign] [/ign][fname]PB[/fname][/oauthor][ign]. [/ign][title
language="en"]Role of tyrosine phosphorylation in sperm
capacitation / acrosome reaction[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Reprod
Biol Endocrinol[/stitle][ign]. [/ign][date
dateiso="20040000"]2004[/date][ign]; [/ign][volid]2[/volid][ign]: [/ign][pages]75-88[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]8[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Córdoba [/surname][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Pintos [/surname][fname]L[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]MT[/fname][/oauthor][ign]. [/ign][title
language="en"]Variations in creatine kinase activity and
reactive oxygen species levels are involved in capacitation of bovine
spermatozoa[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Andrologia[/stitle][ign]. [/ign][date
dateiso="20080000"]2008[/date][ign]; [/ign][volid]40[/volid][ign]: [/ign][pages]370-76[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]9[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Breitbart [/surname][fname]H[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Etkovitz[/surname][ign] [/ign][fname]N[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Role and regulation of EGFR in actin
remodeling in sperm capacitation and the acrosome reaction[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Asian
Journal of Andrology[/stitle][ign]. [/ign][date
dateiso="20110000"]2011[/date][ign]; [/ign][volid]13[/volid][ign]:[/ign][pages]106-10[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]10[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Pawson [/surname][fname]T[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Specificity in signal transduction[/title][ign]: [/ign][subtitle]from
phosphotyrosine-SH2 domain interactions to complex cellular systems[/subtitle][/ocontrib][ign]. [/ign][oiserial][stitle]Cell[/stitle][ign]. [/ign][date
dateiso="20040000"]2004[/date][ign]; [/ign][volid]116[/volid][ign]: [/ign][pages]191-203[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]11[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Cohen[/surname][ign] [/ign][fname]G[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Rubinstein[/surname][ign] [/ign][fname]S[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Gur[/surname][ign] [/ign][fname]Y[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Breitbart [/surname][fname]H[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Crosstalk between protein kinase A and C
regulates phospholipase D and F-actin formation during sperm capacitation[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Developmental
Biology.[/stitle][ign] [/ign][date
dateiso="20040000"]2004[/date][ign]; [/ign][volid]267[/volid][ign]: [/ign][pages]230-41[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]12[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Etkovitz [/surname][fname]N[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Rubinstein [/surname][fname]S[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Daniel [/surname][fname]L[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Breitbart [/surname][fname]H.[/fname][/oauthor][ign] [/ign][title
language="en"]Role of PI3-kinase and PI4-kinase in actin polymerization
during bovine sperm capacitation[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Biology of
Reproduction[/stitle][ign]. [/ign][date
dateiso="20070000"]2007[/date][ign]; [/ign][volid]77[/volid][ign]: [/ign][pages]263-73[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]13[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Jungnickel [/surname][fname]MK[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Sutton[/surname][ign] [/ign][fname]KA[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Wang [/surname][fname]Y[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Florman [/surname][fname]HM[/fname][/oauthor][ign]. [/ign][title
language="en"]Phosphoinositide - dependent pathways in
mouse sperm are regulated by egg ZP3 and drive the acrosome reaction[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Developmental
Biology[/stitle][ign]. [/ign][date dateiso="20070000"]2007[/date][ign]; [/ign][volid]304[/volid][ign]: [/ign][pages]116-26[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]14[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Hancok [/surname][fname]J[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Desikan [/surname][fname]R[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Neill [/surname][fname]S[/fname][/oauthor][ign]. [/ign][title
language="en"]Role of oxygen species in cell signaling
pathways[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Biochem
Society Transaction[/stitle][ign]. [/ign][date
dateiso="20010000"]2001[/date][ign]; [/ign][volid]29[/volid][ign]: [/ign][pages]345-50[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]15[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]de Lamirande[/surname][ign] [/ign][fname]E[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Gagnon [/surname][fname]C[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]A positive role for the superoxide anion in
triggering hyperactivation and capacitation of human spermatozoa[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Int J
Androl[/stitle][ign]. [/ign][date
dateiso="19930000"]1993[/date][ign]; [/ign][volid]16[/volid][ign]: [/ign][pages]21-25[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]16[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Aitken [/surname][fname]RJ[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Fisher [/surname][fname]HM[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Fulton[/surname][ign] [/ign][fname]N[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Gomez[/surname][ign] [/ign][fname]E[/fname][/oauthor][ign]. [/ign][oauthor
role="nd"][surname]Knox[/surname][ign] [/ign][fname]W[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Lewis[/surname][ign] [/ign][fname]B[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Irvin [/surname][fname]S[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Reactive oxygen species generation by human
spermatozoa is induced by exogenous NADPH and inhibited by the flavoprotein
inhibitor dyphenylene iodonium and quinacrine[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Mol Reprod
Dev.[/stitle][ign] [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]47[/volid][ign]: [/ign][pages]468-82[/pages][/oiserial][ign].[/ign][/ocitat][ign].
[/ign][ocitat][no]17[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Córdoba [/surname][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Mora [/surname][fname]N[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]MT.[/fname][/oauthor][ign] [/ign][title
language="en"]Respiratory burst and NAD(P)H oxidase
activity are involved in capacitation of cryopreserved bovine spermatozoa[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Theriogenology[/stitle][ign]. [/ign][date
dateiso="20060000"]2006[/date][ign]; [/ign][volid]65[/volid][ign]: [/ign][pages]882-92[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]18[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]O'Flaherty [/surname][fname]CM[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Beorlegui [/surname][fname]NB[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Beconi [/surname][fname]MT[/fname][/oauthor][ign]. [/ign][title
language="en"]Reactive oxygen species requirements for
bovine sperm capacitation and acrosome reaction[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Theriogenology[/stitle][ign]. [/ign][date
dateiso="19990000"]1999[/date][ign]; [/ign][volid]52[/volid][ign]: [/ign][pages]289-301[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]19[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Aitken [/surname][fname]RJ[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Harkiss[/surname][ign] [/ign][fname]D[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Buckingham[/surname][ign] [/ign][fname]D[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Relationship between iron-catalysed lipid
peroxidation potential and human sperm function[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]J
Reproduction and Fertility.[/stitle][ign] [/ign][date
dateiso="19930000"]1993[/date][ign]; [/ign][volid]98[/volid][ign]: [/ign][pages]257-65[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]20[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Córdoba[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Santa-Coloma[/surname][ign] [/ign][fname]TA[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Beorlegui[/surname][ign] [/ign][fname]NB[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]MT[/fname][/oauthor][ign]. [/ign][title
language="en"]Intracellular calcium variation in
heparin-capacitated bovine sperm[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Biochemistry
and Molecular Biology International[/stitle][ign]. [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]41[/volid][ign]: [/ign][pages]725-33[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]21[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Córdoba[/surname][ign] [/ign][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Pintos[/surname][ign] [/ign][fname]L[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]MT[/fname][/oauthor][ign]. [/ign][title
language="en"]Heparin and quercetin generate differential
metabolic pathways that involve transferases and LDH dehydrogenase in
cryopreserved bovine spermatozoa[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Theriogenology[/stitle][ign]. [/ign][date
dateiso="20070000"]2007[/date][ign]; [/ign][volid]67[/volid][ign]: [/ign][pages]64[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]22[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Wallimann[/surname][ign] [/ign][fname]T[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Dolder[/surname][ign] [/ign][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Schattner[/surname][ign] [/ign][fname]U[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Eder[/surname][ign] [/ign][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Hornemann[/surname][ign] [/ign][fname]T[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]O´Gorman[/surname][ign] [/ign][fname]E[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Ruck[/surname][ign] [/ign][fname]A[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Brdiczka[/surname][ign] [/ign][fname]D[/fname][ign].[/ign][/oauthor][title
language="en"] Some new aspects of creatine kinase (CK):
compartmentation, structure, function and regulation for cellular and
mitochondrial bioenergetics and physiology[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Biofactors[/stitle][ign]. [/ign][date
dateiso="19980000"]1998[/date][ign]; [/ign][volid]8[/volid][ign]: [/ign][pages]229-34[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]23[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Fukui [/surname][fname]Y[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Sonoyama[/surname][ign] [/ign][fname]T[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Mochizuki [/surname][fname]H[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Ono [/surname][fname]H[/fname][ign]. [/ign][/oauthor][title
language="en"]Effects of heparin dosage and sperm
capacitation time on in vitro fertilization and cleavage of bovine oocytes
matured in vitro.[/title][/ocontrib][ign] [/ign][oiserial][stitle]Theriogenology.[/stitle][ign] [/ign][date
dateiso="19900000"]1990[/date][ign]; [/ign][volid]34[/volid][ign]:[/ign][pages]579-91[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]24[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]O'Flaherty[/surname][ign] [/ign][fname]C[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beorlegui[/surname][ign] [/ign][fname]N[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Effect of natural antioxidants, superoxide
dismutase and hydrogen peroxide on capacitation of frozen-thawed bull
spermatozoa[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Andrologia[/stitle][ign]. [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]29[/volid][ign]: [/ign][pages]269-75[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]25[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Beorlegui[/surname][ign] [/ign][fname]N[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Cetica[/surname][ign] [/ign][fname]P[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Trinchero[/surname][ign] [/ign][fname]G,[/fname][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Córdoba[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]M.[/fname][/oauthor][ign] [/ign][title
language="en"]Comparative study of functional and
biochemical parameters in frozen bovine sperm.[/title][/ocontrib][ign] [/ign][oiserial][stitle]Andrologia[/stitle][ign]. [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]29[/volid][ign]: [/ign][pages]37-42[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]26[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Courage[/surname][ign] [/ign][fname]C[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Budworth[/surname][ign] [/ign][fname]J[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Gescher[/surname][ign] [/ign][fname]A[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Comparison of ability of protein kinase C
inhibitors to arrest cell growth and to alter protein kinase C localisation.[/title][/ocontrib][ign] [/ign][oiserial][stitle]British
Journal of Cancer.[/stitle][ign] [/ign][date
dateiso="19950000"]1995[/date][ign]; [/ign][volid]71[/volid][ign]: [/ign][pages]697-704[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]27[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Wu[/surname][ign] [/ign][fname]AH[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Bowers[/surname][ign] [/ign][fname]GN[/fname][/oauthor][ign]. [/ign][title
language="en"]Evaluation and comparison of inmunoinhibition
and inmunoprecipitation methods for differentiating MB and BB from macro forms
of creatine kinase isoenzymes in patiens and healthy individuals[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Clin Chem[/stitle][ign]. [/ign][date
dateiso="19820000"]1982[/date][ign]; [/ign][volid]28[/volid][ign]: [/ign][pages]2017-21[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]28[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Urdal[/surname][ign] [/ign][fname]P[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Landmark [/surname][fname]K[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Basmo [/surname][fname]GM[/fname][/oauthor][ign]. [/ign][title
language="en"]Mononuclear cell magnesium and retention of
magnesium after intravenous loading in patients with acute myocardial
infarction[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Scand J
Clin Lab Invest[/stitle][ign]. [/ign][date
dateiso="19920000"]1992[/date][ign]; [/ign][volid]52[/volid][ign]: [/ign][pages]763-6[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]29[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Leclerc [/surname][fname]P[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]de Lamirande[/surname][ign] [/ign][fname]E[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Gagnon [/surname][fname]C[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Regulation of phosphotyrosine phosphorylation
and human sperm capacitation by reactive oxygen derivates.[/title][/ocontrib][ign] [/ign][oiserial][stitle]Free
Radical Biol and Med[/stitle][ign]. [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]22[/volid][ign] ([/ign][issueno]4[/issueno][ign]): [/ign][pages]643-56[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]30[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Storey [/surname][fname]BT[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Kayne [/surname][fname]FJ.[/fname][/oauthor][ign] [/ign][title
language="en"]Properties of pyruvate kinase and �űagellar
ATPase in rabbit spermatozoa[/title][ign]: [/ign][subtitle]relation
of metabolic strategy of sperm cell[/subtitle][/ocontrib][ign]. [/ign][oiserial][stitle]J Exp Zool.[/stitle][ign] [/ign][date
dateiso="19800000"]1980[/date][ign]; [/ign][volid]211[/volid][ign]: [/ign][pages]361-67[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]31[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Ficarro[/surname][ign] [/ign][fname]S[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Chertihin[/surname][ign] [/ign][fname]O[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Westbrook[/surname][ign] [/ign][fname]VA[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]White[/surname][ign] [/ign][fname]F[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Jayes[/surname][ign] [/ign][fname]F[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Kalab [/surname][fname]P[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Marto[/surname][ign] [/ign][fname]JA[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Shabanowitz[/surname][ign] [/ign][fname]J[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Herr[/surname][ign] [/ign][fname]JC[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Hunt[/surname][ign] [/ign][fname]DF[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Visconti[/surname][ign] [/ign][fname]PE[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Phosphoproteome analysis of capacitated human
sperm[/title][ign]. [/ign][subtitle]Evidence of tyrosine
phosphorylation of a kinase-anchoring protein 3 and valosin-containing
protein/p97 during capacitation[/subtitle][/ocontrib][ign]. J
Biol Chem. 2003; 278: 11579-89.[/ign][/ocitat][ign]
[/ign][ocitat][no]32[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Arcelay[/surname][ign] [/ign][fname]E[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Salicioni[/surname][ign] [/ign][fname]AM[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Wertheimer[/surname][ign] [/ign][fname]E[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Visconti[/surname][ign] [/ign][fname]PE[/fname][/oauthor][ign]. [/ign][title
language="en"]Identification of proteins undergoing
tyrosine phosphorylation during mouse sperm capacitation[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Int J Dev
Biol.[/stitle][ign] [/ign][date
dateiso="20080000"]2008[/date][ign]; [/ign][volid]52[/volid][ign]: [/ign][pages]463-72[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]33[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Satorre[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Córdoba[/surname][ign] [/ign][fname]M.[/fname][/oauthor][ign] [/ign][title
language="en"]Involvement of intracellular calcium and src
tyrosine-kinase in capacitation of cryopreserved bovine spermatozoa[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]InVet.[/stitle][ign] [/ign][date
dateiso="20100000"]2010[/date][ign]; [/ign][volid]12[/volid][ign] ([/ign][issueno]1[/issueno][ign]) [/ign][pages]75-83[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]34[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Koufen[/surname][ign] [/ign][fname]P[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Rück [/surname][fname]A[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Brdiczka[/surname][ign] [/ign][fname]D[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Wendt[/surname][ign] [/ign][fname]S[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Wallimann[/surname][ign] [/ign][fname]T[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Stark[/surname][ign] [/ign][fname]G[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Free radical-induced inactivation of creatine
kinase[/title][ign]: [/ign][subtitle]in�űuence on
octameric and dimeric states of the mitochondrial enzyme (Mi b-CK).[/subtitle][ign] [/ign][/ocontrib][oiserial][stitle]Biochem J.[/stitle][ign] [/ign][date
dateiso="19990000"]1999[/date][ign]; [/ign][volid]344[/volid][ign]: [/ign][pages]413-7[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]35[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Rotem[/surname][ign] [/ign][fname]R[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Paz[/surname][ign] [/ign][fname]GF[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Homonnai[/surname][ign] [/ign][fname]ZT[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Kalina[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Lax[/surname][ign] [/ign][fname]Y[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Breitbart[/surname][ign] [/ign][fname]H,[/fname][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Naor [/surname][fname]Z[/fname][ign]. [/ign][/oauthor][title
language="en"]Calcium-independent induction of acrosome
reaction by protein kinase C in human sperm[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Endocrinology[/stitle][ign]. [/ign][date
dateiso="19920000"]1992[/date][ign]; [/ign][volid]131[/volid][ign]: [/ign][pages]2235-43[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]36[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Lax[/surname][ign] [/ign][fname]Y[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Rubinstein[/surname][ign] [/ign][fname]S[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Breibart [/surname][fname]H[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Subcellular distribution of protein kinase C
alpha and beta I in bovine spermatozoa, and their regulation by calcium and
phorbol esters[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Biol
Reprod[/stitle][ign]. [/ign][date
dateiso="19970000"]1997[/date][ign]; [/ign][volid]56[/volid][ign]: [/ign][pages]454-9[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]37[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Nassar[/surname][ign] [/ign][fname]A[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Mahony [/surname][fname]M[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Morshedi[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Lin[/surname][ign] [/ign][fname]MH[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Srisombut[/surname][ign] [/ign][fname]C[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Oehninger[/surname][ign] [/ign][fname]S[/fname][/oauthor][ign]. [/ign][title
language="en"]Modulation of sperm tail protein tyrosine
phosphorylation by pentoxifylline and its correlation with hyperactivated
motility[/title][/ocontrib][ign]. [/ign][oiserial][stitle]Fertility
and sterility.[/stitle][ign] [/ign][date
dateiso="19990000"]1999[/date][ign]; [/ign][volid]71[/volid][ign]: [/ign][pages]919-23[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]38[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Lin[/surname][ign] [/ign][fname]SC[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Chen[/surname][ign] [/ign][fname]MC[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Huang[/surname][ign] [/ign][fname]AJ[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Salem[/surname][ign] [/ign][fname]B[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Li[/surname][ign] [/ign][fname]KC[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Glucose and its role in generating reactive
oxygen species required for mouse sperm fertilizing ability[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Asian-Australasian
Journal of Animal Sciences[/stitle][ign]. [/ign][date
dateiso="20000000"]2000[/date][ign];[/ign][volid]13[/volid][ign] ([/ign][issueno]6[/issueno][ign]): [/ign][pages]748-6[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]39[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Chen [/surname][fname]SJ[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Allam[/surname][ign] [/ign][fname]JP[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Duan [/surname][fname]Y[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Haidl [/surname][fname]G[/fname][/oauthor][ign]. [/ign][title
language="en"]In�űuence of reactive oxygen species on
human sperm functions and fertilizing capacity including therapeutical
approaches[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Arch
Gynecol Obstet.[/stitle][ign] [/ign][date
dateiso="20130000"]2013[/date][ign]; [/ign][volid]288[/volid][ign]: [/ign][pages]191-9[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]40[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Kaldis[/surname][ign] [/ign][fname]P[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Stolz[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Wyss[/surname][ign] [/ign][fname]M[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Zanolla[/surname][ign] [/ign][fname]E[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Rothen-Rutishauser[/surname][ign] [/ign][fname]B[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Vorherr[/surname][ign] [/ign][fname]T[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Wallimann[/surname][ign] [/ign][fname]T.[/fname][/oauthor][ign] [/ign][title
language="en"]Identification of two distinctly localizated
mitochondrial creatine kinase isoenzymes in spermatozoa.[/title][/ocontrib][ign] [/ign][oiserial][stitle]J Cell Sci.[/stitle][ign] [/ign][date
dateiso="19960000"]1996[/date][ign]; [/ign][volid]109[/volid][ign]: [/ign][pages]2079-88[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]41[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Miyaji [/surname][fname]K[/fname][ign].[/ign][/oauthor][ign] [/ign][title
language="en"]Creatine kinase isoforms in the seminal
plasma and purified human sperm[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Arch
Androl.[/stitle][ign] [/ign][date
dateiso="20010000"]2001[/date][ign]; [/ign][volid]46[/volid][ign]: [/ign][pages]127-34[/pages][/oiserial][ign].[/ign][/ocitat][ign]
[/ign][ocitat][no]42[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Mendes[/surname][ign] [/ign][fname]JOB[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Burns[/surname][ign] [/ign][fname]PD[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]De La Torre-Sanchez[/surname][ign] [/ign][fname]JF[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Seidel[/surname][ign] [/ign][fname]GE.[/fname][/oauthor][ign] [/ign][title
language="en"]Effect of heparin on cleavage rates and
embryo production with four bovine sperm preparation protocols[/title][ign].[/ign][/ocontrib][ign] [/ign][oiserial][stitle]Theriogenology[/stitle][ign]. [/ign][date
dateiso="20030000"]2003[/date][ign]; [/ign][volid]60[/volid][ign]: [/ign][pages]331-40[/pages][ign].[/ign][/oiserial][/ocitat][ign]
[/ign][ocitat][no]43[/no][ign]. [/ign][ocontrib][oauthor
role="nd"][surname]Breininger[/surname][ign] [/ign][fname]E[/fname][ign],[/ign][/oauthor][ign] [/ign][oauthor
role="nd"][surname]Cetica[/surname][fname] PD[/fname][/oauthor][ign], [/ign][oauthor
role="nd"][surname]Beconi[/surname][ign] [/ign][fname]MT.[/fname][/oauthor][ign] [/ign][title
language="en"]Capacitation inducers act through diverse
intracellular mechanisms in cryopreserved bovine sperm.[/title][/ocontrib][ign] [/ign][oiserial][stitle]Theriogenology[/stitle][ign]. [/ign][date
dateiso="20100000"]2010[/date][ign]; [/ign][volid]74[/volid][ign]: [/ign][pages]1036-49[/pages][ign].[/ign][/oiserial][/ocitat][/other][ign].[/ign][/back][ign]
[/ign][/article]


