INTRODUCTION
At the end of 2019, a novel coronavirus was identified as the cause of a cluster of pneumonia cases in China; the infection rapidly spread worldwide, resulting in a global pandemic (1). Coronavirus disease 2019 (COVID-19) is thought to cause acute kidney injury (AKI) by various mechanisms. The proposed mechanisms of kidney injury are numerous and range from a direct viral infection that affects the renin-angiotensin-aldosterone system to hemodynamic instability, the activation of coagulopathy, cytokine storm, and the activation of the immune system (2,3). IgA nephropathy (IgAN) is the most common lesion found to cause primary glomerulonephritis throughout most developed countries of the world. IgAN is typically triggered by upper respiratory infections, and relapse of IgAN has been reported in patients receiving vaccinations (4-8). Here, we present a case of newly diagnosed crescentic IgAN after SARS-CoV-2 infection and mRNA-based SARS-CoV-2 vaccine.
CASE REPORT
A 31-year-old man with no medical history presented with gross hematuria 24 hours after SARS-CoV-2 infection. He reported no personal history of kidney disease, including IgAN. The diagnosis of COVID-19 was made primarily by direct detection of viral reverse-transcription polymerase chain reaction (RT-PCR) from the upper respiratory tract. Chest computed tomography findings were consistent with SARS-CoV-2 infection. He did not have hospitalization during SARS-CoV-2 infection. He was treated with favipiravir and paracetamol. He did not have a urinalysis when he had gross hematuria. Serum creatinine was 0,93 mg/dL (Ref: 0.76-1.27 mg/dL), and the estimated glomerular filtration rate was 109 ml/min/1.73 m2. Hematuria had been resolved spontaneously within three days. He was not referred to nephrology during the gross hematuria attack. However, due to the development of hematuria after the viral infection, its spontaneous recovery, and the absence of a kidney stone story, we think that he had an attack of IgAN even though he did not have a urine test or histopathological evidence. He had been vaccinated with two doses of CoronaVac (Sinovac) three months after the SARS-CoV-2 infection, and he did not describe a hematuria attack after the CoronaVac (Sinovac) vaccine. He had been vaccinated with the Pfizer-BioNTech COVID-19 vaccine one month after the second dose of CoronaVac (Sinovac) vaccine, and gross hematuria occurred 24 hours after the Pfizer-BioNTech COVID-19 vaccine. He was referred to nephrology for consultation. The patient had no history of renal disease, and there was no hematuria in the past urine analysis. Physical examination was normal, and blood pressure was 123/75 mm Hg. He did not have lower extremity edema, rash, lymphadenopathy, or throat erythema. Serum creatinine was 1.15 mg/dL (Ref: 0.76-1.27 mg/dL), and the estimated glomerular filtration rate was 84 ml/min/1.73 m2. A urinalysis showed 3+ protein (ref: negative), 242 red blood cells per high-power field (ref: 0-3), 13 white blood cells per high-power field (ref: 0-4), and 3+ blood (ref: negative). The random urine protein-creatinine ratio was 0.67 g/g (ref: 0-0.2 g/g), estimating 24-hour urine protein excretion of 670 mg. 24-hour urine protein was 1083 mg/day. Kidney ultrasound showed mildly increased echogenicity of average size and cortical thickness. Additional serological work-up for glomerulonephritis was negative, including hepatitis B antigen, anti-hepatitis C antibody, anti-HIV (Human immunodeficiency virus) antibody, and antinuclear and antineutrophil cytoplasmic antibodies. Erythrocyte sedimentation rate, Rheumatoid factor, and C-reactive protein levels were normal. Complements C3 (1.56, ref: 0.9-1.8 g/L) and C4 (0.4, ref: 0.1-0.4 g/L) were normal. Creatinine phosphokinase was 109 U/L (ref: 30-200 U/L), and Immunoglobulin A level was normal. The patient's laboratory results, timeline of infection, and vaccinations are shown inTable 1.
Table 1: Timeline of patient's infection and vaccinations.
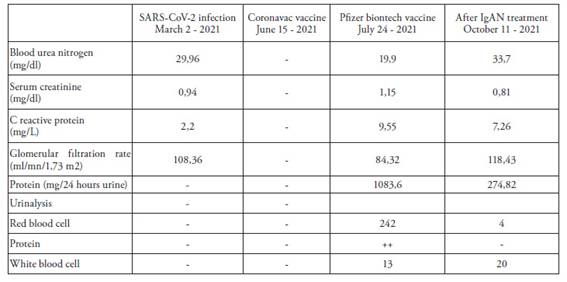
Given the unclear diagnosis, an ultrasound-guided percutaneous renal biopsy was performed. Light microscopy revealed 77 glomeruli, most of which had a mild mesangial expansion, hypercellularity with endocapillary hypercellularity, and adhesion of a capillary loop to the Bowman capsule (Figure 1), 1 of which showed global sclerosis, 1 of which showed segmental sclerosis.
Figure 1: Mild tubular atrophy and interstitial fibrosis are present. H&E x 100.
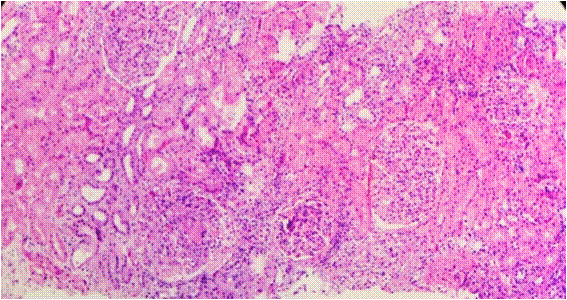
4 of which showed cellular crescent (Figure 2), 1 of which showed fibrous crescent (7.7%), 2 of which showed fibrinoid necrosis.
Figure 2
While a cellular crescent is observed in the left glomerulus, an increase in mesangial cellularity is observed in the other glomerulus. H&E x200.
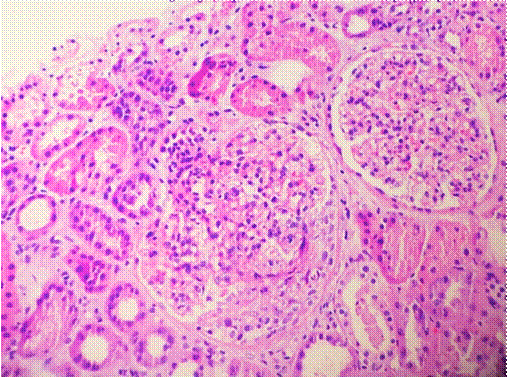
Immunofluorescence revealed 3+ diffuse granular mesangial staining for IgA (Figure 3).
Figure 3: Mesangial accumulation in glomeruli with IgA in direct immunofluorescence examination x40
Staining was positive for C3 and negative for IgG and other immunoglobulins and complement antibodies. Mild tubular atrophy and interstitial fibrosis were observed. Erythrocyte clumps, hemoglobin casts, and hyaline casts were detected in some tubular lumens. Pathologic features were consistent with IgAN with Oxford MEST-C classification as M1-E0-S1-T1-C1, and his risk of a 50% decline in estimated glomerular filtration rate or progression to kidney failure within five years was approximately 9.75%, as per a recent risk prediction model by the International IgA Nephropathy Network. He started on Ramipril 10 mg daily, 1 gr pulse Methylprednisolone for one day, following 1mg/kg/day, and fish oil, which was well tolerated. After eight weeks of therapy, the urine protein-creatinine ratio improved to 0.27 g/g, and creatinine remained stable at 0.83 mg/dL.
DISCUSSION
Here we present a patient with probable de novo crescentic IgAN associated with COVID-19 infection. Our patient's Pfizer-BioNTech COVID-19 vaccine and COVID-19 infection triggered a gross hematuria attack.
IgAN is the most common lesion found to cause primary glomerulonephritis throughout most developed countries. Patients may present at any age, but there is a peak incidence in the second and third decades (9). Upper respiratory tract infections often precede the onset or exacerbation of the IgAN. Various immune triggers for IgAN have been published, including staphylococcal infection, vaccination, and viral infection (4-8). Viral infections contribute to the multihit hypothesis of IgAN and trigger a cascade of events, including an increase in circulating galactose-deficient IgA1 (gd-IgA1) antibodies, development of autoreactive antibodies to gd-IgA1, and formation of immune complexes that deposit in various tissues, including the kidney, triggering an inflammatory response (10). IgAN is an immune-complex disease characterized by mesangial IgA1 deposition with or without concurrent IgG and C3 deposits.
Renal manifestations of SARS-CoV-2 infection encompass the spectrum of kidney injury, including increased serum creatinine levels and asymptomatic urinary abnormalities (11). In patients admitted to the hospital with COVID-19, asymptomatic urinary abnormalities like hematuria and proteinuria or AKI are significantly associated with increased mortality (12). Renal biopsy series have been reported reviewing kidney pathology from patients with COVID-19. In 26 biopsies, Su H et al. reported no samples demonstrated hypercellular or inflammatory glomerular lesions, or crescents, despite including patients with hematuria and proteinuria. The most common pathological finding reported was acute tubular injury, which was suggested to be direct viral effects in the proximal tubule epithelium (13).
Our patient presented with gross hematuria 24 hours after SARS-CoV-2 infection and the Pfizer-BioNTech COVID-19 vaccine. While COVID-19-related AKI is seen in relatively severe hospitalized patients, our patient was managed in an outpatient clinic given the mild COVID-19 presentation during the SARS-CoV-2 infection. Gross hematuria regressed spontaneously within three days after the SARS-CoV-2 infection. He had been vaccinated with two doses of CoronaVac (Sinovac) three months after SARS-CoV-2 infection. Furthermore, he had been vaccinated with the Pfizer-BioNTech COVID-19 vaccine one month after the second dose of CoronaVac (Sinovac) vaccine.
Ran E et al. reported a case of crescentic IgAN following the CoronaVac vaccine (14). However, our patient did not describe a hematuria attack after the CoronaVac (Sinovac) vaccine doses. However, he presented with gross hematuria and subnephrotic proteinuria 24 hours after the first dose of the Pfizer-BioNTech COVID-19 vaccine. People who receive mRNA-based vaccines demonstrate robust antibodies against the receptor-binding domain (RBD) of the S1 protein. Similar to natural infection, due to the powerful stimulation of immune response from mRNA-based vaccines compared to other vaccines, the patients may produce de novo antibodies, leading to IgA-containing immune-complex deposits in the kidney. Besides COVID-19 vaccines, influenza and the recombinant zoster vaccine may cause hematuria and flare-up IgA nephropathy (15). In addition, other vaccines, such as hepatitis B, measles, and pneumococcus, have been linked to renal pathologies like nephrotic syndromes (16). Nevertheless, according to some recent studies, developing glomerulonephritis after vaccination against SARS-CoV-2 may be a rare adverse event. They claimed a temporal association was found for IgAN and minimal change disease, but causality was not firmly established (17).
This case highlights the immunological effects of the novel mRNA-based SARS-CoV-2 vaccines. Nephrologists should be aware of new-onset hematuria or proteinuria observed after SARS-CoV-2 infection or mRNA-based SARS-CoV-2 vaccine.
Recibido: 18-01-2023
Corregido: 27-01-2023
Aceptado: 08-05-2023