I. Introduction
Polymers such as plastics and rubbers pervade our lives, and we come across them in many diferent forms. Their physical properties are therefore of great importance, and an understanding of them is vital for their use in technology and engineering [1]. The blending of diferent polymers or inorganic materials with polymers represents a strategic route to improving the performance of a material, and allows the realization of novel composite systems that enhance the performance of the parent blend [2]. Polyvinyl alcohol (PVA) is a versatile, polyhydroxy polymeric material which has gained the interest of researchers due to its many potential applications, and the scope for easy modification and formation of useful miscible blends with many other polymers. PVA reinforced with different materials like iodine, ferric chloride, barium chloride and other salts have been studied extensively, and these polymeric materials show a signicant modication in their microstructural, electrical and mechanical properties when compared to pure PVA ms [3]. The subject of polymer blends has been one of the primary areas of focus in polymer science and technology over several decades now. As a new area of interest in polymer science, polymer blend technology often represents an important subject [4]. Polymer blends ofer versatile industrial applications through property enhancement and economic benets. The blending of two or more polymers of similar or dissimilar natures has been practiced for many years [5]. Solution blending of different polymers is one of the methods used to obtain new material with a variety of properties, which mainly depend on the characteristics of the parent homo polymers and the blend composition [6]. Polyvinyl alcohol (PVA), a semi-crystalline poly- mer, has been studied widely because of its many interesting physical properties, which arise from the presence of OH groups and the hydrogen bond formation with other polymers or metals. Polyvinyl pyrrolidone (PVP) is a vinyl polymer possessing planar and highly polar side groups due to the peptide bond [7]. The aim of this study is to prepare pure polymer blend (PVA:PVP) lm and salt (CdCl2 H2O) reinforced polymer blend films at different weight ratios (10 wt%, 20 wt%, 40 wt%) using the casting method, and to investigate the efect of salt reinforcement on the dielectric and mechanical properties of the prepared films.
II. Experimental work
In the preparation of polymer blend lms, polyvinyl alcohol powder (produced by Central House (P) Ltd of India with a molecular weight of 13000 g/mol - 23000 g/mol), and Polyvinyl Pyrrolidone powder (produced by the Indian HIMEDIA Company, with a molecular weight of 40000 g/mol) were used. For reinforcement of the blend, cadmium chloride (CdCl2H2O) salt (produced by the Indian HIMEDIA Company) was used.

Figure 2: Log-Log plot of the dielectric constant as a function of the frequency of pure and PVA:PVP polymer blend films reinforced with different weight ratios of CdCl2H2O salt.
Pure PVA:PVP polymer blend film and CdCl2H2O salt-reinforced polymer blend films were prepared at different weight ratios (10 wt%, 20 wt%, 40 wt%) using the casting method. The Differential Scanning Colorimeter (DSC) was performed for all samples and reported elsewhere, and the evidence for the blend nature of the film was con rmed [8]. The PVA and PVP powders with 1 : 1 wt%, and CdCl2H2O powder with the above mentioned weight ratios were dissolved in distilled water by stirring for 1 hr at 60. The solution was then poured into special glass molds placed on a at surface and left until the solvent evaporated to obtain the pure polymer blend film and salt-reinforced polymer blend films. The thickness was measured using a digital micrometer, and was found to be in the range 1450 m - 1455 m. For the purpose of dielectric measurements, an LCR meter (4294A Agilent Precision Impedance Analyzer) was used in the frequency range of 100 Hz - 1 MHz at room temperature, and for the investigation of mechanical properties, the following instruments were used: Tinius Olsen-H10K for the tensile test, Shore D Checkline-dd-100 for the hardness test and Filling Darter Impact Tester of the type FDI-01 for the shock resistance test. Figure 1 shows the sample images required for each test.

Figure 3: Semi-log plot of the loss factor as a function of the frequency of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.
III. Results and discussion
i. Electrical properties
a. Dielectric constant
The dielectric constant (0) was recorded for pure polymer blend PVA:PVP film and CdCl2-H2O salt reinforced polymer blend films at diferent weight ratios (10 wt%, 20 wt%, 40 wt%) at room temperature and within the frequency range of 100 Hz - 1 MHz, as shown in Fig. 2. The dielectric constant decreased with increased frequency for all polymer blend films, which can be explained as follows: In the low frequency region there will be sufficient time for molecular dipoles to rearrange and align themselves in the direction of the external electric eld, but at high frequencies the time is shorter, and less than the time period needed by the molecules for rearrangement in the direction of the external electric eld [9]. The dielectric constant at the same frequency increased with an increase in the weight ratio of added salt. In general, this increase in the value of the dielectric constant is due to increased polarization [10].
b. Loss factor
The loss factor (0) is the ratio of loss of power in electrically insulating materials to the total capacity transported through the insulator; i.e., the loss of energy in the insulating material is directly proportional to the loss factor. The dielectric loss factor of the pure PVA:PVP polymer blend lms and polymer blend films reinforced by CdCl2-H2O salt was calculated at diferent weight ratios (10 wt%, 20 wt%, 40 wt%) at room temperature and within the frequency range of 100 Hz { 1 MHz, as shown in Fig. 3. It can be observed that the loss factor decreases as the frequency increases for all the polymer blend lms. This may be attributed to the enhancement of the charge carriers that takes place across the electric charge area, decreasing the value of the loss factor at high frequencies until the electron's energy is equal to the Fermi level [11]. Another reason for the change in the loss factor with frequency is the polarization mechanism and the multiple interactions between ions and dipoles. This is due to the value of relaxation time [12]. It is also observed that the value of the loss factor at the same frequency increases with an increase in the weight ratio of the added salt. In general, this increase in the value of the dielectric loss factor is attributed to the increase in polarization and the increase in ion charge carriers [13, 14]
c. AC electrical conductivity
The alternating electrical conductivity of the pure PVA:PVP polymer blend films and polymer blend films reinforced by CdCl2-H2O salt was measured at diferent weight ratios (10 wt%, 20 wt%, 40 wt%) at room temperature and within the frequency range of 100 Hz - 1 MHz, as shown in Fig. 4. It is clear that the alternating electrical conductivity increases significantly as the frequency increases for all the polymer blend films, and this increase is due to increased polarization in the samples [15]. It should be noted that the alternating electrical conductivity in a dielectric material is the amount of power lost when an alternating electric eld is exerted, which appears as heat when the dipoles rotate in their positions. The vibration of the charges changes with the alternating electric eld, and therefore depends on the frequency [16]. Moreover, the alternating electrical conductivity at the same frequency increases with an increase in the weight ratio of added salt. This increase is strongly afected by many factors, including the purity of the material and dispersion. In general, this increase in alternating electrical conductivity is attributed to a decrease in dielectric resistance due to the increase of conductive molecules in the polymeric blend films [17], and also because of the number of charge carriers that have a signifficant relaxation time due to the high energy barrier [18].

Figure 5: Stress-strain curves of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.

Figure 6: Tensile strength of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.
ii. Mechanical properties
a. Tensile test
The tensile test was conducted and stress-strain curves were obtained for pure PVA:PVP polymer blend films and polymer blend films reinforced by CdCl2H2O salt at diferent weight ratios (10 wt%, 20 wt%, 40 wt%) . These curves are shown in Fig. 5. The stress-strain of the pure polymer blend lms consists of the elastic deformation region showing a linear relationship between stress and strain. From this region Young's modulus can be estimated from the slope of the straight line. The polymeric material within the boundaries of this region sufers from an elastic deformation due to the stretching and elongation of the polymeric chains without breaking the bonds. This curve deviates from linear behavior due to cracks generated within the polymeric material. These cracks grow and combine with increased stress, creating larger incisions and continuing to grow with stress until a fracture occurs in the sample [19]. In other cases, the fracture begins at the outer surfaces in the sites of deformities or defects such as scratches, holes or internal cracks, which act as areas for concentration of stress. This leads to a rise in the stress value to limits where the strength value exceeds the internal force of cohesion, and thus breakage occurs. When the CdCl2-H2O salt is added to the pure polymer blend, the stress-strain curve changes and we obtain curves with diferent properties. Figures 6, 7 and 8 show the variations in tensile strength, elongation at break and Young's modulus as a function of the weight ratio of the added salt, for all samples. Table 1 shows the values of tensile strength, elongation at break and Young's modulus for all polymer films, determined from the stress-strain curves. The tensile strength of the pure polymer blend films is 15.5 MPa, the elongation at break value is 28:7 % and the value of Young's modulus is 332 MPa; however, when the blend is reinforced with CdCl2-H2O salt, these values change. The tensile strength values increase at the 10 wt% weight ratio , reaching 56.1 MPa, and then decrease with an increase in the weight ratio of the salt added. The elongation at break value starts to decrease with an increase in the weight ratio of salt added, while the value of Young's modulus starts to increase unsystematically to reach its highest value of 7200 MPa at the weight ratio of 40 wt%. The decrease observed in the values of tensile strength, elongation at break and Young's modulus for polymer blend films at some weight ratios of salt reinforcement compared with pure polymer film is due to weak interaction between the molecules and low interstitial adhesion between the composite components, which leads to an increase in the composite fragility [20].

Figure 7: Elongation at break of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.

Figure 8: Young's modulus of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.
The increase found in the values of tensile strength, elongation at break and Young's modulus for the polymer blend films at some weight ratios of salt reinforcement compared with pure polymer film indicates that reinforcement has been achieved. It can be concluded that, at these weight ratios, the added salt is compatible with common addition polymerization and is efectively dispersed in the polymer blend , afecting its mechanical properties [21].
b. Hardness test
Hardness (shore D) for pure polymer blend films and those reinforced by CdCl2-H2O salt are shown in Fig. 9. It is clear from the gure that the hardness of the pure polymer blend films increases with an increase in the weight ratio of salt added, reaching its highest value (32.5) at the 20 wt% weight ratio, and then the hardness decreases with an increase in the weight ratio of salt added. This decrease is due to the high viscosity gained by the prepared material when adding high weight ratios of reinforcing salt to the matrix (polymer blend), which is in the liquid state. The high viscosity makes penetration of the CdCl2-H2O salt inside the interfaces of the polymer blend inefficient and difficult, which leads to the production of many gaps within the prepared composite material when hardened, causing a decrease in the hardness [22]. Table 2 shows the hardness values of all polymer blend films.
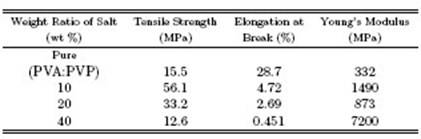
Table 1: Tensile property values of pure and PVA:PVP polymer blend films reinforced with diferent weight ratios of CdCl2-H2O salt.

Figure 9: Hardness of pure and PVA:PVP polymer blend films reinforced with different weight ratios of CdCl2-H2O salt. Figure 10: Fracture energy of pure and PVA:PVP polymer blend films reinforced with different weight ratios of CdCl2-H2O salt.
c. Impact test
The impact test is an important mechanical test used to demonstrate the resistance of a material to collapse by the force of impact under operating conditions, as it measures the actual energy required to break a piece under the test [23]. Fracture Energy for pure polymer blend lms and those reinforced by CdCl2-H2O salt were recorded at different weight ratios, as shown in Fig. 10. It can be seen that the fracture energy value for the pure polymer blend films is 0:392 kg.m2/s, and that this value increases as the weight ratio of added salt increases. In other words, the figure shows that the absorbed energy necessary for fracture increases with an increase in the weight fracture [24], where the CdCl2-H2O salt works to hinder the growth of the crack, and this changes the shape of the crack and its direction. This change in the shape of the crack increases the surface area of the fracture and the spent energy, all of which lead to an increase in the mechanical resistance of the material [25]. The addition of CdCl2-H2O salt to the PVA:PVP pure polymer blend film improved the mechanical properties. The reason for the increase in fracture energy with the increase in the weight ratio of added salt is that a large part of the impact energy projected onto the sample is reduced by the salt, which increases the resistance of the substance [25]. Table 3 shows the fractional energy values of all polymeric films.

Table 2: Hardness values of pure and PVA:PVP polymer blend films reinforced with different weight ratios of CdCl2-H2O salt.

Table 3: Fracture energy values of pure and PVA:PVP polymer blend films reinforced with different weight ratios of CdCl2-H2O salt.
IV. Conclusions
The results for polymer blend films reinforced with CdCl2-H2O salt show that the values of the dielectric constant, loss factor, and alternating electrical conductivity increase with an increase in the weight ratio of CdCl2-H2O salt at the same frequency. In general, this increase is attributed to an increase in polarization. This is why all these polymer blends can be used in the manufacture of electric batteries. The results of the tension test for PVA:PVP polymer blend lms reinforced with CdCl2-H2O salt show a change in the values of tensile strength, elongation at break and Young's modulus as the salt weight ratio increases. It can be concluded that, at these weight ratios, the added salt is compatible with common addition polymerization and is efectively dispersed in the polymer blend, afecting its mechanical properties. The hardness valu rst increases then decreases as the salt weight ratio increases. This decrease is due to the high viscosity gained by the prepared material when adding high weight ratios of reinforcing salt to the matrix (polymer blend), which is in the liquid state. The fracture energy value increases with increasing salt weight ratio, where the salt works to hinder the groh of the crack and this changes the shape of the crack and its direction. This change in the shape of the crack increases the surface area of the fracture and the spent energy, all of which lead to an increase in the mechanical resistance of the material, thus they could be good candidates for hard adhesives with low exibility.
















