Introduction
The Punta Lara Nature Reserve (PLNR) (34°47’33” S & 58o0’28.32” W) is a protected area located on the banks of La Plata River, 12 km north of the city of La Plata (Province of Buenos Aires, Argentina). From the phytogeographical point of view the Reserve is located in the Chaco Domain, Oriental Pampean District of the Pampean Province of the Neotropical Region (Cabrera, 1976; Arana et al., 2017). It covers 6.000 hectares and extends over 8 kilometers of coastline where different environments and communities develop, among them the gallery forests, which constitute the world’s southernmost subtropical riparian forests (Cabrera & Dawson, 1944). Guerrero et al. (2018) postulated that the marginal forest of Punta Lara is a recently installed association, established in the mid-nineteenth century when the climate of the Río de la Plata region presented an increase in rainfall and minimum temperature, a trend that continues until the present. Currently, the Reserve is the core area of the Pereyra Iraola Biosphere Reserve within the framework of the UNESCO MAB Program (Barbetti, 2008). In this zone, the marginal forests develop on the border of incoming streams formed by La Plata River and comprise arboreal species, shrubs, epiphytes and an herbaceous stratum represented by various species of ferns, among other groups of plants.
The first floristic studies in the Reserve were carried out by Cabrera & Dawson (1944), which described 8 species of isosporated ferns. Later,Moschione (1987) cited 16 species of ferns. More recent investigations conducted by this research group reported the presence of 20 native species of isosporate ferns growing in different habitats of the Reserve (Giudice et al., 2011, 2014). Some exotic species such as Adiantum capillus-veneris L., Christella dentata (Forssk.) Brownsey & Jermy, Cyclosorus interruptus (Willd.) H. Ito and Pteris tremula R. Br., are also registered (Giudice et al., 2011; Arana et al., 2020).
Our studies in this area also included the reproductive aspects of some native ferns, as well as the analyzes of soil spore banks as contributions to biodiversity conservation in this area (Ramos Giacosa et al., 2014, 2017; Luna et al., 2016; Ramos Giacosa et al., 2017;Gorrer et al., 2018).
The spores provide useful systematic characters to distinguish genera, and in some cases species (Tryon & Lugardon, 1991). In particular, the characteristics of the spores under light microscope are a relevant source of information for paleobotanical and paleoecological studies, and also in the aerobiological and forensic fields (Harris, 1957; Vaughn et al., 1990; Farfán-Santillán et al., 2016).
Previous descriptions of the spores of some ferns that grow in Buenos Aires Province are found in Michelena (1989, 1993, 1998) andGiudice (1999). Also, some characteristics of the spores of certain taxa growing in the Reserve were provided during the reproductive studies in the genera Ctenitis (Ramos Giacosa et al., 2017), Microgramma C. Presl, and Pleopeltis Humb. & Bonpl. ex Willd. (Gorrer et al., 2018), Amauropelta Kunze, Christella H. Lév. and Goniopteris C. Presl (=Thelypteris Schmidel) (Giudice et al., 2014; Ramos Giacosa et al., 2014).
As a complement of the mentioned works, the objective of this study was to deepen the analyses of the morphological characteristics of the spores of the isosporated ferns inhabiting the Punta Lara Nature Reserve using light microscopy. This information will be useful for systematic, palynological and paleo-environmental studies and conservation in protected areas.
Material and methodsThe study was carried out with fresh materials collected during three years (2016-2018) at different sites within four cores of the Reserve (Fig. 1). The spores belong to the following 16 native taxa: Adiantum raddianum C. Presl, Amauropelta decurtata (Link) Salino & T. E. Almeida, Asplenium claussenii Hieron., A. sellowianum C. Presl ex Hieron., A. ulbrichtii Rosenst., Blechnum auriculatum Cav., Ctenitis submarginalis (Langsd. & Fisch.) Ching, Doryopteris concolor (Langsd. & Fisch.) Kuhn, D. pentágona Pic. Serm., Equisetum giganteum L., Gastoniella chaerophylla (Desv.) Li Bing Zhang & Liang Zhang, Goniopteris burkartii C. Chr. ex Abbiatti, Microgramma mortoniana de la Sota, Pleopeltis macrocarpa (Bory ex Willd.) Kaulf., P. minima (Bory) J. Prado & R.Y. Hirai, Rumohra adiantiformis (G. Forst.) Ching and 4 exotic species very frequent in the study area: Adiantum capillus-veneris L., Christella dentata (Forssk.) Brownsey & Jermy, Cyclosorus interruptus (Willd.) H. Ito and Pteris tremula R. Br. The voucher specimens are deposited in the Herbarium LP of the Museum of Natural Sciences of La Plata (Table 1).
Likewise, the microscopic preparations are preserved in the Laboratorio de Anatomía Comparada, Propagación y Conservación de Embriófitas “Dr. Elias R. de la Sota”, Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata. For the taxa nomenclature, the update of the Catalog of Vascular Plants of the Southern Cone was followed (Zuloaga et al., 2019). The spores were placed on to microscope slides with glycerin jelly without prior treatment and studied and photographed under a Nikon E200 light microscope (LM).
The analyzed characteristics were: color, shape in polar and equatorial view, laesura type and wall ornamentation. The quantitative data refers to the major and minor equatorial diameters, polar diameter and laesura length. The spore’s measures were randomly estimated on 20 spores in each sample. The averages of the 4 quantitative variables were obtained for each species and also for the respective genera and families. With these data, graphics were made to compare the quantitative variables between species, genera and families using Microsoft Excel 2013. The palynological terminology used in the descriptions refers to Tryon & Lugardon (1991). The figures were made using the Corel Draw X3 program.
Results
The morphological characteristics of the spores of each species are given in Table 2. The spore measurements are given in Table 3. The spores are illustrated in Figs. 2-6.

Fig. 1: Study area. The circle include the base station, “El burrito” trail, “Fern ditch”, “El Mirador” Stall, Yabotí Bridge, “Las Cañas” Stream and “La Medialuna” Stall. The triangle symbolizes “La Araucaria” Stall. The rhombus symbolizes “El Ché” trail. The quadrate symbolizes Baldovinos Channel.
The spores showed coloration from yellow, light brown to dark brown, and only in the case of Equisetum they were green (chlorophyllous). Most spores are monolete (thirteen), other trilete (six) and only one alete.
The largest spores belong to the family Polypodiaceae (Microgramma mortoniana 61.4 pm largest equatorial diameter) and the smallest ones to Pteridaceae (Doryopteris pentagona, 28.1 pm and Pteris tremula 30.5 pm).
According to the type of wall ornamentation, we find granular, folded, verrucous, tuberculated, psilated, reticulated and regulated-cristated spores.
Taking into account the morphological characteristics of the spores under LM, a dichotomous key is presented below.
Comparisons regarding the quantitative characteristics (measurements) of the spores are presented in Figs. 7-9.
Key to the identificaron of the ferns of the PLNR using the characteristics of the spores
1. Alete spores, with elaters..........................................................................................Equisetum giganteum
1'. Monolete or trilete spores, without elaters.
2. Monolete spores.
3. Psilate ornamentation....................................................................................Blechnum auriculatum
3'. Verrucate or folded ornamentation.
4. Verrucate ornamentation.
5. Evident verrucae.............................................................................Microgramma mortoniana
5'. Small verrucae, not clearly evident.....................................................................Pleopeltis sp.
4'. Folded ornamentation.
6. Short and subglobose folds.
7. Abundant folds, densely distributed.................................................Ctenitis submarginalis
7'. Scarce folds....................................................................................Rumohra adiantiformis
6'. Alate-cristate, reticulate, alate or echinulate-alate folds.
8. Alate-cristate folds........................................................................................Asplenium sp.
8'. Other types of folds.
9. Reticulate folds..........................................................................Amauropelta decurtata
9'. Equinulate-alate or alate folds.
10. Equinulate-alate folds...........................................................Cyclosorus interruptus
10'. Alate folds.
11. Folds partially fused................................................................Chrystella dentata
11'. Folds totally fused..............................................................Goniopteris burkartii
2'. Trilete spores.
12. With equatorial ridge.
13. Tuberculate ornamentation...................................................................................Pteris tremula
13'.Verrucate ridged ornamentation..........................................................Gastoniella chaerophylla
12'. Without equatorial ridge.
14. Granular ornamentation.........................................................................................Adiantum sp.
14'. Rugulate-cristate or psilate ornamentation.
15. Rugulate-cristate..................................................................................Doryopteris concolor
15'. Psilate................................................................................................Doryopteris pentagona

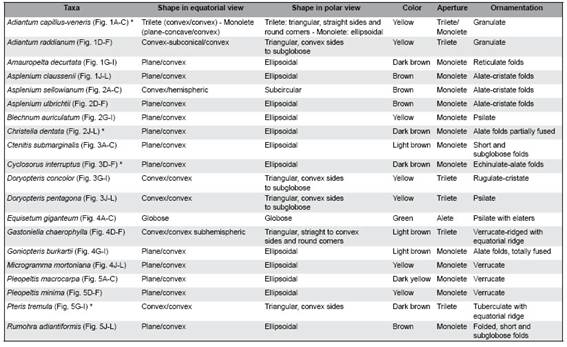
Fig. 2: Spores of Adiantum capillus-veneris, Adiantum raddianum, Amauropelta decurtata and Asplenium claussenii. A-C: Adiantum capillus-veneris: A: Trilete spore in proximal view, granúlate ornamentaron. B: Spore in distal view with straight sides. C: Spore in equatorial view. D-F: A. raddianum: D: Trilete spore in proximal view, granulate ornamentaron. E: Spore in distal view with convex sides. F: Spore in equatorial view with granulate ornamentaron. G-I: Amauropelta decurtata: G: Monolete spore in proximal view with reticulate folds. H: Spore in distal view with prominent wing-like folds at the edges. I: Spore in equatorial view with reticulate folds and wing like-folds at the edges. J-L: Asplenium claussenii: J: Monolete spore in proximal view, alate-cristate folds continuous at the edges. K: Spore in distal view with continuous alate-cristate folds along the edges. L: Spore in equatorial view with alate-cristate folds on the edges and on the body of the spore. Scales= A-L: 10 pm.

Fig. 3: Spores of Asplenium sellowianum, Asplenium ulbrichtii, Blechnum auriculatum and Christella dentata. A-C: Asplenium sellowianum: A: Monolete spore in proximal view, coarse alate-cristate folds are observed. B: Spore in distal view, coarse alate-cristate folds are observed both, on the edges and on the body of the spore. C: spore in equatorial view, coarse alate-cristate folds are observed. D-F: A. ulbrichtii: D: Monolete spore in proximal view, low alate-cristate folds with erose margin are observed. E: Spore in distal view, alate-cristate folds with erose margin are observed at the edges and on the body of the spore. F: Spore in equatorial view, alate-cristate folds with erose margin are observed. G-I: Blechnum auriculatum: G: monolete spore in proximal view. H: Spore in distal view with psilate ornamentaron. I: Spore in equatorial view. J-L: Christella dentata: J: Monolete spore in proximal view, continuous alate folds are observed at the edges. K: Spore in distal view, short alate folds, partially fused in the body of the spore. L: Spore in equatorial view, short alate folds and erose margin are observed. Scales= A-L: 10 pm.
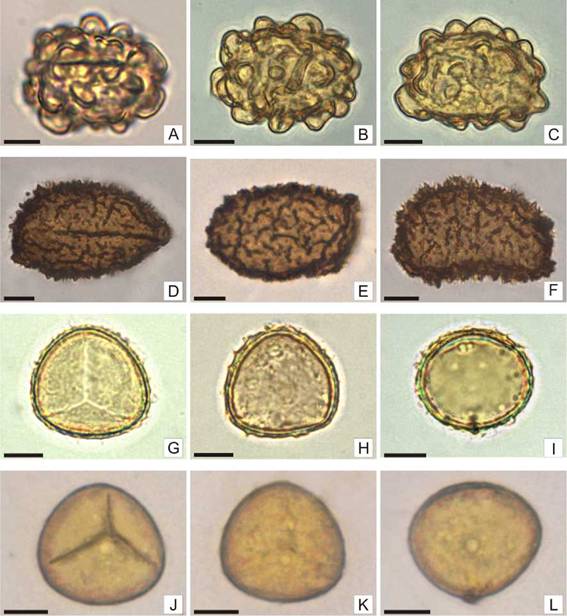
Fig. 4: Spores of Ctenitis submarginalis, Cyclosorus interruptus, Doryopteris concolor and Doryopteris pentágona. A-C: Ctenitis submarginalis: A: Monolete spore in proximal view, short and subglobose folds are observed. B: Spore in distal view, short and subglobose folds are observed over the entire surface. C: Spore in equatorial view, short and subglobose folds are observed. D-F: Cyclosorus interruptus: D: Monolete spore in proximal view, echinulate-alate folds are observed. E: Spore in distal view, short echinulate-alate folds are observed mainly in the body of the spore. F: Spore in equatorial view with short echinulate-alate folds. G-I: Doryopteris concolor: G: Trilete spore in proximal view, a surface with low rugulate-cristate are seen at the edges. H: Spore in distal view, low cristae or rugulae form the ornamentaron. I: Spore in equatorial view, low cristae or rugulae are observed at the edges. J-L: D. pentagona: J: Trilete spore in proximal view, convex sides. K: spore in distal view, psilated. L: Spore in equatorial view. Scales= A-L: 10 pm.
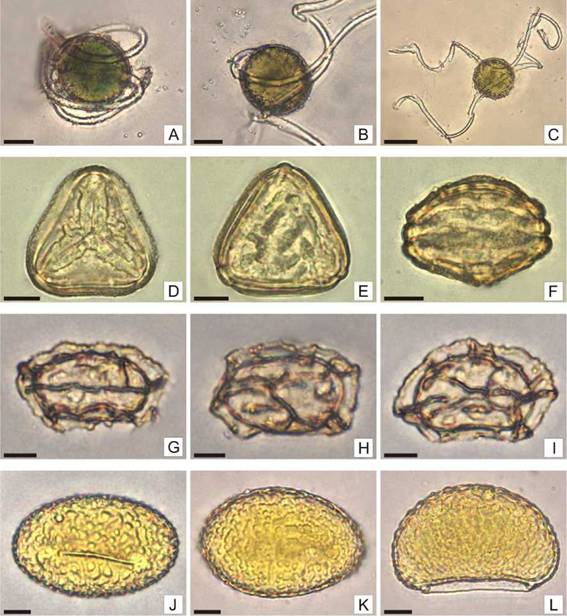
Fig. 5: Spores of Equisetum giganteum, Gastoniella chaerophylla, Goniopteris burkartii and Microgramma mortoniana. A-C: Equisetum giganteum: A: Alete spore, globose, chlorophyllous, with translucent elaters wound on the spore. B: Spore with elaters partially unrolled, psilated. C: Spore with elaters totally unrolled. D-F: Gastoniella chaerophylla. D: Trilete spore in proximal view, irregular ridges-verrucae surrounding the laesura. E: Spore in distal view, irregular ridges-verrucae are observed. F: Spore in equatorial view, an equatorial ridge is observed at the equator. G-I: Goniopteris burkartii: G: Monolete spore in proximal view, alate folds are observed at the edges. H: Spore in distal view, alate folds both on the edges and on the body of the spore are observed. I: Spore in equatorial view, alate folds totally fused. J-L: Microgramma mortoniana: J: Monolete spore in proximal view, the ornamentation is made up of verrucae. K: Spore in distal view, uniformly distributed verrucae are observed. L: Spore in equatorial view, verrucae tend to be small towards laesura. Scales= A-B: 20 pm; C: 40 pm; D-L: 10 pm.

Fig. 6: Spores of Pleopeltis macrocarpa, Pleopeltis mínima, Pteris trémula and Rumohra adiantiformis. A-C: Pleopeltis macrocarpa: A: Monolete spore in proximal view, very low verrucae are observed at the edges. B: Spore in distal view, small verrucae, not clearly evident are observed at the edges of the spore. C: spore in equatorial view. D-F: P minima: D: Monolete spore in proximal view, small verrucae not clearly evident are observed at the edges of the spore. E: Spore in distal view. F: Spore in equatorial view. G-I: Pteris tremula: G: Trilete spore in proximal view, tuberculated. H: Spore in distal view, smaller tubercles towards the equator are observed. I: Spore in equatorial view, an equatorial ridge is observed at the equator, the tubercles are seen larger towards the poles. J-L: Rumohra adiantiformis: J: Monolete spore in proximal view, continuous folds are observed at the edges. K: Spore in distal view, short and long folds are observed at the edges. L: Spore in equatorial view, few short, subglobose folds are observed. Scales= A-L: 10 pm.
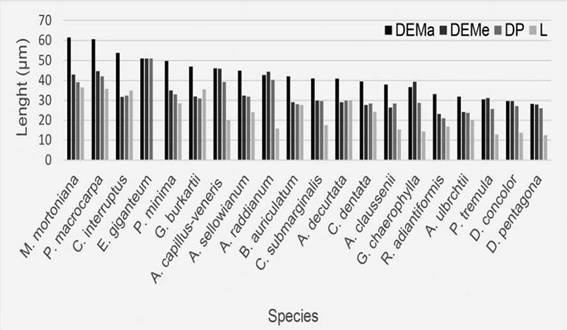
Fig. 7: Average spore size by species in the Punta Lara Natural Reserve. Abbreviations= DEMa: major equatorial diameter; DEMe: minor equatorial diameter; DP: polar diameter; L: laesura.
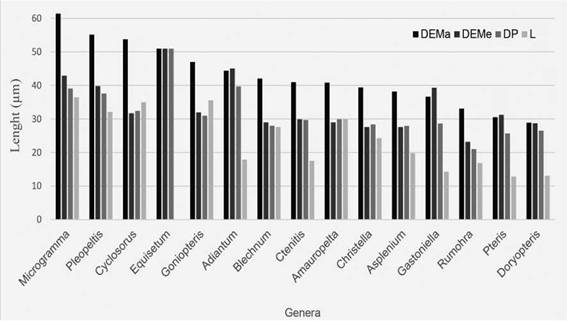
Fig. 8: Average size of the spores by genera in the Punta Lara Natural Reserve. Abbreviations= DEMa: major equatorial diameter; DEMe: minor equatorial diameter; DP: polar diameter; L: laesura.
Discussion and Conclusions
The morphological characteristics of the spores allowed the identification of most fern taxa at a specific level, except for the genera Adiantum, Asplenium and Pleopeltis. According to Simabukuro et al. (1998, 2000), the spores of related species within these genera have very similar morphological characteristics.
The largest spores belonged to the genera Microgramma and Pleopeltis, both epiphytic taxa. They also showed verrucate ornamentation. According to Tryon et al. (1990), epiphytic fern spores have walls with complex ornamentation.
In the exotic species A. capillus-veneris both, trilete and monolete spores, were found. According to Michelena (1989) and Ramos Giacosa (2014) observations in Pteridaceae and Anemia respectively, it is common to find different types of spores (trilete, monolete and intermediates) in the same specimen, and also at different stages of development (normal, aborted and immature). These variations were mentioned as characteristic of species where meiosis does not develop normally, thus achieving apogamy, hybridization and polyploidy (Wagner, 1974; Devi, 1977).
Detailed knowledge of spore morphology is an important tool for paleo-environmental reconstructions (Sánchez-Dzib et al., 2009; Poliakova & Behling, 2016). In this way, the knowledge of current palynological floras allows comparing with fossil ones and to establish relationships between them (Solé de Porta & Murillo-Pulido, 2005).
The spores of A. claussenii, C. dentata, C. interruptus, D. pentagona, G. burkartii and P. tremula are illustrated for the first time under the light microscope.
The information provided here constitutes a contribution to soil spore bank analyses, as well as fossil plant identification and aeropalinological studies, all of which employ spore characteristics for taxonomic identification.
Contributions of authorsDAG, GEG and JPRG analyzed the data. DAG and JPRG edited the figures and tables. JPRG and MLL revised the language. DAG, PCB and MLL collected the material. All authors participated in the writing of the manuscript.












 uBio
uBio