KEY POINTS
Current knowledge
• Fracture healing and nonunion management is concep tually understood as the diamond: osteogenic, osteo inductive, osteoconductive, and biomechanical. It was introduced in 2007 and has gained popularity and util ity. Hydroxyapatite is the most abundant and strongest component in bone.
Contribution to the current knowledge
• This article highlights other facets in fracture healing that lead to a 6-facet diamond with the inclusion of osteoim munology and vascularity, mirroring the hydroxyapatite strong constitution.
Bone structure dynamically responds to common daily basic loading and straining activities (e.g., walk ing, running, swimming) and in extreme circumstances such as trauma (i.e., fractures), tumors, and surgery. The capacity to respond and heal is finely tuned and reflected by its natural composition: residing cells (e.g., osteoblasts, osteocytes, osteoclasts, progenitor cells) and extracellular matrix1. Of note, the extracellular matrix is constituted of approximately 40% organic (e.g., collagens, and non-collagenous proteins) and 60% inorganic matrix, which major inorganic constituent is hydroxyapatite (HA, Ca5(PO4)3OH), providing compressive strength1-3. Non-collagenous proteins allow biomineralization, while the col lagen provides the structured template for hydroxyapatite deposition1,4. From a chemical perspective, hydroxyapatite is structurally an hexagon2,3,5-7. It has been widely studied and implemented in many orthopaedic procedures and implants due to its bioactive profile5,7-12.
The fracture healing, from a secondary healing per spective, can be schematically divided into three overlap ping biological phases: inflammatory, repair, and remod eling. This involves intramembranous and endochondral ossification mechanisms of bone formation, which is determined by fracture gap and strain13. Giannoudis et al work, entitled “Fracture healing: The diamond concept”, has transcended the vast literature gaining recognition in such appreciation of fracture healing and setting a model or template for understanding fracture healing phenomenon and for the management of nonunion (up to 10% of all fractures)14. It has received wide acceptance in the orthopaedic world as a framework for analysis of nonunions and as a decision-making tool when planning multifaceted interventions, more so in the setting of signifi cant bone defects or recalcitrant nonunions. This 4-corner work is conceptually framing bone healing as a “diamond” by osteogenic cells (i.e., mesenchymal cells, progenitor cells), an osteoconductive scaffold (i.e., grafts, synthetic fillers), growth factors (i.e., osteoinductive cytokines) and lastly the biomechanical environment (i.e., strain, stability, cellular mechanoreceptors)12,15-18. The aforementioned conceptual frame of bone healing and nonunion treatment called the “diamond” concept comes short in reality and does have a number of additional facets to the original four that are worth mentioning, such as vascularity of the zone of injury, the containment of the graft, the timing of intervention, the profile of the patient (i.e., age, comorbidi ties, immune system) and surgical technique17.
The diamonds per-se have multiple facets, corners, and edges, and they can be represented by many polygons in nature. Probably, the strongest shape is the hexagon due to its mechanical strength and stability, just like the hy droxyapatite in bone19-22. Various structures, correspond ingly the hexagon (e.g., honeycombs), in nature are not a coincidence and have inspired mankind to replicate in engineering due to their structural stability and reliability23.
Though from basic science knowledge advancement in the case of osteoimmunology and procedural practice in the case of vascularity; these facets seem individual factors, other than part of the whole fracture healing “diamond” concept. Hence, this work outlines these two other facets that should be integrated and recognized into the conceptual framework “diamond” model when treating fractures. This work will not delve into types of nonunion, nor infection, or patient comorbidities that oftentimes compromise fracture healing17.This article will provide an overview of the original 4 facets of the “diamond” model, and will explore more in-depth the proposed ones that configure an hexagon14.
The four-facet diamond
Biomechanical environment
Strain is a relative measure of deformation an object has in response to loading and is influenced by stability. In the clinical setting, stability at the fracture surfaces is the degree of load-dependent displacement16. When a fracture occurs, the load transmission is affected, the he matoma fills the gap and eventual callus formation takes place. The degree of motion at the fracture surfaces will determine the strain and is fundamental for primary or secondary bone healing17. Primary bone healing occurs where there is absolute stability, defined as bone surface contact < 0.15 mm or strain < 2%. It occurs primarily as intramembranous ossification and can be seen in non-displaced fractures or with anatomic reduction and fixa tion techniques (e.g., compression plate, lag screws)15. Secondary bone healing occurs with relative stability and occurs primarily as endochondral ossification. The initial strain tolerance can be around 100%, but as the callus matures and calcifies the contact area increases, and motion at the fracture decreases, then becoming around 2-10% which is tolerable for healing17. It can be seen in comminuted or displaced unstable fractures, with splint ing or casting, or with non-rigid fixation techniques (e.g., bridge plating, intramedullary nailing)17. If the strain falls outside that range, fracture healing is hampered and may lead to delayed healing or nonunion17. The progenitor/stem cells and residing bone cells (i.e., osteoblasts, osteoclasts) through mechanosensation and mechanotransduction sense and respond to mechanical conditions determin ing their proliferation and the secretion of cytokines and enzymes24. Under appropriate mechanical stimuli stem cells can undergo chondrogenic or osteogenic differen tiation while osteoblasts and osteoclasts tailor the bone resorption/reconstruction balance25. Moreover, in the set ting of osteosynthesis, if implant loosening and instability take place, component wear and abrasions can stimulate macrophages and osteoblasts towards a pro-inflammatory and pro-osteolytic activity26.
Osteogenic cells
At the fracture hematoma, the advent of neighboring or local progenitor and stem cells (i.e., from the periosteum, bone marrow, muscle) responds to the extracellular matrix debris, growth factors, and cytokines15. This parallel to an initial inflammatory process leads to a progenitor/stem cell proliferative response. There is a concomitant increased vascular permeability that allows more stem and immune cell chemotaxis. The fibrin matrix is progressively replaced by a forming callus due to fibroblasts and osteoclasts. The stem cells found in the callus, depending on the cytokine profile, mechanical strain, and oxygen tension of the en vironment will proliferate and differentiate into osteoblasts [bone morphogenic protein (BMP), lower strain and higher oxygen tension] or chondrocytes (higher strain and lower oxygen tension)25. This results in a combination of a pe ripheral or cortical hard callus tissue (e.g., osteoblasts and collagen I) of predominant intramembranous ossification, and a central or medullary soft callus (chondrocytes and collagen II) of predominant endochondral ossification15.
Osteoconductive scaffold
Naturally, the extracellular matrix provides an environment for cell adhesion, migration, and cues for osteogenic cells. However, when there is a significant gap and impending nonunion, some procedures can be performed to over come this deficiency: autograft, allograft, vascularized bone graft, and Masquelet membrane, among others17. An ideal bone graft has high osteoconductivity, high osteoin ductivity, and high osteogenicity; due to retained structure, and residing factors and cells27,28. Autologous bone grafts remain the gold standard material as it minimizes the risk for rejection and provides a highly osteoconductive and os teoinductive environment (e.g., iliac crest). Notwithstand ing, autografts have significant disadvantages: donor-site morbidity, risk of infection, potential nerve damage, and increased blood loss due to the longer surgical time and reimplantation of the graft28,29. Further, autograft supply is limited in cases of large bone defects and is ultimately not a feasible option for patients with poor bone quality (i.e., osteoporosis). These disadvantages have led to the increased use of cadaveric bone allografts27. These un dergo rigorous preparation and the processed bone lacks osteogenic cells and has limited growth factors, which may lead to graft failure27,30. Indeed, low osteoconductivity and low osteoinductivity of commercially available allografts have been reported as reasons for failure in animal mod els of spinal fusion31. To overcome this limitation and enhance stable bone formation and fusion, there has been an interest in developing biologic adjuvant therapies for allografts or graft alternatives such as growth factor supplementation and/or adding osteoprogenitor cells32,33. Regarding graft alternatives, such as synthetic grafts (e.g., coralline, silicate ceramic, tricalcium phosphate), mimic the mineral portion of bone but cannot provide an opti mal healing environment27. However, the demineralized bone matrix alternative is an allograft-derived substance containing primarily collagen I and BMPs, hence is both osteoconductive and osteoinductive in the presence of progenitor cells27.
Osteoinduction
As the fracture hematoma develops, a vast repertoire of signaling molecules such as interleukins (IL) and growth factors are spilled locally and systemically. They are secreted by platelets, macrophages, stem cells, chondrocytes, fibroblasts, osteoblasts, and endothelial cells14. These factors initiate and orchestrate cellular events in the healing environment. They guide stem cell proliferation and differentiation. Though multiple cells secrete these factors, they are extremely intertwined in the “stem-immune” cell cross-talk defined below26,34. The most remarkable factors that promote osteogenesis are insulin-like growth factor (IGF), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), and transforming growth factor-beta (TGFβ). The latter includes BMPs35.
Towards the hexagon
Osteoimmunology
The role of immunology in the bone microenvironment can be often overlooked. It has become an entity by itself but in extreme relationship with other factors. Bone is in a constant dynamic process of resorption and reabsorp tion, in which maximum magnitude of expression could be reflected by the fracture and its healing phases. Both, the immune system and bone remodeling cells are intertwined in a cross-talk that regulates each other, termed osteoim munology by Arron and Choi (Figure 1)26,36,37. The fracture leads to the formation of hematoma which temporarily acts as a scaffold or matrix rich in cytokines, immune cells, and progenitor cells38.Polymorphonuclear cells are the first to intervene in a stepwise fashion followed by macrophages and lymphocytes. These secrete chemo kines [i.e., IL-1, IL-6, IL-10, alpha tumornecrosis factor (TNFα), monocyte chemoattractant protein 1 (MCP-1), alpha chemokine CXC ligand-1 (CXCL-1α), macrophage inflammatory protein 1 (MIP-1)] that attract and activate monocytes and macrophages34. The Osteomacs are resid ing peri-fracture macrophages that take a quick participa tory influence in initiating intramembranous ossification; whereas inflammatory macrophages are those recruited during endochondral ossification13. In addition, from a phe notypic perspective, two different types of macrophages have been identified. Although it represents a simplistic bipolar manner, and more types have been recognized, M1 and M2 represent an antagonistic though necessary interaction for proper healing, which is also present in other tissues34. M1 has a predominant inflammatory action and secretes pro-inflammatory cytokines (i.e., IL-1, IL-6, TNFα, MCP-1) and aids in clearing debris, while M2 has a predominant anti-inflammatory effect [i.e.,IL-10, TGFβ, BMP2, vascular endothelial growth factor (VEGF)], en hance mesenchymal cell recruitment and lead to regenera tion13,26,34,39. The receptor activator of nuclear factor Kappa ligand (RANKL) - osteoprotegerin (OPG) signaling axis is fundamental in osteoclastogenesis and the inflammatory or anti-inflammatory cytokine profile will modulate bone resorption40. Both factors RANKL/OPG are synthesized by mature osteoblasts and osteoblast precursors, but inflammatory cells produce them too40. In the cascade of events, the lymphocytes are recruited and can be broadly divided into T and B populations. For instance, the T-lymphocytes secrete RANKL and IL-17 which recruit and activate osteoclasts, whereas B-lymphocytes besides dampening the inflammation, produce OPG which down regulates osteoclasts further. Moreover, IL-17 enhances mesenchymal stem cells’ anti-inflammatory activity and aid in osteoblastic maturation34. Vast literature shows that an unbalanced immune response leads to deficient fracture healing (e.g., diabetes, rheumatoid arthritis and lupus), as well as mechanical instability can perpetuate inflam mation and osteolysis (e.g., inappropriate bone fixation, inadequate strain and component wear)16,26.
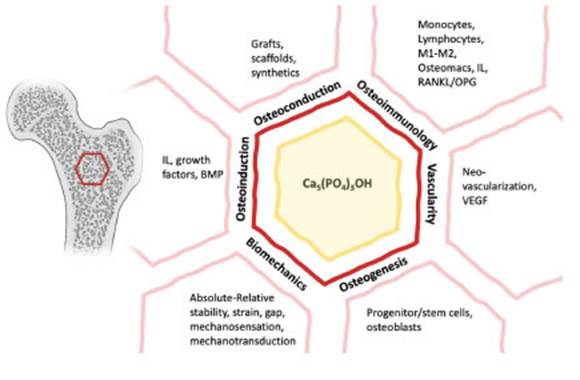
Fig. 1 Progression to the hexagon-diamond model with the inclusion of osteoimmunology and vascu larity. Note: IL, Interleukin; BMP, bone morphogenic protein; Ca5(PO4)3OH, hydroxyapatite; M1-M2, macrophages 1-2; RANKL/OPG, receptor activator of nuclear factor Kappa ligand/osteoprotegerin; VEGF, vascular endothelial growth factor.
Vascularity
In another facet of our so-called hexagonal “diamond”, vascularity is fundamental for healing as well (Figure 1). Angiogenesis is crucial during endochondral ossification for fracture healing and physeal bone growth41. Its inhibi tion leads to fibrous tissue and nonunion42. Awareness in the scientific community of the VEGF pathway in endo chondral and intramembranous ossification has gained popularity and is probably the most important41,43,44. Within the fracture hematoma, VEGF allows neovascularization in a relative injured hypoxic environment allowing soft callus formation through the delivery of trophic factors, nutrients, and osteoprogenitor cells38,45. Other factors are also implicated in angiogenesis and bone healing such as FGF, IGF, and placental-GF, which in their absence hinder fracture healing and normal bone growth45. Aside from the microenvironment, the blood supply for most long bones can be divided into periosteal and endosteal circulation, both nurturing the cortical bone (outer 1/3 and inner 2/3, respectively)46,47. The endosteal circulation can be com promised when intramedullary fixation is performed, but this is compensated by the periosteal circulation vessels’ proliferative response in the following weeks after sur gery48. A metanalysis focused on the vascular anatomy of lower extremity long bones found the distal third segment to have the poorest vascular supply, and speculate that this could explain the higher nonunion rate in this region47. Hence, the importance of the zone of injury at a fracture site and the amount of soft tissue damage to the perios teum (i.e., periosteal circulation). The periosteum also provides a substrate of local trophic factors and progenitor cells49. This led to a better understanding of the importance of surgical techniques and choice of types of fixation (e.g., intramedullary versus extramedullary implants), as well as the principle of minimizing periosteal stripping during surgery49-53. For example, when convenient, the benefit of using intramedullary nailing and minimally invasive plate osteosynthesis minimize fracture exposure, and soft tis sue stripping, preserving local vascularity and osteogenic/ osteoinductive factors50,51.














