Introduction
Food production faces the challenge of keeping up high levels of quality, considering aspects of food safety and production systems with fair remuneration for producers (8). Grain storage arises as a consequence of the randomness and seasonality of agricultural production. Inside stored commodities, the temperature and humidity conditions favor the appearance of insect pests that find the food and protection to display their multiplication potential. It has been estimated that 5-30% of post-harvested losses worldwide are due to insect damage (25). The damages are qualitative and quantitative, including reduction of hectoliter weight, increased commercial rejection levels, alterations in the nutritional value, deterioration of industrial features and decreased seed germination power (13). Some species in the Order Coleoptera, due to their ubiquity and high destructive potential, constitute one of the greatest entomological problems of stored grains. The “weevil” (Sithophilus oryzae L.) and the “cereal borer” (Rhyzopertha dominica F.) are primary infestation beetles that initiate the deterioration of healthy grains, the larvae feed on the endosperm, leaving holes that facilitate the entry of secondary infestation species (13).
Despite the negative consequences associated with the utilization of synthetic insecticides, these substances are still the main solution utilized in preventive and curative treatments of stored grains. The use of chemical-synthetic insecticides involves a series of disadvantages: such as the presence of toxic residues in the grain, intoxication of users and consumers, contamination of the environment, and development of insect resistance (1,4). The urgent need for a control method that ensures the elimination of insect pests, leads in many cases to incorrect and excessive applications of these harmful products, risking food safety and therefore the health of the consumer, generating a negative environmental impact and the rejection of grains in the market (13).
The current demand for healthier food and the change in production paradigms require the total or partial replacement of synthetic pesticides by non-polluting methods. In this sense, a biological alternative, not detrimental to the environment that is also safe for the producers and the consumers is urgently needed to control stored grain pests. Current trends in integrated pest management are oriented towards preserving the environment together with the use of natural biopesticides with less toxicity. Among these products are the entomopathogenic fungi (14,20).
The species within the genera Beauveria and Metarhizium are widely used due to their specificity and effectiveness as biological insecticides (11,22,26,27). Several studies have demonstrated the capacity of entomopathogenic fungi to protect stored seeds and have demonstrated their insecticidal capacity to control different beetles (3,9,10,13,14) highlighting the importance of conducting bioassays for the selection of highly virulent isolates given the high genetic variability presented by these microorganisms.
The hypotheses tested were that native strains are able to control several species of stored grain pests and that the fungus is able to persist on the surface of wheat grains. Thus, this study was conducted to test the efficacy of a native strain of B. bassiana to control two main stored grain pests such as S. oryzae and R. dominica and also evaluate the persistence of the fungus on wheat grains in laboratory conditions.
Material and methods
Insect rearing
Two main stored grain pests R. dominica and S. oryzae, were selected to perform bioassays. The insects were acquired from the Department of Agricultural Zoology (Faculty of Agronomy, National University of Buenos Aires, Argentina) and a laboratory colony was established at the Spegazzini Institute, La Plata National University, Argentina. The insects were kept inside glass containers (400 mL) and provided with wheat grains (cultivar Klein Capricornio, Cauda Semillas, Chacabuco, Argentina) as food source. The colonies were maintained in a climatic chamber under controlled conditions of temperature and humidity (26 ±2°C and 70±5% RH).
All trials were carried out with adults of 7-10 days of age.
Fungal strain
The fungal strain used to carry out the laboratory tests was provided by the mycological collection of the Spegazzini Institute, La Plata National University, Argentina. The Beauveria bassiana LPSc 1227 strain (GeneBank Accession number MG012792) was isolated from Schistocerca cancellata (Orthoptera: Acrididae) in Santiago del Estero Province, Argentina during 2016. This strain was selected based on its entomocidal capacity (15). The inocula was obtained from cultures maintained in potato dextrose agar (PDA) for a week at 25°C in the dark. Conidia were harvested with a sterile loop, placed in test tubes and stirred for 2 minutes using a vortex. The conidial concentration was determined as in Goettel & Inglis (1997). The conidia were counted using a Neubauer chamber under a light microscope and the concentration was adjusted to 1×108 conidia/ml. The viability of the conidia was determined according to Goettel & Inglis (1997). The fungal suspensión (400 μl) was inoculated into slides containing a thin layer of PDA culture media. Slides were kept for 24 h in Petri dishes containing a moistened filter paper to allow conidia germination. Conidia were considered germinated when the germ tube exceeded half its length. Three repetitions were made at different times and 300 conidia were counted on each case. The conidia exhibited a 99% germination rate.
Mortality test
Rhyzopertha dominica and S. oryzae adults were inoculated with the fungal solution (1×108 conidia/ml) using a hand glass sprayer (20 mL). Control insects were sprayed with a conidia free solution of Tween 80 ® (Merck) 0.01% (v/v). Afterwards, adults were individualized in Petri dishes containing wheat grain as food source and placed inside a climatic chamber under controlled conditions (24±2°C, 75%). Mortality was recorded daily for 14 days. Humid chambers were set up according to Goettel & Inglis (1997) to confirm death by mycosis.
As data did not meet normality insect mortality was analyzed using the Wilcoxon test.
Survival curves and mean survival time (MST) were estimated using the Kaplan-Meyer analysis. Pairwise comparisons between survival curves were made by Logrank test (6,15). Infostat software was used to performe the statistical analyses (5).
Choice test
A dual-choice olfactometer with static air (supplementary material) was utilized to evaluate the insect preference according to Mitina et al. (2020) methods. One pot (20 cm3) containing 20 g of treated wheat grains was placed at one end of the tube and a container with untreated grains (control) was placed at the other end. Treated group grains were sprayed with the conidial suspension (1× 108conidia/ml). In the control group, grains were sprayed with a Tween 80 solution. Insects were placed individually at the center of the olfactometer, and the chosen direction was recorded. The olfactometer was rotated to ensure that the behavior of the insects depended only on the repellent action. Individuals who did not show a response after 10 minutes were not considered in the analysis. A total of 100 insects of each species were employed in this experiment.
To characterize the olfactory response of weevils, the “index of aggregation” (19,29) was calculated using the following formula:
IA= (O-K)/ (O+K) × 100%
where:
O = number of insects in the tube with the treated sample
K = number of insects in the control tube
If differences are obtained between the mean number of insects in the treatment and control, and the value of the index is positive indicates an attractive effect of the sample. On the other hand, a repellent action of the sample is recorded when the value of the index is a negative number. If no differences are observed a neutral action of the sample is inferred (19,29).
Seed persistence
To evaluate the conidial persistence in wheat over time, seeds (100 g) were autoclaved for 20 minutes, and then were sprayed with 30 ml of the fungal suspension (1×108conidia/ml). Inoculated seeds were placed in Erlenmeyer flasks (250 ml) and were maintained at 24°C in the darkness. Three seeds were randomly selected every week for 45 days and were placed in Petri dishes containing PDA. Control seeds were not mixed with the fungal inocula. The Petri dishes were kept under controlled conditions of temperature (25°C) and in darkness for seven days. The persistence was recorded when fungal colonies grew around seeds.
Results
Mortality tests showed significant differences between treatments for both insect species, R. dominica (W=135, p < 0.0001) and S. oryzae (W=55, p=0.0001). The B. bassiana strain controlled 89%±0.073 of R. dominica adults and 80% ±0.14 of S. oryzae (Figure 1).
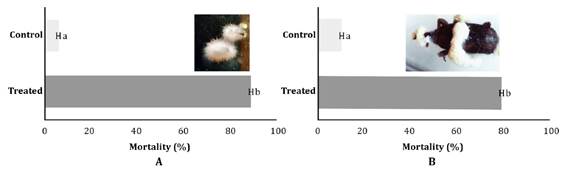
Different letters show significant differences according to Wilcoxon test (<0.05).
Letras diferentes indican diferencias significativas de acuerdo con el test de Wilcoxon (<0,05).
Figure 1 Percentage of adult mortality after the treatment with Beauveria bassiana LPSc 1227 strain. A: Rhyzopertha dominica and B: Sitophilus oryzae. Figura 1. Mortalidad porcentual de adultos de A: Rhyzopertha dominica y B: Sitophilus oryzae inoculados con la cepa de Beauveria bassiana LPSc 1227
The Kaplan-Meier analysis exhibited a mean survival time (MST) of 4.27 ±0.19 days for R. dominica adults and 4.27 ±0.20 days for S. oryzae. Results of long rank test (X2) for the comparison of the Kaplan-Meier curves did not present significant differences between the survival of both stored grain pests (log-rank test p>0.05) (Figure 2).
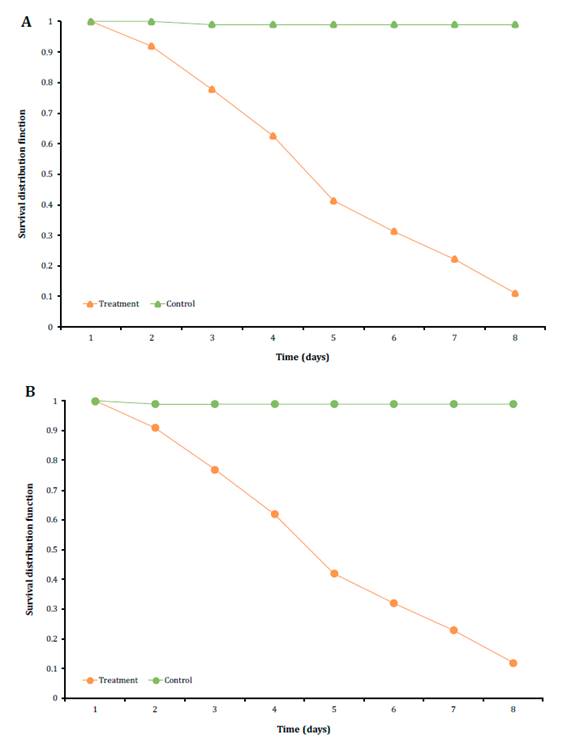
Figure 2 Kaplan-Meier survival curves of Rhyzopertha dominica A: and Sitophilus oryzae B: after the treatment with Beauveria bassiana LPSc 1227 strain. Figura 2. Curvas de supervivencia de Kaplan-Meier A: Rhyzopertha dominica y B: Sitophilus oryzae luego del tratamiento con la cepa de Beauveria bassiana LPSc 1227
Dual choice tests demonstrated that B. bassiana LPSc1227 presented a repellent action against both stored grain pests (Table 1).
Table 1 Effect of Beauveria bassiana on treated wheat grains on the preference of Sitophilus oryzae and Rhizopertha dominica. Tabla 1. Efecto de Beauveria bassiana sobre la preferencia por granos tratados de Sitophilus oryzae y Rhizopertha dominica para granos de trigo con y sin tratamiento de Beauveria bassiana.
After 45 days, 100% of wheat seeds studied preserved viable conidia on the surface (Figure 3).
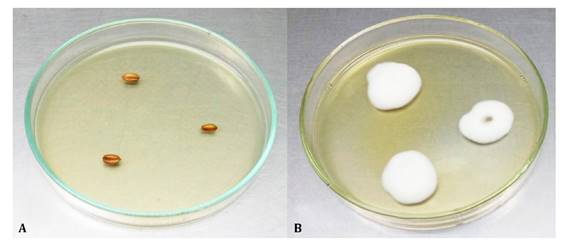
A: Control (not inoculated) and B: Treated with Beauveria bassiana LPSc 1227. A: Control (no inoculadas) y B: Tratadas con Beauveria bassiana LPSc 1227.
Figure 3 Persistence of Beauveria bassiana conidia 45 days after the wheat grain treatment. Figura 3: Persistencia de conidios de Beauveria bassiana en semillas de trigo luego de 45 días.
The results provided by this investigation constitute a starting point in the utilization of the B. bassiana strain LPSc 1227 as an effective entomopathogen to control two main stored grain pests.
In this study, B. bassiana LPSc 1227 exhibited mortality levels of 80-89% against R. dominica and S. oryzae respectively, showing a good performance to control both coleopteran species in vitro. Many studies presented similar results where the effectiveness of the entomopathogens relied on the fungal strain and the pest species tested. Wakil et al. (2021 b) found that R. dominica was the most susceptible species to B. bassiana and M. anisopliae. Also, Reza Pourian & Alizadeh (2021) reported that an isolate of B. bassiana was effective in killing 60-73% of Callosobruchus maculatus (F.) (Chrysomelidae) and Oryzaephilus surinamensis (L.) (Silvanidae). Kordali et al. (2021) registered high mortalities (from 62.6% to 100%) of C. maculatus adults using different species of entomopathogenic fungi. Furthermore, Yanar et al. (2019) also found that the S. granarius mortality varied depending on the fungal isolate utilized registering up to 70% mortalities. When analyzing fungal entomopathogens against R. dominica, Musso et al. (2020) reported that B. bassiana strains were the most effective in controlling adults causing up to 65% mortality.
Mean survival time (MST) constitutes an important parameter to describe when characterizing entomopathogens that gives an approximation of the pathogenicity rate of the fungus. In this study, median lethal times were similar for both insects (4.27±0.19 for R. dominica and 4.27±0.20 for S. oryzae) confirming the good performance of the fungus.
In the Same fashion, El Khourry et al. (2022) estimated for four different stored grain pests MST of 3.5±0.3 for Cathartus quadricollis (Guerin-Meneville) (Coleoptera: Silvanidae), of 3.8±0.6 days for Callobrosuchus maculatus F. (Coleoptera: Chrysomelidae), of 3.8±0.2 for Sitophilus granarius L. (Coleoptera: Curculionidae) and of 4.1±0.2 for Oryzaephilus surinamensis L. (Coleoptera: Silvanidae) using a B. bassiana strain. Similarly, Kassa et al. (2002) when studying several strains of B. bassiana to control Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae) and Protephonus truncates Horn (Coleoptera: Bostrichidae) registered MST that ranged between 2.85±0.05 to 6.28±0.41 days. On the contrary, higher MST have been found by other authors indicating a poor entomodicidal capacity of the strains employed, for instance Al-Zunti et al. (2023) when studying the effect of B. bassiana on larval stages of Tribolium castaneum Herbst (Coleoptera: Tenebrionidae) found MST of 5-6 days and in the case of R. dominica, Musso et al. (2020) obtained MST values for B. bassiana strains of 8 to 9 days.
The differential susceptibility of different species of stored grain insect pests towards fungal entomopathogens has been attributed to differences in the composition of the insect cuticle, to the conidial concentration or to fungal specificity (3,4,27). The entomocidal capacity of the B. bassiana LPSc 1227 strain towards two of the main primary stored grain pests represents an important finding in the search for the formulation of an effective bioinsecticide.
Fungal-insect interactions are of great interest to understand the fundamental behavioral processes that occur between insects and pathogens. This issue remains crucial when trying to exploit fungal entomopathogens as biological control agents. Results provided by this study show that the B. bassiana strain inflicted a repellent effect on both insect species. Many studies revealed that insects tend to avoid the presence of fungal entomopathogens (16,17,23). This behavior has been attributed to volatile compounds released by fungi and to the capacity of insects to detect these specific signals. In this regard, Selitskaya et al. (2016) confirmed that B. bassiana strain Yuk-4 had a strong repellent effect towards the granary weevil. Similar results were also obtained by Mitina et al. (2020) using B. bassiana. The authors obtained negative values of the index of aggregation, showing repellence towards the fungus. Also, Selitskaya et al. (2014) investigated the behavior response of S. oryzae to several Fusarium strains and found differential responses of the insects according to the strains.
Seed persistence of conidia on wheat grains over prolonged periods may provide extra protection against stored primary pests since it may contribute to suppressing progeny production (27). Results in this study demonstrated that the strain B. bassiana LPSc 1227 remains viable on the seed surface for at least 45 days.
When developing a microorganism-based product the study of multitrophic interactions should be considered and included as part of the basic research of an entomopathogen, future research will contribute to elucidating further properties of the promising strain studied.
Conclusion
This study demonstrates that the native strain of B. bassiana LPSc 1227 can effectively control stored grain pests and also persist on the surface of wheat grains.
Further research will contribute to elucidating additional insecticidal features of the B. bassiana LPSc 1227 strain against S. oryzae and R. dominica, two primary pests of stored grain. Additionally, it aims to evaluate whether the conidia present on the seed surface after 45 days retain their germination and insecticidal capacity.














