Birt-Hogg-Dubé (BHD) syndrome is a geno dermatosis of autosomal dominant inheritance characterized by genetic mutations in the FLCN gene located in chromosome 17p11.21,2. It encodes foliculin, a protein that through an inap propriate either inhibition or activation of the mTOR pathway may cause skin lesions, renal cancer, and characteristic lung lesions3.
Clinically, it can be observed in the skin as fibrofolliculomas. Histologically, this is a lesion typically vertical or perpendicular to the epi dermis, centered in the hair follicle, and that presents thin epithelial projections4. It should be noted that despite the fact that skin lesions are a warning sign for a dermatologist, not all patients with Birt-Hogg-Dubé syndrome present them.
Within the extracutaneous manifestations, there is a predisposition to present renal tumors which are characteristically bilateral and multi focal, mostly of the histological type of hybrid forms of oncocytoma and chromophobe carci noma4,5.
Regarding pulmonary compromise, up to 80% of patients with this syndrome have lung cysts which may be asymptomatic but that could have up to 50 times more risk of developing pneumo thorax compared to the normal population. The location of the cysts is mainly in the lung bases and at the subpleural level.
Clinical case
We present the case of a 38-year-old male with bron chial asthma treated with budesonide (160 mcg) and for moterol (4.5 mcg) and no history of smoking, who con sulted for isolated hemoptysis event, three weeks after recovery from COVID-19 infection, with a presumptive di agnosis of post-COVID-19 pulmonary complication.
A chest tomography was requested, showing multiple bilateral, basal and adjacent to mediastinum air cyst. The biggest one (28 × 27 mm) in the left lower lobe (Fig. 1A). The lesions were followed up for 3 months, a period in which did not show changes in its size, nor association with interstitial lesions. Neither was evidence of compro mise in computed tomography of the abdomen and pelvis with intravenous contrast, excluding kidney affectation. In this context, different diagnoses were proposed, such as Langerhans cell histiocytosis, lymphangioleiomyoma tosis, lymphocytic interstitial pneumonia and BHD syn drome (Table 1).
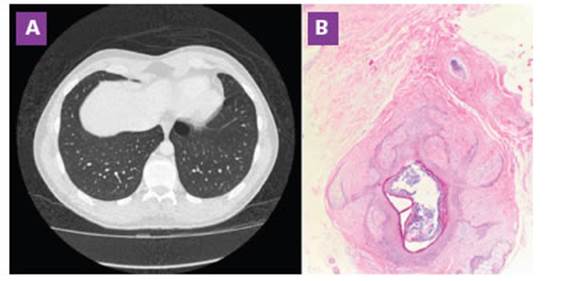
Figure 1 A: Chest tomography. Left lower lobe air cyst (28 x 27 mm) B: Skin biopsy (Hematoxylin/eosin stain x40): circumscribed tumoral lesion with central cystic cavity surrounded by concentrically arranged collagen fibers
During the follow-up of the patient, physical exami nation revealed firm, whitish, papular skin lesions 2 to 4 mm in diameter, asymptomatic throughout the thorax. An interconsultation with Dermatology was requested, who biopsed this lesion, resulting in a circumscribed tumor lesion with a central cystic cavity surrounded by concentrically arranged collagen fibers, compatible with the diagnosis of fibrofolliculoma (Fig. 1B).
Within the family history of the patient, there was a maternal aunt who presented spontaneous pneumotho rax on three occasions; a maternal grandmother with recurrent adrenal carcinoma; and maternal grandfather with leukemia.
Given the suspicion of a genetic disorder associated with skin lesions and cystic lung lesions, a multi-genetic panel was requested (BRCA1/BRCA2/TP53/FLCN) resulting in a pathological variant of the FLCN gene (exon 1 deletion, heterozygous) associated with autosomal dominant BHD syndrome. This variant is a large deletion that occurs in a non-coding region of the FLCN gene. It does not change the encoded amino acid sequence of the FLCN protein. Studies have shown that a similar copy number variant alters FLCN gene expression6. For these reasons, this vari ant has been classified as pathogenic. In this context, the molecular finding explained the clinical condition. Genetic counseling was requested from relatives: both the grandmother and the aunt were diagnosed with the disease. His mother, during the follow-up process, under went a chest tomography that showed cystic lesions in the lungs, but the genetic study is still in process. These patients are currently under clinical follow-up, and fam ily screening is going on.
The patient did not repeat new episodes of hemop tysis.
Discussion
The follow-up of patients after suffering from SARS-CoV-2 virus disease has led us to carry out imaging studies that can generate the diagnosis of incidentalomas, which require a differential diagnosis approach and follow-up to carry out an early treatment if necessary7.
The management of BHD syndrome must be carried out by a multidisciplinary group of physicians. The two essential components of the clinical follow up of the BHD syndrome are the management of recurrent spontane ous pneumothorax and screening of kidney tumors. The high probability of pneumotho rax is well known, nearly 30% will have it as personal medical history before 40 years old. The optimal method and timing of treating BHD associated pneumothorax is unclear and controversies exist as randomized prospective trials are lacking. Some reviews suggested that BHD-associated pneumothorax should be managed in the same way as in the general population; others argue that with the lower likelihood of spontaneous resolution and re currence, pneumothoraxes should be man aged more aggressively8,9.
Renal tumors are usually multiple and bilat eral, frequently appearing at a median age of 30 to 40 years. In comparison with most inherited renal cancers syndromes, that are commonly related to a single histologic tumor type, BHD syn drome is related to a wide range of tumor histologies, most typically chromophobe tumors and hybrid chromophobe/oncocytic tumors. Clear cellular carcinoma, papillary carcinoma, and blended-kind carcinoma can also occasionally be seen. Some experts recommend abdominal imaging starting at age 21 (or following diagnosis) and at least every 36 months until a mass is identified, at which time interval imaging is determined in the individual patient based upon the size and growth rate of the tumor. Ultrasonography may miss small isoechoic renal masses, and, therefore, computed tomography imaging or magnetic resonance imaging to minimize radiation exposure should be used when possible5. Favorably, the reported patient did not present pneumothorax or renal involvement.
In conclusion, a new medical paradigm has emerged as a result of the SARS-CoV-2 virus pandemic in a number of ways. The increased neumological follow-up made possible early diagnosis of pathologies less common in everyday practice. This case serves as an illustration of dealing with a challenging pathology in terms of the differential diagnosis of cystic lung diseases. The patient and his family should be treated in a multidisciplinary manner focusing on the treatment of potential complications, avoiding late diagnosis of possible oncological pathology, and providing genetic counseling for the patient and his family10.
It is interesting to note that neither Argen tina’s epidemiological statistics nor internation al management guides specify the screening method, frequency, or ideal age for this condi tion; which provides an excellent opening for future research.















