INTRODUCTION
Pascalia glauca (fig. 1 A), which is widely distributed in Argentina and neighboring countries (Burkart and Carera, 1953), causes several deaths in animals from different production systems. It is distributed in northern Chile, Paraguay, Uruguay and the north-central region of Argentina (Micheloud and Odriozola, 2012). P. glauca is described as toxic for cattle, sheep, pigs, horses, goats and llamas (Morán and Kosik, 1965; Riet-Correa, 1978; De Diego and Solla, 1991; López et al., 1991; Collazo and Riet-Correa, 1996; Rodriguez Armesto et al., 2003; Magnano et al., 2011). Reports about this toxicity in goats are imprecise and old (Ragonese and Milano, 1984) and experimental works include mainly cattle and sheep (Collazo and Riet-Correa, 1996). The toxic principle of P. glauca is a carboxyatractyloside (Schteingart and Pomilio, 1984) that can block mitochondrial ATP synthesis, oxidative phosphorylation and other metabolic reactions in hepatocytes (Obatomi and Bach, 1998; Bruneton, 2001) and renal tubular cells (Obatomi and Bach, 1996). The liver is the most affected organ since it is severely exposed to absorbed toxins (Plumlee et al., 2004), causing diffuse hemorrhagic necrosis with acute liver failure (Micheloud and Odriozola, 2012) microscopically characterized by centrilobular necrosis with hemorrhages (Stalker and Hayes, 2007). This work aims to clarify P. glauca toxicity in goats, characterize clinical and pathological findings and determine the necessary dose to induce poisoning.
MATERIALS AND METHODS
The study protocol was approved by the UCASAL Animal Care and Use Committee (CICUAL number 53-18-17). The requirements of the Argentine Animal Protection Policy (Ley 14346) were always fulfilled.
P. glauca samples in vegetative and early flowering states were collected, dried for seven days at room temperature and subsequently shredded to a particle size of 2 to 5 millimeters. Before grinding, some samples of the collected plant were sent to the CMSC herbarium of the National University of Salta for botanical identification.
Twelve adult Saanen goats were divided into 4 groups of 3 animals each. Before the experiment, a clinical examination was performed on all the selected animals to determine that they were in good health. The exposed groups were divided as follows: T1 (animals 4 to 6), T2 (animals 7 to 9) and T3 (animals 10 to 12) were given a dose of 4 g/kg, 6g/kg and 8g/kg of dry matter respectively by the orogastric route. The control group (CG) was integrated by animals 1 to 3 and was given 8 g/kg of dry shredded alfalfa only. After this, all the animals were sent to a paddock with ad libitum feeding and water. There were no toxic plants in the paddock that could interfere with the experiment. Clinical examination was done every two hours and blood samples were collected at the beginning of the experiment and then 6, 12, 24 and 48 hours later and serum levels of aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT) and alkaline phosphatase (ALP) were determined following the instructions of the manufacturer (Wiener Lab®). The analysis of the variables obtained was performed by non-parametric ANOVA for measurements repeated over time using the InfoStat software.
RESULTS
Clinical findings
Animals from CG and T1 groups did not show clinical signs and their health status and behavior were normal both during the experiment and for 15 days after the experiment.
In contrast, in the T2 and T3 groups, clinical signs started after 14 hours (range 8-24) and 10 hours (range 6-12) after ingestion of the plant, and death occurred within 21 hours (range 12-30) and 29,3 hours (range 10-48) respectively. The main clinical signs observed consisted of anxiety, anorexia, ruminal atony, tachycardia, tachypnea, and abnormal recumbency, including an antalgic position to avoid pressure on the right epigastric region. One animal showed a staggering gait and head-pressing (fig. 1 B). At an advanced stage, animals showed recumbency, myoclonus, pedaling movements, seizures and death. Only two animals died early without apparent clinical signs.
Laboratory findings
Table 1 shows the level of Aspartate aminotransferase (AST), Gamma-glutamyl transpeptidase (GGT) and alkaline phosphatase (ALP). Hepatic enzyme levels increased significantly within 12 to 48 hours after consumption in the T2 and T3 groups presumably due to the dose ingested. AST and ALP were the most increased enzymes in those groups.
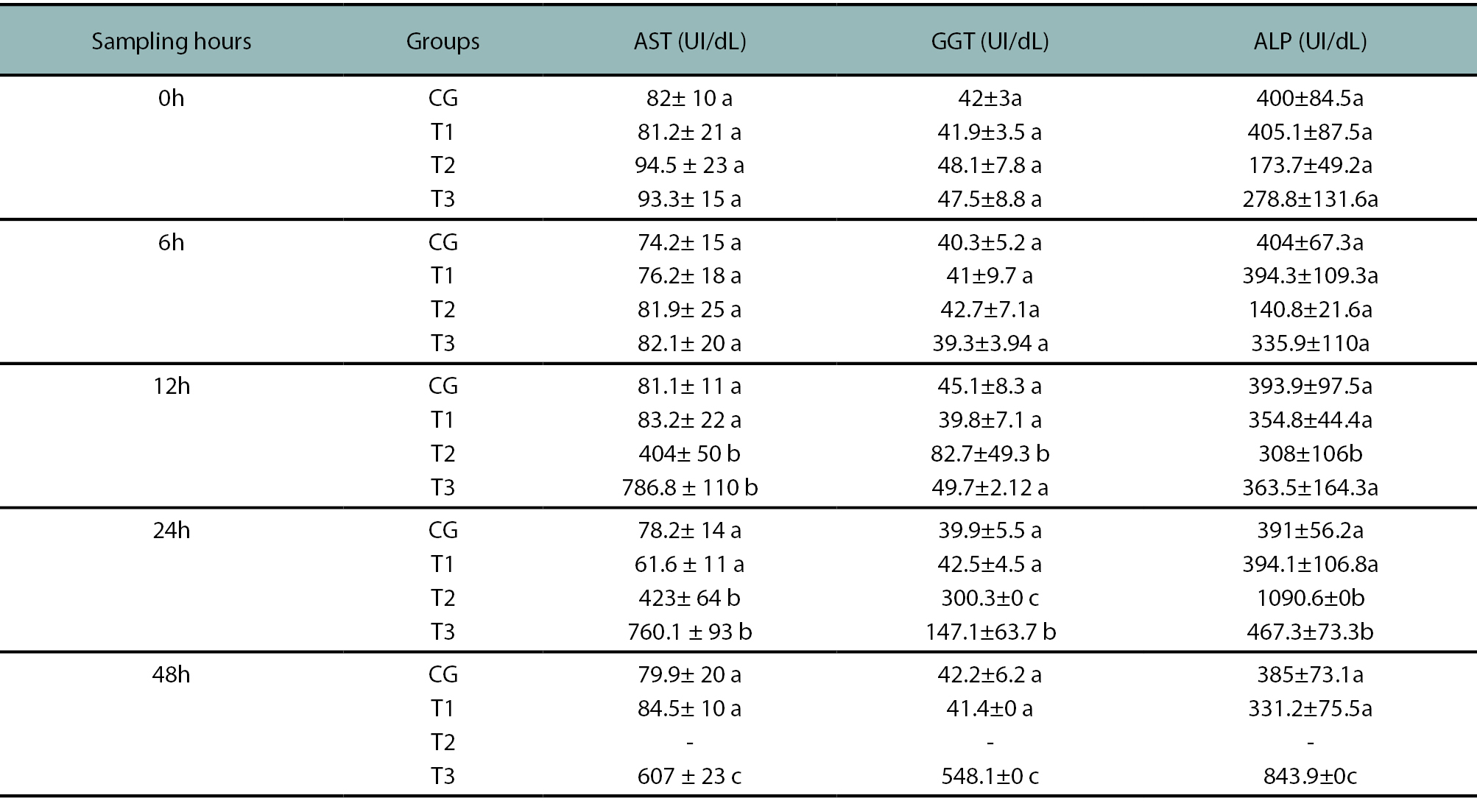
Table 1.Hepatic enzyme activity at different stages of the experiment. AST: Aspartate aminotransferase (AST), Gamma-glutamyl transpeptidase (GGT) and alkaline phosphatase (ALP). Different letters show significant differences between sampling times (p < .05).
Macroscopic findings
Necropsies were performed on all the animals included in the experiment. Groups T2 and T3 showed liver injuries at different stages. The main finding consisted of liver congestion with a diffuse reticulated appearance with dark red areas alternating with pale areas in correspondence with necrotic and normal areas (fig. 1 C). The liver of animals N 7 and 10 showed a mild lobular pattern while in animals N 8 and 11 this pattern was even more pronounced. In comparison, the liver of animals N 9 and 12 were the most affected with severe injuries and a marked lobular pattern. Animals N 9 to 12 also showed endematous gallbladder. Abomasal and intestinal edema and congestion, renal congestion and petechiae in serous membranes were also found. The animals from the control group did not show evidence of injuries in any organ or tissue.
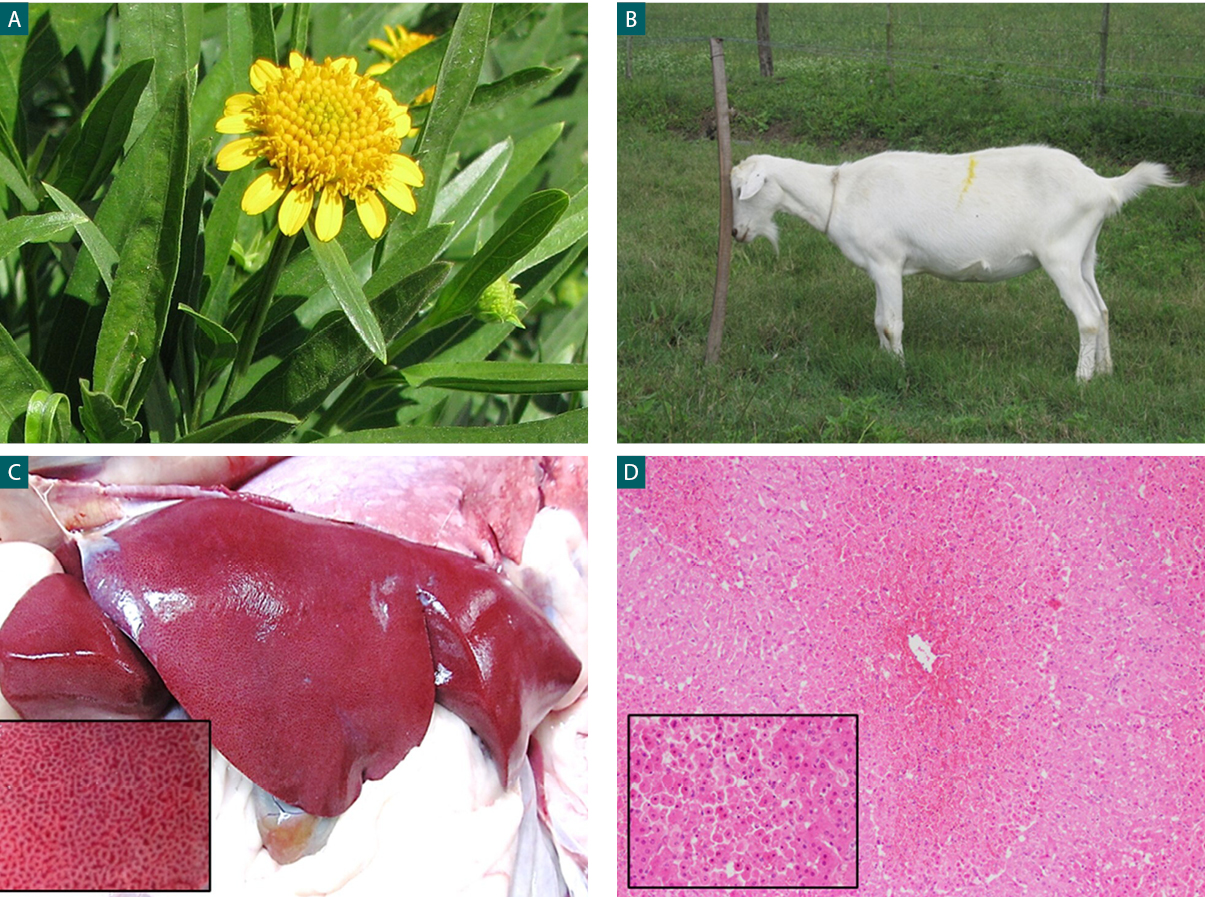
Figure 1.A- P. glauca inflorescence. B- Poisoned goat head-pressing. C- Liver. A reticular pattern in the parenchyma. D- Hemorrhagic centrilobular necrosis (arrowhead). H&E, 10X. Insert: note the nuclear (karyorrhexis and pyknosis) alterations (H&E, 100X).
Microscopic findings
The most frequent finding in the liver was diffuse centrilobular necrosis with hemorrhages (fig. 1 D).
The severity of the injuries in the affected animals was categorized in table 2. Regarding necrosis, severity was classified as follows: (+) 25-30% of affected lobule surface area, then (++) 31-60% and (+++) more than 61% respectively. In reference to hemorrhage, (+) means small isolated spots of hemorrhage, (++) means moderate to extensive hemorrhage foci and (+++) means massive hemorrhage. Hemorrhage was limited to necrotic areas only. In animals 9, 10 and 11 there was intermediate area hepatocyte atrophy and hydropic degeneration in periportal hepatocytes. Animal 10 liver showed severe massive necrosis. Animals 7, 10 and 11 also showed neutrophilic infiltration in isolated clusters of 2 to 3 cells. Other lesions found renal congestion with hydropic degeneration in tubular cells and eosinophilic material in the tubular lumen and glomeruli. In addition, the central nervous system showed gray matter edema and congestion, neuronal degeneration and white matter vacuolization.
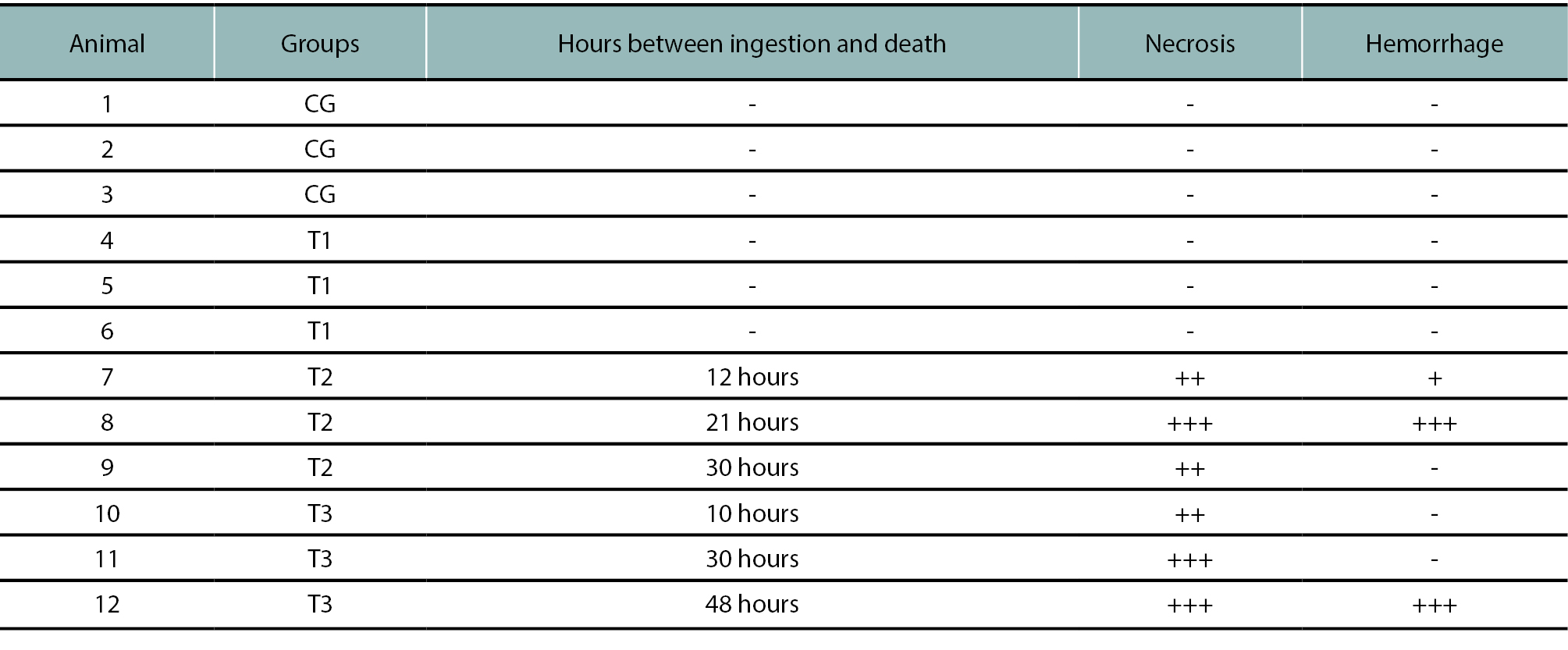
Table 2.Microscopic lesions severity in the liver. -: score 0; +: score 1; ++: score 2; +++: score 3. Necrosis: (+) 25-30% of affected lobule surface area, (++) 31-60% and (+++) more than 61% respectively. Hemorrhage: (+) isolated spots of hemorrhage, (++) moderate to extensive hemorrhage foci and (+++) massive hemorrhage. CG: control group; T1: 4 g MS /kg; T2: 6 g MS/kg; T3: 8 g MS/kg.
DISCUSSION
Although goats are known to be susceptible to P. glauca poisoning, no other experimental poisoning works were found in the consulted literature. Goats showed sensitivity with a toxic dose of 6 g/kg of dry matter, being markedly higher than the toxic dose described in cattle and sheep of 5 to 10 g/kg of green plant (Collazo and Riet-Correa, 1996), 4 to 10 g/kg of green plant (INTA, 2007) and 4 g/kg of green plant in cattle (Odriozola, 2003). Besides that, the absence of clinical signs or biochemical findings related to liver damage in group 1 with a toxic dose of 4 g/kg of dry matter suggests that the toxic dose must be higher than in cattle and sheep to induce poisoning in goats. rather than in cattle and sheep. However, the differences between these groups can also be due to varying concentrations of carboxyatractylosides in the plants used in this work. That is why we recommend submitting the samples to a reference laboratory to determine the toxin concentration and detect variations that could affect or change any results. Despite differences in the amount of plant used in all the groups, once the minimum toxic dose is ingested, death occurs within 48 hours after consumption (Giannitti et al., 2013). In general, there are no differences between clinical signs in goats, cattle and sheep (Collazo and Riet-Correa, 1996; Micheloud and Odriozola, 2012; Giannitti et al., 2013), and the differences between groups in this work might be due to idiosyncrasy more than for any other reason.
The biochemical evidence showed that the most useful diagnostic tool was the detection of AST (Rodríguez et al., 2003) since its serum levels increased in correspondence with a larger amount of plant ingested by animals. Its early elevation showed a peak at 6 to 24 hours after ingestion. After 24 hours, its serum levels decreased to subnormal values probably because of enzymatic depletion due to massive damage. GGT and ASP did not show a marked elevation, their levels increased only after 24 hours of ingestion and remained high until the death of the animals, thus, their utility is reduced in cases like this, where clinical signs and death happen before serum levels increase can be detected (Kaneko et al., 2008).
The most consistent pathologic finding was centrilobular hepatic necrosis, macroscopically characterized by an enhanced reticular pattern. This pattern was more evident in animals with a longer clinical course. Microscopically, centrilobular necrosis was seen in all cases, but it was more severe in animals 7 and 8 from group 2 and animal 12 from group 3. In these mentioned cases, hemorrhages were also found in correspondence with necrotic areas. Animals 9 from group 2 and 10 and 11 from group 3 also showed extensive damage but with no vascular changes. Thus, two different patterns of centrilobular necrosis can be defined: a hemorrhagic and a non-hemorrhagic pattern. It might be possible that these patterns are associated with the disease evolution in each animal rather than with the amount of plant ingested. Changes in the central nervous system might be attributed to hepatic encephalopathy (Butterworth et al., 2009).
CONCLUSIONS
This work allowed us to demonstrate the susceptibility of goats to P. glauca poisoning. A toxic dose was also determined, being significantly higher than the dose needed to cause the disease in cattle and sheep. Additionally, a clinicopathological characterization was made and, except for certain differences, clinical signs are similar to cattle (Rivero et al., 2010) and sheep (Liboreiro et al., 2021). Although these findings cannot be conclusive, goats seem to be more resistant to P. glauca poisoning than other species. In accordance with case studies from the Diagnostic Service INTA-Salta, poisoning outbreaks are frequently observed in mixed flocks containing sheep and goats, where animals ingest the plant directly from the ground, but deaths occur only in sheep (Medina et al., 2022). This supposed resistance in goats can also be related to some metabolic and disposition profile differences between goats, sheep and cattle previously demonstrated with some drugs (Galtier et al., 1981; Bogan et al., 1987; Sundlof and Whitlock, 1992; Alvinerie et al., 1994). In these cited cases, goats proved to have a faster and more effective mechanism to metabolize and eliminate certain compounds rather than the other species. For these reasons, we consider that it is important to keep studying goats concerning plant poisoning to delve into the physiopathology and toxicokinetics differences between this species and other ruminants and thus be able to explain certain differences regarding toxic doses and clinical presentations.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.














