INTRODUCTION
Dental resin cements are used to fill the space between indirect restorative material and tooth preparation or implant abutment to prevent dislodgement of the restoration during the masticatory function 1 . Because of their high retentive strength, resistance to wear, and the low solubility of bonded ceramic restorations in relation to anterior and extensive posterior restorations, clinical studies describe a survival rate of up to 81% 2 .
Three kinds of resin cement are available on the market, classified according to their curing mechanism: light-cured, self-cured and dual-cured 3 . Self- and dual-cured alternatives can be used for all cementation applications. However, light-cured resin cements should be limited to porcelain veneers and glass-ceramic restorations, which allow the curing light to penetrate the porcelain. Despite the reformulations of resin cements introduced on the market, the improvements have been related to physicochemical properties and a reduction in the clinical steps 4 . Notwithstanding these improvements, the thickness, microstructure, and shade of the ceramic continue to adversely affect the degree of conversion of methacrylate-based dental resin cements, and pose a risk associated with the toxic effects of residual monomers released into the oral tissues adjacent to dental restorations, such as the mucosa and dental pulp 4 .
The main monomers are 2,2-bis[4-(2-hydroxy-3-methacryloxypropoxy) phenyl] propane (BisGMA) and urethane dimethacrylate (UDMA), in combination with co-monomers of lower viscosity, such as triethylene glycol dimethacrylate (TEGDMA) and 2-hydroxyethyl methacrylate (HEMA) 3, 4 . Toxins may be released both early in the process, owing to defective photopolymerization, and over time, owing to erosion and degradation, influenced by thermal, mechanical, enzymatic and chemical factors 5 . It is estimated that about 1.5 to 5% of the methacrylic groups remain unreacted 6, 7 . In fact, TEGDMA (triethylene glycol dimethacrylate) and UDMA (urethane dimethacrylate) have been detected in dental composite water and artificial saliva eluates by High Performance Liquid Chromatography (HPLC) in concentrations higher than those reported to be cytotoxic in primary human oral fibroblast cultures 8, 9 .
Resin monomer cytotoxicity has been related to the depletion of glutathione (GSH), the major non-enzymatic antioxidant of cells 10-12 . This depletion is accompanied by augmented levels of reactive oxygen species (ROS), causing damage to biomolecules and consequent cell death 13,14 . TEGDMA has been found to induce lipid peroxidation and mitochondrial damage in gingival hGFs, leading to cell death 13 . HEMA, and especially TEGDMA, have been found to induce the formation of micronuclei, leading to chromosomal aberrations in vitro , and TEGDMA has increased the frequency of gene mutations in mammalian cell cultures by more than ten-fold 15 . DNA damage has also been assessed through single-cell microgel electrophoresis (Comet) assay, which indicates the initiation of DNA strand breaks by TEGDMA and HEMA in human lymphocytes and salivary gland tissue 14, 16 .
Considering the reduced cellular detoxifying potency of monomer-exposed cells due to glutathione depletion, it is crucial to the survival of these cells to have an adaptive response that can activate the expression of enzymatic components of the antioxidant system. Thus, this study aimed to evaluate the cytotoxicity and antioxidant gene expression of three resin cements with different polymerization processes. The genes analyzed were those encoding superoxide dismutase 1 (SOD1), peroxiredoxin 1 (PRDX1) , glutathione peroxidase (GPX1) , catalase (CAT) and glutathione synthetase (GSS). SOD1 accelerates the conversion of the superoxide anion radical to hydrogen peroxide, while PRDX1, GPX1 and CAT convert hydrogen peroxide to water. GSS is the second enzyme in the GSH biosynthesis pathway. The tested hypothesis was that the polymerization process would affect toxicity and antioxidant gene expression in human fibroblasts.
MATERIALS AND METHOD
Resin cements and specimen preparation
All the tested materials and their compositions are listed in Table 1. The materials were hand-mixed in a flow chamber, following the manufacturer’s instructions to use a 1:1 ratio, a metal spatula, and a block of waterproof paper. Briefly, the materials were placed in sterile bipartite metal molds (1-mm high and 14 mm-diameter), inserted in a single increment, and pressed between polyester strips and glass slides to prevent the formation of an oxygen-inhibited surface layer. Next, they were polymerized according to the characteristics of each cement. Although VLII is a dual-curing cement, it may be applied only with a light-curing technique, and was used for this sole purpose. Photoactivation was performed with a light-emitting diode, at a curing intensity of 1000 mW/cm2, set to standard power, with a 9.6-mm lens diameter and wavelength of 395-480 nm, and kept plugged into an electrical outlet (VALO™, Ultradent Products, South Jordan, UT, USA). Each disc side was exposed for 20 s. All the samples were subjected to disinfection by ultraviolet light for 20 minutes on each side.
Table 1 Material, type, composition, manufacturer, and batch number of cements used in the study
|
Material |
Type |
Composition* |
Manufacturer/ Batch # |
||
|---|---|---|---|---|---|
|
RelyX Ultimate RXU |
Dual-curing Adhesive resin cement |
50-60% glass powder 20-30% methacrylated phosphoric acid esters 10-20% TEGDMA 1-10% silane-treated silica < 1% sodium persulfate |
3M/ESPE St. Paul, MN, USA 130720 |
||
|
VariolinkII (Base) VLII |
Dual-curing (Applied in the light-curing technique only) Adhesive resin cement |
10-25% Bis-GMA 2.5-10% UDMA 2.5-10% TEGDMA |
Ivoclar Vivadent Schaan, Liechtenstein U25861 |
||
|
RelyX U200 RXU200 |
Dual-curing Self-adhesive resin cement |
45-55% glass powder 20-30% methacrylated phosphoric acid esters 10-20% TEGDMA 1-10% silane-treated silica < 3% sodium persulfate |
3M/ESPE St. Paul, MN, USA 160920 |
||
Cell Culture
To obtain samples, three healthy subjects were submitted to a gingival biopsy after approval by the Institutional Ethics Committee (protocol #1.303.768). Briefly, primary cultures were isolated using the explant technique, after being enzymatically digested by a 0.25% trypsin-EDTA solution for 1 h at 37oC. The cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM), supplemented with 10% fetal bovine serum and 1% antibiotic-antimycotic solution (penicillin-streptomycin), and incubated under standard cell culture conditions (37oC, 100% humidity, 95% air and 5% CO2).
Extract Preparation and Cell Exposure
Extracts were obtained by incubating the resin cements of the specimens in DMEM medium, in a proportion of 0.2 g/mL at 37°C for 24 h 17 . The cells were plated on 6-well (1x106 cells/well) or 24-well (2x104 cells/well) plates. After 24 h, the culture medium was removed and replaced by the different extracts taken from the 3 cements - RelyX Ultímate™ (RXU), Variolink™n (VLII), and RelyX U200™ (RXU200) - or in the DMEM medium (control). The cells were cultured under standard cell cultivation for up to 3 days, as described above.
Cell Viability (Trypan blue exclusión test)
After 24 h, 48 h and 72 h of exposure to different cements extracts, vital trypan blue exclusion was used to evaluate cell viability. Cells were removed enzymatically from the plates, and the cell pellet obtained from the centrifugation was suspended in 1 mL of medium. Ten gL of the cell suspensión was added to 10 gL of Trypan Blue solution 0.4% (Sigma-Aldrich, St. Louis, MO, USA) and the solution was gently mixed for 30 sec. After that, the solution was left to stand for 5 min. Then, 1 pL of this solution was placed in a hemocytometer (Neubauer-Fisher Scientific, Pittsburgh, PA, USA), and observed for cell count and analysis using a phase microscope (Nikon Eclipse TS100, Tokyo, Japan).
Expression of antioxidant genes (quantitative real-time PCR)
After 24 h of exposure to different cements extracts, total RNA was extracted from cell cultures using Trizol reagent (Thermo Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. Briefly, cells were collected and homogenized with 1 mL of Trizol, and the aqueous and organic phases were separated by adding chloroform (0.2 mL), followed by centrifugation (12,000 g, 15 minutes, 4°C). RNA was precipitated from the aqueous phase with 0.5 mL of isopropanol (12,000 g, 15 minutes, 4°C), washed with 75% ethanol, and suspended in water. Reverse transcription was performed using 1 pg of each RNA sample, treated with 1 U DNase I, and the RevertAid H Minus First Strand cDNA Synthesis Kit (Thermo Scientific). Briefly, reactions were initiated by using 1 pg of RNA, 0.5 pg of oligo (dT) 18, 1 mM of dNTP mix, 200 U of RevertAid H Minus M-MuLV Transcrip-tase, and 20 U of RiboLock RNase Inhibitor at 42°C for 60 minutes. The reactions were then terminated by heating the samples at 70°C for 5 minutes. The reactions were initiated by using 40 ng of cDNA and 0.3 pM of pairs of primers, added to the Maxima SYBR Green qPCR Master Mix (Thermo Scientific) (Table 2). The primer sets were as follows: Peroxiredoxin I (PRDX1; Forward 5 ’ -GGATTCTCACTTCTGT-CATCTAGCA-3’; Reverse 5 ’ -TGTTCATGGGTC-CCAGTCCT-3’), Glutathione Peroxidase I (GPX1; Forward 5 ’-CCGACCCCAAGCTCATCA-3 ’; Reverse 5’-GAAGCGGCGGCTGTACCT-3’), Catalase (CAT; Forward 5 ’ -GATAGCCTTCGACCCAAG-CA-3’; Reverse 5 ’ -ATGGCGGTGAGTGTCAG-GAT-3’), Superoxide dismutase 1 (SOD1; Forward 5’-AGGTCCTCACTTTAATCCTCTATCCA-3’; Reverse 5 ’-ACCATCTTTGTCAGCAGTCACATT -3’), Glutathione synthetase (GSS; Forward 5 ’-ATTTGACCAGCGTGCCATAGAG-3 ’; Reverse 5’-TCCAGAGACCCCTTTTCAGAGATATC-3’), and for internal gene reference Glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Forward 5 ’-ACCCACTCCTCCACCTTTGA-3 ’; Reverse 5’-TGTTGCTGTAGCCAAATTCGTT-3’). The con-ditions required to elicit a reaction consisted of main-taining the samples 10 minutes at 95°C, followed by 40 cycles at 95°C. Each cycle consisted of keeping the samples 15 s at 95°C, and 1 min at 60°C. The rela-tive expression among the samples was calculated by comparing the threshold cycle values, based on the 2-AACt formula. The GAPDH gene was used to nor-malize the expression levels.
Table 2 Analyzed genes and sequences of the primers used
|
Gene name and symbol |
Gene Bank (NM) |
Sequences of primers |
|---|---|---|
|
Peroxiredoxin I PRDX1 |
181696.1 * 181697.1 * 002574.2* |
F 5'-GGATTCTCACTTCTGTCATCTAGCA-3' R 5'-TGTTCATGGGTCCCAGTCCT-3' |
|
Glutathione Peroxidase I GPX1 |
000581.2 * 201397.1* |
F 5'-CCGACCCCAAGCTCATCA-3' R 5'-GAAGCGGCGGCTGTACCT-3' |
|
Catalase CAT |
001752.3 |
F 5'-GATAGCCTTCGACCCAAGCA-3' R 5'-ATGGCGGTGAGTGTCAGGAT-3' |
|
Superoxide dismutase 1 SOD1 |
000454.4 |
F 5'-AGGTCCTCACTTTAATCCTCTATCCA-3' R 5'-ACCATCTTTGTCAGCAGTCACATT -3' |
|
Glutathione synthetase GSS |
000178 |
F 5'-ATTTGACCAGCGTGCCATAGAG-3' R 5'-TCCAGAGACCCCTTTTCAGAGATATC-3' |
|
Glyceraldehyde-3- phosphate dehydrogenase GAPDH |
002046.3 |
F 5'-ACCCACTCCTCCACCTTTGA-3' R 5'-TGTTGCTGTAGCCAAATTCGTT-3' |
RESULTS
Cell viability
The toxicity of the resin cements was assessed by exposing the fibroblasts to the respective extracts, and then determining the number of viable cells (Fig. 1). A significant reduction was observed in the number of viable cells after 24 h of exposure to RXU and RXU200 extracts, but not to VLII, compared to the control medium (p<0.05). All the cements caused a reduction in the viable cell number, but the more drastic effects were observed with exposure to the RXU and RXU200 extracts (5-fold), compared to the control medium (p<0.05; Fig. 1).
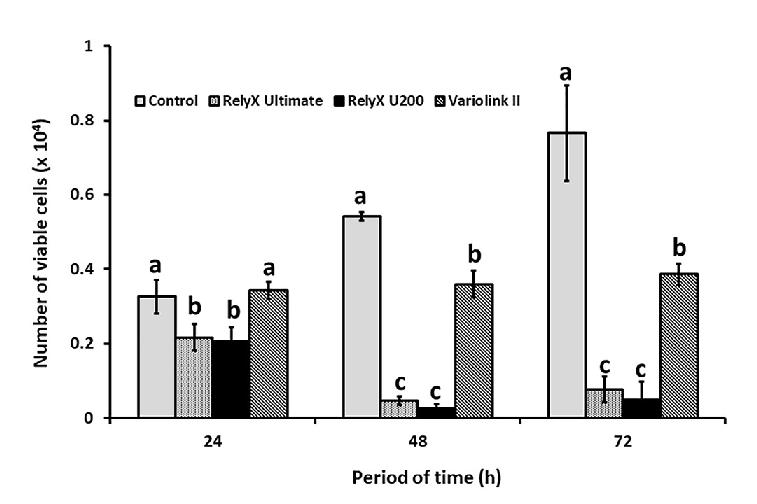
Fig. 1 Influence of the resin cement extracts on the viability of human gingival fibroblasts. Gingival fibroblasts were exposed to the extracts of RelyX Ultimate, RelyX U200 and Variolink II, or to the culture medium (Control) for 24, 48 and 72 h, and the number of viable cells was obtained by the vital trypan blue exclusion test. The data represent mean and standard deviation. Different lowercase letters show that there was significant difference for viable cell number for ANOVA and Bonferroni tests (p<0.05).
Expression of antioxidant genes
To gain better understanding of how well resin cements induce oxidative stress and antioxidant adaptive cellular response, the expression of PRDXI, GPX1, CAT, SOD1 and GSS antioxidant genes was analyzed in fibroblasts exposed to the resin cement extracts, and the levels were compared to those of the cells grown in the culture medium (control). This analysis showed a significant increase in PRDX1 transcript levels, caused by the extracts of all the tested cements (p <0.05), as well as SOD1 induction by the RXU200 and VLII cement extracts (p <0.05) (Fig. 2). The other genes (GPX1, CAT and GSS) were induced significantly only by the RXU200 extract (p <0.05). The highest levels of expression of these genes were observed with the exposure to the RXU200 cement extract (Fig. 2).
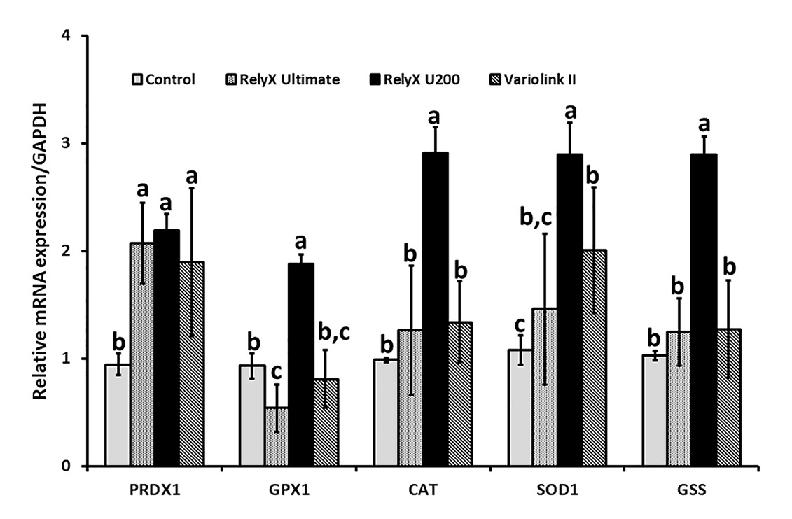
Fig. 2 Influence of the resin cement extracts on the expression of antioxidant genes in human gingival fibroblasts. Gingival fibroblasts were exposed to the extracts of RelyX Ultimate, RelyX U200 and Variolink II or to the culture medium (Control) for 24 h, and the expression of PRDX1, CAT, GPX1, SOD1 and GSS was assessed by qPCR. The data represent mean and standard deviation. Different lowercase letters for each gene show differences by ANOVA and Bonferroni tests (p<0.05).
DISCUSSION
In the present study, the toxicity of different cements was assessed by exposing fibroblasts to the extracts obtained from resin materials. The results demonstrated that VLII cement was less toxic than RXU and RXU200, as observed by a greater number of viable cells during the 72 h of exposure to the corresponding extracts. One of the determining factors for the concentration of residual monomers is the polymerization process. It is important to emphasize that only the basal surface of this cement was used for this analysis, since polymerization ofthe cement is dependent exclusively on light, according to the photoactivation process, thus eliminating the need to mix the base and catalyst pastes. RXU and the RXU200 cements required dual polymerization, and presented the most exacerbated toxic effects, which were statistically similar. This could be attributed to the longer time required for polymerization, leading to more methacrylic groups remaining unreacted. In agreement, Kurt et al. showed that all resin cements were toxic to fibroblasts, but that RXU200 had the greatest toxic effect 9 .
It is assumed that the combination of different methacrylates potentiates their toxicity, compared with uncombined forms. Ratanasathien et al. studied the isolated effects of methacrylates, and determined that the most toxic cements, in decreasing order, are those containing Bis-GMA, followed by UDMA and TEGDMA, which are less toxic 18 . In our study, all the cements were formulated with Bis-GMA. However, considering the associations of methacrylates, TEGDMA had the lowest cytotoxicity, and VLII had comparatively lower toxicity, hence corroborating the findings of Ratanasathien et al. On the other hand, toxicity studies on isolated methacrylate components reported that TEGDMA and Bis-GMA are the most toxic monomers, compared to EDMA and UDMA 19, 20 . Different experimental conditions may be associated with the divergent results in the study mentioned. It is known that even small amounts of monomers can induce toxic effects in cells, and that toxicity increases significantly at higher concentrations 21 .
Lefeuvre et al. suggested that the death of cells exposed to TEGDMA, BIS-GMA and other resin monomers occurs due to an intracellular increase in ROS subsequent to a decrease in the GSH antioxidant agent 13 . These monomers were found to cause drastic and rapid GSH depletion in gingival fibroblast pulp cells 10, 22 , based on a suggested mechanism involving the formation of GSH-monomer adducts 23, 24 . In line with this mechanism, N-acetylcysteine (NAC), ascorbate and Trolox antioxidants were shown to prevent TEGDMA-induced toxicity, and partially restore GSH levels in gingival fibroblasts 10 . In addition, Kurt et al. showed that the TEGDMA released from resin cements (including RXU200) increased in artificial saliva over time (1, 24 and 72 h) 9 . They also showed that stimulated ROS production increased the genotoxicity of resin cements more than 3-fold relative to that of the control, and decreased cell viability in L-929 mouse fibroblasts exposed to the cements, compared to non-exposed fibroblasts. In line with ROS involvement in resin cement toxicity, our results showed an increase in PRDX1 expression in human gingival fibroblasts exposed to all the cement extracts, compared to those left unexposed. This increase indicates an attempt by the cells to survive a disturbance in their redox balance by increasing their antioxidant defense.
ROS generation is expected to be lower when an exclusively light-cured system is applied, such as that used with VLII cement. This is because the formation of these species is totally dependent on the presence of photons at a depth that does not exceed a few millimeters 4 . On the other hand, when chemical curing is used, free radicals are formed throughout the bulk of the curing material, regardless of depth. Hence, use of the dual cements requires a broader defense response. In fact, our results showed that not only PRDX1 antioxidant genes, but also RXU200 cement caused an increase in the transcript levels of all the other genes studied (GPX1, CAT, SOD1 and GSS) . However, this broader effect was not observed in cells exposed to RXU. Since the curing system cannot explain the different expression pattern induced by the two RX dual cements, and their composition is very similar, we can only speculate that it could be related to the concentration of the ingredients. Although exact percentages are declared as trade secrets in manufacturers’ safety data sheets, it is known that sodium persulfate may reach a higher percentage by weight in RXU200 (<3 for RXU200 and <1 for RXU). The same is true for the percentage of tert-butyl peroxy-3,5,5-trimethylhexanoate (<0.50 for XU200 and <0.25 for RXU). Persulfate salts and organic peroxides are radical initiators of cross-linking methacrylate monomers, and have strong oxidizing properties. It has been reported that the ammonium salt of persulfate-induced ROS generation in mast cells and basophils 25 , and also MCF-7 breast cancer cells, leads to oxidative stress 26. Thus, we can hypothesize that the higher percentage of these chemical initiators in RXU200 may have led to higher levels of ROS formation in the cells, activating additional signaling pathways responsible for the induction of a greater variety of antioxidant genes. Even so, the suggested activation of the oxidative stress response does not seem entirely efficient. This may be the reason why a drastic reduction in the number of viable cells was observed when the cells were exposed to any of the dual cements, especially after 48 h. Regarding the results of the exposed VLII base cells, ROS formation was mostly attributed to residual monomers left unpolymerized, rather than to the release of residual chemical initiators. Thus, PRDX1 and SOD1 induction by the cells exposed to this cement may have contributed to their survival during the exposure interval (72 h). These results support the proposition that cement cytotoxicity was at least partly related to the formation of ROS.
The findings of the present study showed that biological principles must be considered during operative and restorative procedures; hence, it is critical to determine the professional clinical choice of whether to involve the patient. Therefore, it is generally accepted that in vitro tests, mostly based on cell culture systems, must precede in vivo approaches when testing the health risk of dental materials. This is a major concern and ongoing issue.















