INTRODUCTION
Renal transplantation, partial nephrectomy, revascularization of renal artery, trauma, and hydronephrosis can cause ischemic renal injury which is the common cause of acute renal failure. Also, renal ischemia may result in chronic renal failure1-2 Although restoration of blood flow saves kidney from damage, reperfusion-induced nephrotoxicity may occur in kidney. Previous studies have shown that NO (nitric oxide) and reactive oxygen species (ROS) play a role in ischemic injury.3-4 These ROS (excessive production) can damage most of the cellular components.5 Abundant amounts of oxygen (O2) which is present in the reperfused ischemic tissue induces excessive free oxygen radicals which reduce antioxidant defense mechanisms.6 In addition, inflammation as well as free oxygen radicals have been shown to be responsible for the severity of ischemia/reperfusion (I/R) injury.7 Inflammation with I/R injury is considered "aseptic inflammation".8 Aseptic inflammation, like septic inflammation, is accompanied by an accumulation of neutrophils and macrophages and an increase in proinflammatory mediators.9 It also increases the activity of inducible nitric oxide synthase (iNOS) and causes NO formation.10 In the light of previous studies antioxidant and anti-inflammatory activity may be beneficial in pre-reperfusion and post-reperfusion periods of ischemic tissue.
Naringin is a flavonoid that is anti-inflammatory, antiviral, anticancer, antimutagenic, antiallergic, analgesic, hypotensive activity, blood cholesterol-lowering effect, and has anti-inflammatory activity at doses of 100 mg/kg.11 In a study, naringin was shown to inhibit the NF-κB / COX-2-caspase-1 pathway.12 However, it has been reported that the enzyme iNOS reduces activity by decreasing NO production.13 For this purpose, we aimed to reveal the possible protective effect of naringin against ischemia-reperfusion induced renal injury according to evidence of biochemical and enzyme analyses.
MATERIALS AND METHODS
Twenty-four Wistar albino female rats weighing 250-300 g were obtained from Cukurova University Animals Research Center. They were placed in a controlled room with controlled temperature (21 ± 2°C) and humidity (60 ± 5%), in which a 12:12 hour light-dark cycle was maintained. The rats were fed with standard commercial pellets and water ad libitum. This study was approved by the Animal Care Committee and Ethics Committee of Çukurova University. All of the procedures were performed according to accepted standards of Guide for the Care and Use of Laboratory Animals. The rats were randomly divided into three groups: (1) control group (C), in which the rats were only performed right nephrectomy (n=8); (2) right nephrectomy and left kidney ischemia (1 h) and reperfusion (24 h) group (I/R) (n=8); (3) I/R/N group received 50 mg/kg naringin orally once a day for two weeks before I/R (n=8). Intraperitoneal (IP) ketamine (75 mg/kg) and xylazine (8 mg/kg) were preferred as anesthesia method for rats. After right nephrectomy through the right dorsolateral incisions were done in all rats, the left renal artery and vein were isolated for 30 minutes without further surgical intervention to allow recirculation in rats in the I/R groups. Later, ischemia was induced by clamping the left renal vessels for 1 hour. Left kidney reperfusion was achieved during 24 h reperfusion. After the bleeding control, the skin and subcutaneous tissues were sutured and the surgical procedure was terminated. All rats were decapitated 24 hours after surgery. Blood samples were taken for biochemical analysis in the tail veins of each rat just before decapitation. After decapitation, the left kidneys were quickly isolated and stored at -80°C for Quantitative Analysis by ELISA.
Quantitative Analysis
Tissue Homogenization
Kidney samples were homogenized by modified RIPA Buffer and by using ultrasonication on ice. Then tissue homogenates were centrifuged (10.000 rpm +4ºC) for 10 minutes, and pellets were discarded, and supernatants were saved for quantitative analyses (ELISA).
Total Protein Analyses
We used the Bradford method for the determination of total protein levels and the standardization of samples of homogenization.
Quantitative Analyses of Homogenizates
Expression of cyclooxygenase-2 (COX-2), cytosolic phospholipase A2 (cPLA2), inducible nitric oxide synthase (iNOS), caspase-3, B- cell lymphoma-2 (BCL-2), Bcl-2 associated x protein (Bax), serum creatinine (Cr), tumor necrosis factor α (TNF-α), interleukin 6 (IL-6) were measured by using enzyme-linked immunosorbent assay (ELISA) (Shanghai Sunred Biological Technology Co. Ltd) kits according to the manufacturer’s instructions.
STATISTICS
Data from samples were shown as means ± S.E.M., and differences between groups were tested by analysis of variance (ANOVA) corrected for Bonferroni multiple comparisons. P values less than 0.05 were considered to be significant.
RESULTS
Effect of naringin on the serum parameters
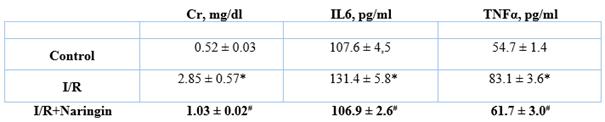
The serum parameters are summarized in Table 1. The serum levels of Cr, IL6, and TNFα were significantly higher in the I/R group when compared to the C group. Naringin treatment reduced levels of Cr, IL6 and TNFα when compared to I/R group, respectively.
Effect of naringin on inflammatory and apoptotic mediator in kidney
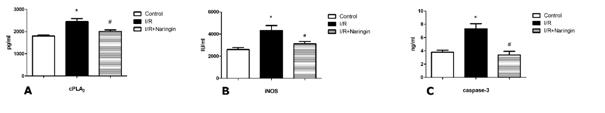
While renal I/R caused a decrease of bcl-2 (1.72 ± 0.20 pg/ml) levels, and increased COX-2 (11882 ± 642 pg/ml), cPLA2 (2448 ± 139 pg/ml), iNOS (4331 ± 438 IU/ml), cleaved caspase-3 (7.33 ± 0.76 ng/ml) and Bax (2.33 ± 0.44 ng/ml) levels, the treatment of naringin reversed these kidney effects (7.47 ± 60.35 pg/ml; 9299 ± 327 pg/ml; 2001 ± 78 pg/ml; 3112 ± 220 IU/ml; 3.38 ± 0.54 ng/ml; 2.33 ± 0.44 ng/ml, respectively) (p <0.05). (Figure 1-2)
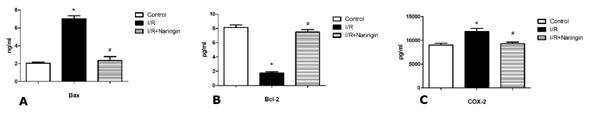
Figure 1: 1A. Effect of naringin on I/R induced Bax expression (n=8). 1B. Effect of naringin on I/R induced Bcl-2 expression (n=8). 1C Effect of naringin on I/R induced COX expression (n=8). Statistical analysis: ANOVA. Post hoc: Bonferroni. (*: for control p < 0.05, #: for I/R p < 0.05)
DISCUSSION
In this study, we evaluated inflammatory and apoptotic mediators together. Studies have shown that renal ischemia causes an increase in COX-2, cPLA2 and iNOS enzymes, which are inflammatory mediators.(10, 14) Renal ischemia increased pro-apoptotic enzymes, which are cleaved caspase-3 and Bax while decreased antiapoptotic enzymes, which is bcl-2. This study showed that the use of naringin decreases renal I/R injury. In this study, while one kidney was nephrectomized, the artery of the other kidney remained clamped for one hour to create I/R. Because the peak serum Cr following ischemia occurs at 24-hour of reperfusion, reperfusion was evaluated after 24 hours in our study.15
Inflammation has an important role in nephrotoxicity.16 The COX-2 enzyme, one of the inflammatory mediators of nephrotoxicity, and the prostaglandins synthesized by this enzyme also play a role. Studies have shown that selective COX-2 inhibitors attenuate severity of nephrotoxicity.17 In a study, naringin was shown to inhibit the NF-κB/COX-2-caspase-1 pathway.12 The expression of inducible iNOS, particularly in inflammatory cells, can have detrimental effects, including direct effects of NO on various proteins and enzymes. It is also associated with the generation of NO, reactive oxygen species (ROS), peroxynitrite and other reactive species.18 In a study by Gutiérrez-Venegas, they found that naringin attenuated inflammatory events by inhibiting the expression of iNOS, reducing NO production.13) Studies have also shown that reperfusion induces oxidative stress which increases intracellular calcium level and increased calcium level leads to elevation of the activity of phospholipase A2 enzyme.19 Oxidative stress is induced by excessive ROS and free superoxide radicals. ROS both plays role as a mediator and in the damage of cellular components. While it acts as a second messenger in the signal transduction pathways cause damage cell in excessive production.20 In excessive production of ROS antioxidant defense system of cell fails. In a study showed that oxygen species also modulates NF-kB,p53 and AP-1.21 H2O2 which is a member of ROS plays a role in various physiological stimuli.21 Oxidative stress plays a pivotal role in apoptosis. Apoptosis is essential for cellular function, survival and shaping of organs. It is also known as programmed cell death. Studies show that antioxidants block or delay apoptosis. Bcl-2, a member of apoptotic pathway, is an anti-apoptotic mediator and this mediator prevents apoptosis by an anti-oxidative mechanism.22 Studies have shown that naringin has great antioxidant capacity.23
In a study, Amini et al. aimed the effects of co-administration of trimetazidine and narigin on renal I/R injury in rat models. Administration of the trimetazidine, narigin, and their combination decreased the plasma level of Bax mRNA expression, microRNA-10a, and caspase-3, but increased Bcl-2 mRNA expression in the kidney tissue. In addition, antioxidant activity, renal blood flow and Cr clearance were improved. They concluded that trimetazidine, narigin, and their combination might be beneficial as potent therapeutic factors against renal I/R injury.24 In a study by Singh et al., investigating the protective effect of naringin against renal I/R injury in a rat model, they found that naringin alleviated renal dysfunction, reduced morphological changes, high thiobarbituric acid reactive substances levels, and restored depleted renal antioxidant enzymes.25 Differently in both studies, in our study, narigin was shown to decrease renal I/R damage with inflammatory and apoptotic mediators such as bcl-2 COX-2 cPLA2, iNOS, cleaved caspase-3 and Bax.
CONCLUSION
In the light of our findings it seems that naringin protects I/R induced nephrotoxicity by decreasing inflammation and apoptosis.
Conflict of interest: Authors declare no conflict of interest.
Ethical approval: This study was approved by the Animal Care Committee and Ethics Committee of Çukurova University. All of the procedures were performed according to accepted standards of Guide for the Care and Use of Laboratory Animals.
Acknowledgments: This study was funded by Cukurova University Research Fund (TSA-2016-6136).