INTRODUCTION
Coronavirus disease 2019 (COVID-19) is an infec tious disease caused by a strain of coronavirus called severe acute respiratory syndrome corona virus 2 (SARS-CoV-2).1 The first cases were seen in Wuhan, China, in late December 2019, and from there it has spread practically all over the world. It was officially recognized as a pandemic by the World Health Organization (WHO) on March 11, 2020.1-3
According to PAHO (Pan American Health Organization) as of April 1, 2021, the number of confirmed cases worldwide reached 128 million, and more than 2 million deaths. In Argentina, the data available up to now indicated 2,348,821 cases and 55,858 deaths associated with COVID-19.
The disease mainly affects tissues that express high levels of ACE2 (angiotensin converting en zyme 2 receptor), including the lungs, heart, and gastrointestinal tract.2 The presence of underlying comorbidities is associated with a higher risk of progressing to severe COVID-19. Advanced age, cardiovascular disease, diabetes mellitus, arte rial hypertension, chronic obstructive pulmonary disease (COPD), cancer (especially hematologic neoplasms, lung cancer, and metastatic disease), chronic kidney disease, obesity, and smoking have been mentioned as the main risk factors for devel oping severe disease.1,2,4,5
Numerous studies have highlighted the clinical characteristics of the disease, showing that 80% of patients with COVID-19 present mild disease with an overall mortality rate of 2-5%.1,5,6,7,8,9 However, a subset of approximately 15% of patients showed moderate lung involvement and required hospi talization, and 5% had severe respiratory failure, resulting in death in half of these cases.1,6,7,9,10
The analytical alterations that have been asso ciated with the worst prognoses include: lympho penia, elevation of liver enzymes, of LDH (lactate dehydrogenase), of inflammatory markers such as PCR and ferritin, D-dimer, troponin, and CPK (creatine phosphate kinase), prolonged prothrom bin time, and acute kidney injury. Coagulation test alterations have also been observed, with a state of hypercoagulability that conditions a tendency to thrombosis.1,6,11
Mortality rates of severe forms of the disease are relatively high (half of the cases)1,6,7,9,10, so early identification of risk factors related to the severity of the disease could facilitate the imple mentation of timely interventions and probably help reduce the fatality rate.
In this context, the present research was con ducted with the purpose of correlating the initial symptoms of COVID-19 with the severity of the disease. Also, to determine the main comorbidities associated with a poor prognosis and describe the main elevated biomarkers in COVID-19.
MATERIALS AND METHODS
A retrospective, cross-sectional, observational cohort study was conducted in patients diagnosed with COVID-19 who attended the Emergency Department of the Sanatorio Güemes between January 1st and March 31st, 2021, who se diagnosis had been confirmed by reverse transcription polymerase chain reaction (RT-PCR).
Sanatorio Güemes is a high-complexity private health care institution located in the Autonomous City of Buenos Aires, with a unique information repository for each patient, which is centralized through an Electronic Health Record (EHR). All patients, upon admission to the institution, have voluntarily and officially signed informed consent for the use of their data for research purposes. The information of this study was obtained from the EHR database and was entered into an electronic form designed specifically for this purpose, which was accessible only to the authors of the study, thus preserving the anonymity of all participants.
Inclusion criteria: the study included individuals over 18 years of age with a diagnosis of COVID-19 confirmed by RT-PCR in throat and nasopharyngeal swab.
Exclusion criteria: individuals with suspected COVID-19 without confirmation at the time of the analysis and those with positive COVID-19 antibodies (but without RT-PCR or with negative RT-PCR) were excluded. Additionally, pa tients whose medical records didn’t include demographic data, comorbidities, and initial symptoms of the disease, and those who hadn’t been followed up by the Sanatorio Güemes until the time of medical discharge were excluded.
Clinical information, laboratory data, and assessment of disease severity correspond to data obtained upon hospital admission. Initial symptoms were defined as the first symp toms to appear at the onset of the illness. Lymphopenia was defined as a lymphocyte count < 1000/μL or a percentage of less than 20%. Thrombocytopenia was defined as a platelet count < 100,000/mm3; and thrombocytosis, as a count > 450,000/mm3. A PCR value of ≥ 10 mg/l was considered elevated; D-dimer was considered elevated with a value of ≥ 0.5 mg/l; LDH was considered elevated with values of > 250 U/L, and for ferritin, it was defined as elevated with values of > 300 ng/mL in men and > 150 ng/mL in women.
Based on the National Institutes of Health (NIH) guidelines, individuals who test positive for SARS-CoV-2 but don’t have symptoms compatible with COVID-19 are considered to have an asymptomatic infection. Mild disease: individuals who have any of the various signs and symp toms of COVID-19 but do not show breathing difficulty, dyspnea, or abnormal chest imaging. Moderate disease: individuals who show evidence of lower airways illness during clinical evaluation or imaging and have an oxygen saturation (SpO2) ≥ 94% on room air at sea level. Severe disease: individuals who have SpO2 < 94%, arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio of < 300 mmHg, respiratory rate > 30 breaths/min, or pulmonary infiltrates > 50%. Critical disease: people who have respiratory failure, septic shock, and/or multi-organ dysfunction.4 In order to perform statistical analysis with practical relevance for the study’s objectives, the population was divided into three groups: mild disease (asymptomatic and mild cases), moderate disease, and severe disease (severe and critical cases).
Descriptive statistics were used to analyze the median (interquartile range [IQR]), frequencies, and proportions. Categorical variables were compared using the Chi-square test and Fisher’s exact test when the expected frequencies were less than 5. Odds ratio (OR) and 95% confidence in terval (CI) were used to estimate the association between initial symptoms, comorbidities, and severe COVID-19 (p <0.05 was considered statistically significant). Statisti cal analyses were performed using SPSS software (version 26.0).
RESULTS
A total of 413 patients with diagnosis of COVID-19 confirmed by RT-PCR were included. The median age was 37 years ([IQR] 28-48), 228 cases (55.2%) were female, and 185 (44.8%) were male. A total of 154 cases (37.3%) presented with one or more comorbidities at the time of diagnosis (Table 1).
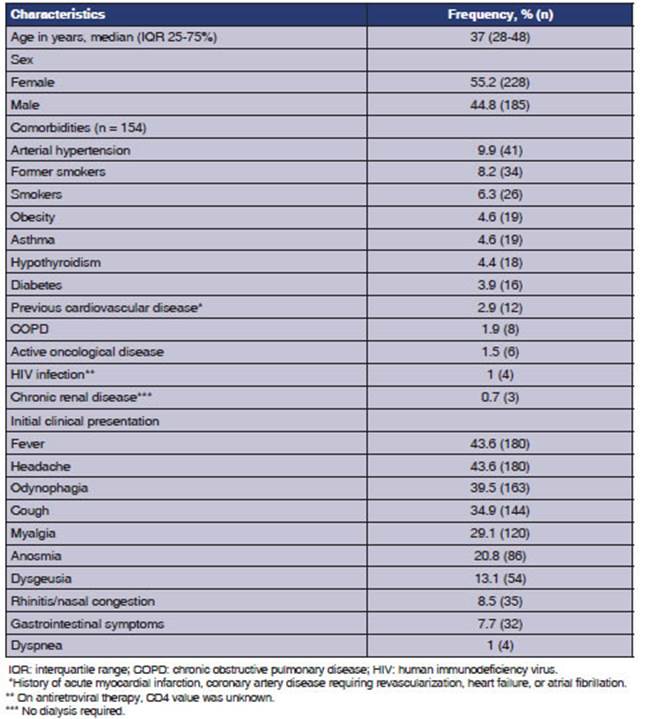
Table 1 General characteristics of patients, comorbidities and initial clinical presentation (n = 413)
Among the main comorbidities, 9.9% were hy pertensive, 8.2% former smokers, 6.3% current smokers, 4.6% asthmatic, and 4.6% obese. The remaining characteristics are detailed in Table 1.
The initial clinical presentation was character ized by the presence of fever and headache, each in 43.6% of cases; 39.5% had odynophagia, 34.9% cough, 29.1% myalgia, 20.8% anosmia, 13.1% dysgeusia, 8.5% rhinitis/nasal congestion, 7.7% gastrointestinal symptoms, and only 1% dyspnea.
Regarding laboratory findings, these were requested according to the treating physician’s criteria, so we do not have data on all patients. LDH was requested in 73 cases, of which 91.8% had elevated LDH; the PCR was elevated in 85.7% of 77 patients; elevated ferritin was found in 85.1% of 47 cases; D-dimer was elevated in 42.3% of 26 patients; lymphopenia was found in 31.1% of 90 cases; thrombocytopenia was present in 3.4% of 89 patients, and no evidence of thrombocytosis was found in any of the studies.
Of the 413 patients, 82.6% (341) were indicated home medical care and daily telephone follow-up; 16.9% required hospitalization in a general ward, and 0.5% had to be hospitalized at the intensive care unit (ICU) upon their admission to the insti tution. The median time from symptom onset to medical consultation was 3 days (IQR 2-4).
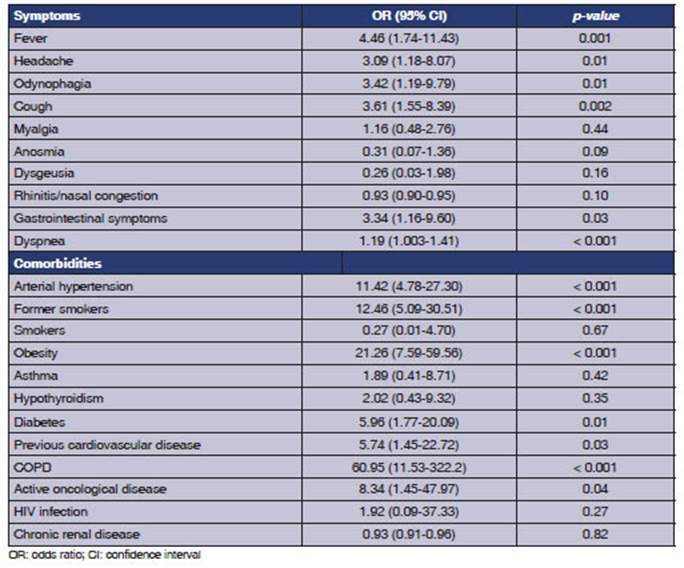
81.1% developed mild disease, 12.8% moderate disease, and 6.1% severe disease. When correlating initial symptoms with severe COVID-19, the sever ity of the disease was significantly associated with dyspnea (OR = 1.19, CI: 1.003-1.41, p < 0.001), fever (OR = 4.46, CI: 1.74-11.43, p 0.001), cough (OR = 3.61, CI: 1.55-8.39, p 0.002), odynopha gia (OR =3.42, CI: 1.19-9.79, p 0.01), headache (OR = 3.09, CI: 1.18-8.07), and gastrointestinal symptoms (OR = 3.34, CI: 1.16-9.60, p 0.03) (Table 2).
As for comorbidities, those significantly associ ated with severe disease were arterial hypertension (OR = 11.42, CI: 4.78 - 27.30, p < 0.001), obesity (OR = 21.26, CI: 7.59 - 59.56, p < 0.001), COPD (OR = 60.95, CI: 11.53 - 322.2, p < 0.001), former smoking (OR = 12.46, CI: 5.09 - 30.51, p < 0.001), diabetes (OR = 5.96, CI: 1.77-0.09, p 0.01), previ ous cardiovascular disease (OR = 5.74, CI: 1.45- 22.72, p 0.03), and active oncological disease (OR = 8.34, CI: 1.45 - 47.97, p 0.04) (Table 2).
All patients who received home hospitalization completed their telephone follow-up without any reported deaths or need for hospitalization. Among the patients who required hospitalization (72 cases), the median length of hospital stay was 6 days (IQR 3.25-9.75). Of those initially hospitalized in a general ward (70 cases), 11.4% subsequently required ICU care. Mortality among hospitalized patients was 8.3% (6 cases). All 6 deceased patients required ICU stay and mechanical ventilation, and they all showed symptoms and comorbidities asso ciated with greater severity. The overall mortality rate was 1.5% (6/413 patients).
DISCUSSION
Our work describes the main clinical character istics, comorbidities, and commonly requested laboratory tests in cases of COVID-19 and the as sociation between initial symptoms and comorbidi ties that are related to the severity of the disease. As relevant data, 37.3% showed comorbidities at the time of diagnosis, with arterial hypertension being the main one, followed by a history of past smoking, current smoking, and obesity; similar to what has been observed in national studies such as that of Pulido et al.12 The first symptoms to ap pear were fever and headache in 43.6% of cases, followed by odynophagia and cough. These data are consistent with several studies published in the literature.3,5-7,13,14
Out of our entire population, 81.1% developed a mild disease, 12.8% moderate, and 6.1% had severe disease. These data are related to several studies that showed that 80% of patients with COVID-19 show mild disease.1,5-8 15% of patients had moder ate lung involvement, which required hospitaliza tion, and 5% had severe respiratory failure, which resulted in death in half of these cases.1,6,7,9,10
As a relevant finding of our study, we found that the initial symptoms of COVID-19 that are associated with the development of severe disease are: dyspnea (p < 0.001), fever (p 0.001), cough (p 0.002), odynophagia (p 0.01), headache (p 0.01), and gastrointestinal symptoms (p 0.03). Likewise, comorbidities associated with the worst prognoses were: arterial hypertension (p < 0.001), obesity (p < 0.001), COPD (p < 0.001), former smoking (p < 0.001), diabetes (p 0.01), previous cardio vascular disease (p 0.03), and active oncological disease (p 0.04).
As of the writing of this study, there are no national publications that assess the initial symp toms and their relationship with the severity of the disease. When comparing our results with the international literature, we saw that He et al3 found in a population of 3,326 patients (se lected from 20 articles, all based on the Chinese population), that from the initial symptoms, cough (p < 0.001), fever (p < 0.001), dyspnea (< 0.001), diarrhea (p < 0.001), fatigue (p < 0.01), expectora tion (p < 0.01), myalgia (p < 0.001), hemoptysis (p < 0.001), abdominal pain (p < 0.001), and an orexia (p < 0.001) were associated with the risk of progressing to severe disease.
Rubio et al7 conducted a cluster analysis of the Spanish SEMI-COVID-19 registry including 11 phenotypic variables in a cohort of 12,066 hospitalized patients. The study identified 4 phe notypic groups that predicted hospital prognosis. The groups associated with poor prognosis were C1 (cases that showed the isolated triad of fever, cough, and dyspnea) and C4 (diarrhea, vomiting, and/or abdominal pain). On the other hand, pa tients who belonged to group C2 (dysgeusia and/ or anosmia) showed the best prognosis, along with group C3 (arthromyalgia, headache, and odynophagia).
Regarding comorbidities, several studies on dif ferent population groups have described that the presence of these comorbidities is associated with a higher risk of progressing to severe COVID-19. Advanced age, cardiovascular disease, diabetes mellitus, arterial hypertension, COPD, cancer, chronic kidney disease, obesity, and smoking are the main risk factors associated with severe COVID-19.1,2,4,5 Our findings mostly agree with published data, however; in contrast, former smoking (p < 0.001) was associated with a risk of severe disease. This result is consistent with what was published by Zhang et al15 in a meta-analysis of 25 articles, which suggested that there was a sig nificant relationship between former smokers and the risk of severe COVID-19, compared to current smokers (combined OR: 1.85, 95% CI: 1.33-2.55, I2 = 67.7%, p < 0.001).
Different studies have identified the main bio markers that are associated with a higher risk of developing a worse course of the disease. In this regard, Tjendra et al5 conducted a review of 43 cohort studies and 11 systematic reviews (includ ing the meta-analysis) where they found that upon admission, most patients showed lymphopenia, neutrophilia, thrombocytopenia, prolonged PT, elevated D-dimer, high lactate levels, lower oxygen saturation, high neutrophil-to-lymphocyte ratio (> 5.0), and high systemic immune inflammation index (> 500). All patients had high initial levels of PCR and IL-6 (> 10 pg/ml), and a large subset of patients had elevated levels of LDH, D-dimer, procalcitonin, and ferritin. The presence of these abnormalities was associated with critical illness. Similarly, Zhou et al14 conducted a retrospective cohort study including 191 patients with COVID-19. In comparison to survivors, non-survivors more frequently showed high LDH (p < 0.001), high procalcitonin (p < 0.001), increased levels of serum ferritin (p < 0.001), and elevated IL-6 (p < 0.001). When comparing these results with those obtained in our study, all patients who developed severe disease had mainly elevated levels of LDH, PCR, ferritin, and D-dimer.
The overall mortality rate was 1.5%, similar to what has been reported in different publications that mention a fatality rate between 1.4% and 4.3%.16,17
Our study has some limitations. As a retrospec tive study, collected data comes from electronic medical records, which are subject to biases that are inherent in data collection. The results only correspond to the experience of a single hospital center rather than multiple centers. In addition, several records were excluded because they did not have the minimum data necessary for the study or medical follow-up until final discharge. Biomarkers were requested according to the treat ing physician’s criteria, so we do not have that particular data on all patients. Although we found significant associations with disease severity, more multicenter studies are necessary, with a larger number of participants, to replicate our findings in a large percentage of the national population.
In conclusion, patients diagnosed with CO VID-19 whose initial manifestations include dyspnea, fever, cough, odynophagia, headache, and gastrointestinal symptoms have higher risk of developing a severe course of the disease. High blood pressure, obesity, COPD, former smoking, diabetes, previous cardiovascular disease, and active oncological disease are associated with a poor prognosis.
In order to prevent disease deterioration, it is vital to identify initial symptoms and comorbidi ties related to poor outcomes as soon as possible