INTRODUCCIÓN
Continental Chile is located in the south-western region of South America (between 17º30’-56º30’S and 66°-75°W), but with additional insular territories along its coast such as islands and archipelagos distributed in the Pacific Ocean, among them Isla de Pascua, Archipiélago de Juan Fernández, Isla Mocha, and Isla Grande de Chiloé are the most representative. Geographically, continental Chile spans the dry desert to the north, the Andes mountain range to the east, the Pacific Ocean to the west, and Antarctica to the south (Moreira Muñoz, 2011). Along its latitudinal configuration Chile displays a mosaic of environments extending along the length of more than 4,329 km, with marked climatic north-to-south gradients. In addition, the longitudinal gradient of Chile is marked by differences in altitude (up to 6,000 m.a.s.l.) which give rise to microclimate variations and thus to environments that are favorable for plant growth. In fact, 55% of its territory is covered by vegetation of which 45% corresponds to floristic endemism. In addition, insular Chile is an interesting mosaic covering a surface area of 374 km2, with an endemism that varies between 64% and 87%, depending on the island (Villagrán and Hinojosa, 2005; CONAF, 2013; Urbina Casanova et al., 2015; Jara Seguel and Urrutia, 2018).
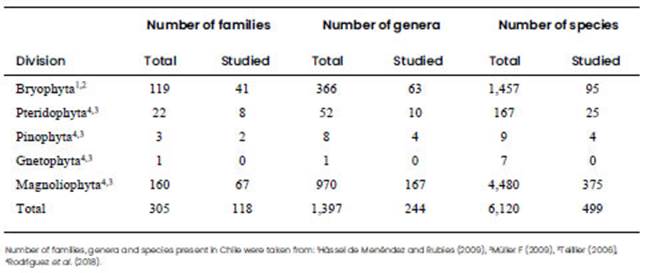
The total richness of land plants for continental and insular Chile has been estimated at ca. 6,120 species belonging to Bryophyta (1,457 species), Pteridophyta (167 species), Pinophyta (nine species), Gnetophyta (seven species) and Magnoliophyta (4,480 species) (Teillier, 2006; Hässel de Menéndez and Rubies, 2009; Müller, 2009; Stuessy and Baeza, 2017; Rodriguez et al., 2018), thus representing ca. 1.9% of the worldwide flora which has been estimated at 330,000 species (Christenhusz and Byng, 2016; Villagrán, 2020).
Methods and techniques for the analysis of plant genome diversity have emerged over the decades, particularly the estimation of nuclear DNA content or C-value, a feature that has also been related to chromosome number and ploidy level (Bennet and Leitch, 2005a; 2005b; Leitch and Leitch, 2012; Pellicer et al., 2018). Today, cytogenetics is recognized as a powerful tool to understand genetic variation processes, genome structure and dynamics, as well as evolution and speciation (Leitch and Leitch, 2012). Cytogenetics has also supported taxonomic circumscriptions in various plant groups (Widham and Yatskievych, 2003; Jara Seguel et al., 2010; Jara Seguel and Urrutia, 2012; Guerra, 2012) and in several cases has related extant species with extinct species found in fossil records (Bonde et al., 2004).
The first review reporting the state-of-the-art on cytogenetics of Chilean flora was carried out in angiosperms and it gathered information for approximately 139 continental and insular species (2.8% of total angiosperms) across 78 publications from years 1924 to 2010 (Jara Seguel and Urrutia, 2012). Recently, the flora of some protected wild areas in the southern zone has also been studied based on the diversity of chromosome numbers (Jara Seguel et al., 2020). In addition, the flora of insular Chile has also been a focus of interest from a cytogenetic point of view, with a number of reviews reporting valuable information on chromosome number. The mechanisms behind cytological evolution have also given rise to various hypotheses in regard to the islands (e.g., Juan Fernández Archipelago, Stuessy and Baeza, 2017). However, a large part of continental and insular Chilean flora has not been studied, while cytoevolutionary processes based on genome structure and dynamics are still poorly understood (Jara Seguel and Palma Rojas, 2021).
In this review we document the advances in cytogenetic studies of Chilean land plants, focusing our analysis on the number of publications on this subject, their taxonomic representation and geographical range, as well as the cytogenetic markers that were analyzed and their resolution to determine the genomic characters of the species. In this context, the diversity of Chilean land plants and its contribution to worldwide diversity is reported in this review.
Number of publications
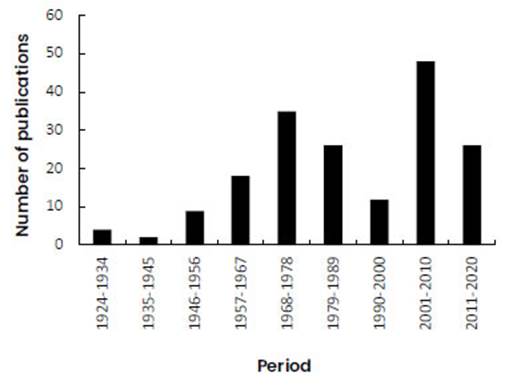
One hundred eighty articles on cytogenetics of Chilean land plants have been published from 1924 to 2020 (Figure 1). The literature on cytogenetics has increased significantly in the last two decades, with 74 articles published between 2001 and 2020. This reflects not only the growing interest of Chilean researchers in the study of cytogenetic characters of the native flora, but also that of foreign researchers in studying chromosome variation in intercontinental and insular floras (Sanders et al., 1983; Spooner et al., 1987; Sun et al., 1990; Lammers and Hensold, 1992; Rahman et al., 2001; Hanson et al., 2003; Kiehn et al., 2005; Talluri and Murray, 2009; Zonneveld, 2012; Hizume and Kan, 2015; Souza et al., 2015; Lujea and Chiarini, 2017; Sassone et al., 2018). Notwithstanding, growth in cytogenetic contributions depends in part on the interest of specialized botanical journals in publishing the data. Thus, only 43 articles have been published in five Chilean journals since 1954, 19 of which correspond to the last decade (2011 to 2020), whereas most of the reports on the cytogenetics of Chilean land plants since 1924 have been published in foreign journals, in many cases authored by foreign cytogeneticists.
Taxonomic representation and geographical range
In our revised literature, listed in the version 4.0 of Chilean Plants Cytogenetic Database (Jara Seguel and Urrutia Estrada, 2021), we found cytogenetic data for 499 Chilean species belonging to Bryophyta, Pteridophyta, Pinophyta and Magnoliophyta, which represent 244 genera and 117 families accounting for ca. 8.2% of total Chilean land plant species (Table 1). However, the number of studied species is likely to be higher than that reported here, particularly with regard to chromosome number data, due to the difficulty in compiling information from old sources of publication. A particular case is Bryophyta, for which little knowledge exists in Chile due to the scarcity of taxonomy specialists for this plant group. A similar situation has been observed in Pteridophytes although there is evidence of incipient advances in knowledge regarding chromosome number and polyploidy (Jara Seguel et al., 2006; Morero et al., 2015). With this in mind, reviews such as “Flora de Chile” (Rodríguez and Quezada, 1995; Rodriguez et al., 2018), “Libro Rojo de la Flora Nativa” (Squeo et al., 2001; 2008), “Plant Geography of Chile” (Moreira Muñoz, 2011), and contributions to the identification and biogeography of Chilean bryophytes (Müller, 2009; Larraín, 2007; 2010a; 2010b; Villagrán, 2020), may play a fundamental role in updating the taxonomic knowledge, in addition to increasing data on the geographic distribution, biogeography, endemism and conservation status of Chilean land plants.
The largest number of continental Chilean species that have been cytogenetically studied is present in a long latitudinal gradient from 17º30’ to 56ºS, with many of the taxa located within the continental biodiversity hotspots (Arroyo et al., 2006). In addition, cytogenetic data of native flora from Juan Fernández Archipelago are also available and are included within insular hotspots (Stuessy and Baeza, 2017). There is data for only one species from Easter Island (Baeza, 1996) and for several Chilean species shared with the Falkland Islands in the southern tip of Patagonia (Moore, 1967). Currently, two frost-resistant angiosperm species have been described for Chilean Antarctica (at 62°S), Colobanthus quitensis (Kunth) Bartl. and Deschampsia antarctica E. Desv. (Molina Montenegro et al., 2012), with karyotype and cytomolecular data reported for populations of D. antarctica included in the Argentinian flora (Cardone et al., 2008; Amosova et al., 2015).
An important number of taxa are undersampled in certain geographical extensions (ca. 91.8% of the total species), such as those from southern Chile along South Patagonia (>53º S) including continental and insular lands, as well as those from high altitudes in both the Nahuelbuta coastal mountains and the Andean range. Efforts should be focused on the cytogenetic study of these plant groups, paying special attention to either local endemic taxa or to highly specialized species adapted to their environment (e.g., parasites, xerophytes, hydrophytes, halophytes, carnivorous). In the case of hydrophytes and marsh plants, a number of advances exist with regard to chromosome numbers described for species of nine genera, among them, Pinguicula (2n=16, 26; Lentibulariaceae), and Potamogeton (2n=ca. 78, Potamogetonaceae) (Casper and Stimper, 2009; Chepinoga et al., 2012). To date, ca. 455 species of hydrophytes and marsh plants have been described for the Chilean flora (Hauenstein, 2006).
Major cytogenetic characteristics that were studied here for Chilean land plants are summarized in Table 2. Chilean plants contribute to the diversity of the worldwide flora with an endemism that reaches between 45% on the continent and 64-87% in the islands. Along continental territories many species or genera are ancient, a large part of them proceeding from Gondwanan forests with a long evolutionary history, such as the austral Antarctic floristic element (ca. 20-55 Mya), or others proceeding from Neogene subtropical paleoflora (ca. 15 Mya). In the northern zone, modern floral elements are more recent in origin (ca. 4-15 Mya) (Scherson et al., 2017). Various genera and families are currently shared with other geographical regions such as part of North America, Central America, and Australasia. In this way, Chilean flora acquires relevant value as a natural heritage that must be recognized and protected by the worldwide community in its different biological levels. Proof of their value is the designation of extensive Chilean areas as hotspots for worldwide biodiversity (Arroyo et al., 2006).
Chromosome number and polyploidy
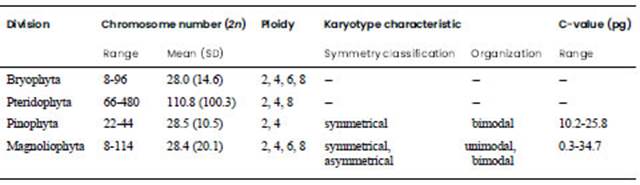
As a genome feature, the chromosome number includes the so-called A-chromosomes and B-chromosomes or supernumerary elements. The A-chromosome number is the most studied cytogenetic feature within Chilean land plants, while B-chromosomes have been found in three Chilean angiosperm species. In this work all 499 gathered land plant species have data on chromosome number, in many cases including subspecies, varieties or natural hybrids.
Chilean angiosperm species cytologically studied to date amount to 375 including 67 families and 167 genera, which represent 6.1% of total land plants and 8.4% of total angiosperms. The mean somatic chromosome number (2n) estimated here for Chilean angiosperms was 28.4+20.1 (n~14.2) ranging from 8 in some Hypochaeris species (Asteraceae), which are diploids, to 114 in the possible polyploid Libertia chilensis (Molina) Gunckel (Iridaceae) (Jara Seguel and Urrutia Estrada, 2021). This mean chromosome number is lower than the 32 previously documented including only 139 species (Jara-Seguel and Urrutia, 2012), therefore new data have made it possible to update the previously reported statistical records. In addition, this mean 2n number was similar to that described for plants from the Cerrado phytogeographic domain in Brazil (ca. 2n = 28, n~14; Roa and Telles, 2017) with 699 studied species. Both n numbers were lower than the gametic number (n=16) estimated for worldwide angiosperms, with a range that varied from 4 to ca. 640 (Soltis and Soltis, 2000; Leitch et al., 2010). Several Chilean families show relatively high levels of cytogenetic variation with diverse basic chromosome numbers. In this context, cytoevolutionary mechanisms that modify the chromosome number have been described, such as, end-to-end fusions, Robertsonian traslocations, and polyploidization in families such as Alstroemeriaceae, Amaryllidaceae and Asteraceae, all studied on the basis of chromosome counts and karyotype morphology (Buitendijk and Ramanna, 1996; Weiss Schneeweiss et al., 2003; Baeza and Schrader, 2005a; Jara Seguel et al., 2006; 2010; Baeza et al., 2007a; Palma Rojas et al., 2007; Chacón et al., 2012a).
B-chromosomes have been reported for three monocot species: Alstroemeria angustifolia Herb. subsp. angustifolia, Alstroemeria hookeri subsp. recumbens (Herb.) Ehr.Bayer (Alstroemeriaceae) both located between 32°S and 33°S in Central Chile (Buitendijk and Ramanna, 1996; Sanso, 2002), and Lapageria rosea Ruiz & Pav. (Philesiaceae) (Hanson et al., 2003; Jara-Seguel and Zúñiga, 2004). L. rosea inhabits two distinct zones, at 30°S in northern Chile (Fray Jorge National Park) and from 33°S to 40°S in the forests of southern Chile. B-chromosomes are supernumerary elements (additional to the standard complement, or A-genome) that are present in ca. 1,300 plant species worldwide, and are mostly distributed in monocots or in plants with large genomes but low chromosome numbers (Camacho et al., 2000; Houben, 2017). It is likely that B-chromosomes may have an adaptive role on a geographic scale (Peruzzi et al., 2011) but they do not appear to have had a relevant role in the adaptation and evolution of Chilean angiosperms due to their low presence in the species studied to date (less than 1% of the species).
The fossil record suggests that angiosperms appeared ca. 145-125 Mya, with an increment in diversity between 112-96.6 Mya (De Bodt et al., 2005; Leitch and Leitch, 2012). The origin of polyploidy in angiosperms has also been widely debated. The ancestral number may be x=7 and x=8, and various numbers higher than x=9 (e.g., x=10, 12, 14, 19, 21) are suggested to be of ancient polyploid origin (Stebbins, 1971; Goldblatt, 1980; Soltis and Soltis, 2000; Soltis et al., 2003; De Bodt et al., 2005; Leitch et al., 2010; Leitch and Leitch, 2012; Carta et al., 2020). However, no evidence exists of a clear link between ancestral chromosome numbers and ancient polyploidization events, suggesting that further insights are needed to elucidate the organization of genome packaging into chromosomes (Carta et al., 2020). In fact, several estimations suggest a wide variation in the percentage of polyploid angiosperms, ranging from 30% to 80% of the species studied worldwide, and molecular evidence points to ancient genome duplication at the base of monocots and dicots (Soltis et al., 2003; Bennett, 2004; Leitch et al., 2010). Bennett (2004) had already questioned this wide range, remarking that a difference of 40-50% represents over 100,000 species, which he considers an unacceptably high error rate for this key element of plant evolution. Nevertheless, these percentages may be still higher because a representative number of plant species have not been studied in various regions around the world according to current data, among them, flora from Chile (91.8%), New Zealand (20%), Italy (65%), Slovenia (40%), Poland (60%) and other countries (Peruzzi et al., 2011; 2012).
Within the Chilean angiosperms, ca. 70 continental species were found to be polyploid (ca. 21.5% of the continental studied species), whereas for the endemic taxa of Juan Fernández archipelago the level of polyploidization is estimated to be ca. 66% (Sanders et al., 1983). These data suggest that polyploidy has been a relevant evolutionary mechanism in Chilean oceanic islands, possibly providing greater adaptive capacity to survive in changing environments (Sanders et al., 1983; Stuessy and Baeza, 2017). As an example of polyploidy within continental and insular Chilean angiosperm taxa, mention can be made of the tetraploidy present within the families Lamiaceae (2n=44, x=11), Onagraceae (2n=44, x=11), Asteraceae (2n=44, 80; x=11, 20; 2n=ca. 94), Amaryllidaceae (2n=32, x=8), and the hexaploidy present within Apiaceae (2n=48, x=8) (Covas and Schnack, 1946; Sanders et al., 1983; Grau, 1987; Kiehn et al., 2005; Talluri and Murray, 2009; Palma Rojas et al., 2012) and Campanulaceae (2n=42; x=7) (Lammers and Hensold, 1992). However, the highest variation in ploidy levels is found within the Poaceae family with tetra, hexa, hepta, and octoploid species (2n=24, 36, 42, and 48, x=6) (Baeza, 1996). The polyploidy described for Chilean Poaceae is consistent with the estimation that 80% of all Poaceae described worldwide are polyploid, with events of whole genome duplication dating back 50-70 million years, close to the origin of the family (ca. 89 Mya) (Leitch et al., 2010). Another particular case of polyploidy is the endemic genus Leucocoryne (Amaryllidaceae), where the tetraploid species 2n=18 and an exceptional case of 2n=19 have derived from cytotypes 2n=10 (x=5) by Robertsonian translocation and chromosome duplication (Crosa, 1988; Jara Arancio et al., 2012; Souza et al., 2015). To date different ploidy levels have been reported in various families of Chilean plants, but evolutionary implications of the polyploidy have not been studied in detail using cytogenetic and molecular methods. According to our dataset we estimated that ca. 21.5% of the continental species and ca. 63.1% of insular species studied so far are polyploids. This suggests that polyploidy has been a more important mechanism in the diversification of insular angiosperms than of continental ones, as described by Peruzzi et al. (2011) when comparing floras from a continental country (Italy) and an insular country (New Zealand). Up to now, only the Leucocoryne genus has been subject to estimations of polyploidy (Jara Arancio et al., 2012; Souza et al., 2015) and DNA C-value (Sassone et al., 2018). This has provided an interesting framework to understand evolutionary trends, strengthened by phylogenetic studies of their chromosome and molecular evolution (Souza et al. 2015) and also by analyzing biome shifts in relation to climatic niche evolution along their distributional area (Jara Arancio et al., 2013). We think that Leucocoryne may be good as a model plant to study evolutionary aspects regarding the origin of polyploidy due to the interesting genome data that has been documented so far.
In contrast to the quantity of cytogenetic data available for Chilean angiosperms, chromosome number information in gymnosperms is scarce, with only four Chilean species described to date. Three species are diploids such as Austrocedrus chilensis (D. Don) Pic. Serm. & Bizzarri, Pilgerodendron uviferum Florin (both 2n=22; Cupressaceae), and Araucaria araucana (Molina) K. Koch (2n=26; Araucariaceae) whereas Fitzroya cupressoides (Molina) I.M. Johnst. is tetraploid (2n=44; Cupressaceae) (Price et al., 1973). F. cupressoides is a long-lived tree, dating back at least 4,000 years in some individuals, and inhabits from 39° to 43°S at an altitude of between 100 and 1,200 m.a.s.l. Currently the species inhabits areas which were glacial refuges during Quaternary glaciations and are a relic of continuous forests of that period. Besides, this species reaches great height and because of its slow growth requires optimal conditions for germination (Premoli et al., 2000). In this regard, some hypotheses suggest that F. cupressoides may be allotetraploid, having originated by hybridization between the sympatric A. chilensis and P. uviferum (both 2n=22) (De Azkue, 1982). Unfortunately, no cytomolecular studies have been carried out to gather evidence to prove hybridization between A. chilensis and P. uviferum or/neither to corroborate genome duplication in F. cupressoides. Moreover, the findings regarding polyploidy in F. cupressoides support previous assumptions maintaining that Cupressaceae is one of the few conifer families having polyploid species (Leitch and Leitch, 2012). Despite the few species studied we estimated a mean chromosome number 2n=28.5+10.5 (n~14.2) for Chilean gymnosperms in a range between 2n=22 and 2n=44. The remaining five species have not yet been studied. Chilean gymnosperms date from Gondwana times, being related to species from the Australasia Region and in a number of cases with species from parts of Africa (Araucariaceae, Podocarpaceae) (Setogushi et al., 1998; Scapa and Catalano, 2013), whereas Cupressaceae is the only family with a virtually worldwide distribution and is represented in all continents except Antarctica (Yang et al., 2012). Gymnosperms had their origin ca. 290 Mya in the Carboniferous period with greater species diversity during the Mesozoic Era (Leitch and Leitch, 2012). To date, chromosome numbers for gymnosperms worldwide have been reported by various authors (Khoshoo, 1961; Marchant, 1968; Delevoryas, 1980; Hanson, 2001; Leitch et al., 2001; Sedel’nikova et al., 2011; Murray and De Lange, 2011), although the number of species has not been accurately determined. With this information in mind, we counted almost 150 species available in cytogenetic cadasters and estimated a mean chromosome number 2n=25.05+4.35. This estimation is consistent with the data previously reported by Murray (2013), who described a narrow range of chromosome numbers within the division (2n=14-66, with a predominant 2n=24). Chromosome numbers of the gymnosperms from New Zealand range between 2n=18 and 2n=38, and are represented by five families, three of which are shared with Chile (Araucariaceae, Cupressaceae, and Podocarpaceae) (Murray and De Lange, 2011) thus presenting greater variation and a higher number of studied species compared to Chile. Nonetheless, polyploidy is rare within the worldwide gymnosperm genera, with tetraploidy constituting the most evident level (36, 44, and 48 chromosomes) and octoploidy an extreme case reported in Ephedra (2n=56, x=7) (Sedel’nikova et al., 2011; Leitch and Leitch, 2012). In addition, B-chromosomes have been reported for almost 11 species around the world (Sedel’nikova et al., 2011) although not for the Chilean taxa.
Pteridophytes show notable cytological differences compared to both angiosperms and gymnosperms and are known to date from as far back as 400-380 Mya. Fossil records for the division have been obtained from rock strata belonging to the Silurian and Devonian periods of the Paleozoic Era. Currently, ca. 12,000 species are recognized worldwide (Pellicer et al., 2018) of which ca. 167 species from various families and genera (the major part of Gondwanan distribution) are present in Chile (Rodríguez, 1995; Teillier, 2006; Rodríguez et al., 2018). Chromosome number has only been determined for 25 Chilean species, across eight families and ten genera representing 14.5% of the total. We estimated a mean chromosome number 2n=110.8+100.3 (n~55.4) for Chilean pteridophytes in a range between 2n=22 (diploids) and ca. 2n=480 (polyploids) (Jara Seguel and Urrutia Estrada, 2021). The n value is close to the gametic chromosome number described for homosporous ferns distributed around the world which is n=57 (Soltis and Soltis, 2000).
Many fern species from various families worldwide have been described as polyploids (Brownlie, 1958; Löve et al., 1977; Roux, 1997; Bennett and Leitch, 2001; Obermayer et al., 2001; Perrie et al., 2003; Jara Seguel et al., 2006; Hennequin et al., 2010; Dubuisson and Schneider, 2010; Morero et al., 2015). Ploidy levels might be higher in pteridophytes compared to angiosperms and gymnosperms, with an estimated percentage of polyploids up to 95% (Soltis and Soltis, 2000). In the case of Chilean ferns, some species show high ploidy levels, such as the octoploid Polystichum subintegerrimum (Hook. & Arn.) R.A. Rodr. (2n=8x=328, x=41; Jara Seguel et al., 2006; Morero et al., 2015), and the tetraploids Polystichum plicatum (Poepp. ex Kze.) Hicken ex Hosseus, P. andinum Phil., P. chilense (H. Christ) Diels, P. multifidum (Mett.) T. Moore, and P. tetragonum Fée (2n=4x=164; Morero et al., 2015) all belonging to the Dryopteridaceae family. The high variation in ploidy levels described for Dryopteridaceae is striking and their phenotypic effects have been studied evaluating the so-called gigas effect on morphological features such as spore size (among other characteristics) which is larger in octoploid species from New Zealand than in similar tetraploids [P. oculatum (Hook.) J.B. Armstr. and P. wawranum (Szyszyl.) Perrie both tetraploid, and P. neozelandicum Fée an octoploid] (Perrie et al., 2003). However, adaptive advantages of the morphological differences have not been explained although they may be related to the geographical distribution and climates in which each species inhabits. Tetraploidy has been described in Asplenium dareoides Desv. (2n=4x=144; Aspleniaceae), Adiantum chilense Kaulf. (2n=4x=116; Adiantaceae), Hymenophyllum cruentum Cav. [Syn. Hymenoglossum cruentum (Cav.) C. Presl], H. caudiculatum Mart., H. fuciforme Sw., and H. ferrugineum Colla (all 2n=4x=72, x=18; Hymenophyllaceae) (Jara Seguel et al., 2006; Jara Seguel and Urrutia Estrada, 2021). A particular case is the chromosome number of Ophioglossum vulgatum L. (Ophioglossaceae) with a meiotic n=240 (Verma, 1958), which coincides with a high somatic 2n=480 (Krivenko et al., 2017). Notably, a number of diploid species have strikingly high chromosome numbers, as is the case of Equisetum bogotense Kunth. (Equisetaceae) which is widely distributed in Southern Chile, with 2n=216 (Jara-Seguel et al., 2006), whereas other genera include species with a lower 2n number, among them Hymenophyllum (2n=22, 24, 26, 28), Blechnum (2n=66) (Blechnaceae) and Megalastrum (2n=82) (Jara Seguel and Urrutia Estrada, 2021). However, 2n=66 and 2n=82 are high diploid numbers in comparison with some Chilean angiosperms (up to 2n=114) and gymnosperms (2n=22). Several of these high chromosome numbers present in the homosporous families Dryopteridaceae and Equisetaceae, 2n=82 and 2n=216 respectively, have been considered ancient polyploids, although studies based on isozyme analysis have shown that they are diploid, which has given rise to interesting debates among cytogeneticists and molecular biologists (Haufler and Soltis, 1986).
Bryophytes is a very diverse plant group with a total of 17,250 species recognized worldwide (Pellicer et al., 2018) and their origin has been dated back to ca. 470-407 Mya since the Ordovician period according to fossil records (Cox, 2018). Chilean bryophytes are extremely diverse with ca. 1,457 species being recognized (Müller, 2009), many of which have been related to taxa from New Zealand (ca. 181 species) as well as to neotropical flora (Blöcher and Frahm 2002). Data on chromosome number are scarce for Chilean bryophytes, with only 95 studied species including liverworts and mosses (6.5% of the total, according to a list published by Fritsch, 1991) belonging to 41 families and 63 genera. The mean somatic chromosome number estimated here for Chilean bryophytes in general was 28.0+14.6 (n~14) with a range from 2n=8 in the liverwort Ricciocarpos natans (L.) Corda (Ricciaceae) to 2n=96 (possible octoploid) in the moss Tortula muralis Hedw. (Pottiaceae). Fritsch (1991) gathered data on chromosome number for 1,550 taxa of bryophytes distributed around the world, but did not provide an estimated mean chromosome number. Przywara and Kuta (1995) estimated in ca. 2,242 the species of bryophytes with known chromosome number worldwide, ranging from n=4 to n=10 in Anthocerotae, n=3 or 8 to n=48 in Hepaticae, and n=4 to n=96 in Musci. In the case of New Zealand flora, a mean somatic chromosome number of 19.4+9.04 was reported for 63 species of mosses (Peruzzi et al. 2014).
Based on our estimation, which is still preliminary, it is remarkable that the mean chromosome number of Chilean bryophytes (mostly mosses) is higher than that estimated for New Zealand species, despite the substantial bryophytic similarity between both floras.
Thus, the high richness of bryophyte species so far described for Chile may be correlated with a high diversity in chromosome number, although future studies covering a more extensive geographic span would be helpful to corroborate this assumption.
Karyotypes
Karyotype morphology has primarily been studied for angiosperms and only one gymnosperm species. In the case of pteridophytes and bryophytes, karyotype studies are still a pending task and the scarce studies available to date could be a consequence of the presence of large chromosome numbers and, in many cases, the existence of small chromosomes which make measurements difficult. Nevertheless, the use of computerized methods for image analysis can potentially facilitate studies in the near future such as those performed in various plant groups many years ago (Bauchan and Hossain, 2001; Munot et al., 2011).
The first techniques used for obtaining the first karyotypes reported for Chilean plants, including the first known chromosome numbers, used histological sections of somatic tissues and male gametophytes (Whyte, 1929; Titov de Tschischow, 1954; Cave, 1966). Later, squash techniques were performed on root tip meristems treated with different antimitotic reagents, followed by fixation and stain procedures, all accepted within standard methods (Singh, 2003). Nomenclature for the description of chromosome morphology mainly follows Levan et al. (1964), which in many studies is combined with other methods to determine karyotype asymmetry (Stebbins, 1971; Arano and Saito, 1980; Romero Zarco, 1986; Paszko, 2006; Peruzzi and Eroğlu, 2013, Eroğlu 2015). In addition, interchromosomal relationships based on the ratio between the largest and shortest pair of chromosomes has provided valuable information on karyotype unimodality or bimodality.
Karyotype data have been collected for 84 angiosperm species belonging to ten families (Alstroemeriaceae, Amaryllidaceae, Asteraceae, Fabaceae, Krameriaceae, Luzuriagaceae, Myrtaceae, Philesiaceae, Poaceae and Solanaceae) all encompassed within the orders Asparagales, Asterales, Fabales, Liliales, Myrtales, Poales, Solanales and Zygophyllales. In several genera of Liliales -where the karyotypes are asymmetric and bimodal-, the largest chromosome pair is three to seven times longer than the shortest pair (e.g., Alstroemeria, Lapageria, Luzuriaga) (Jara Seguel et al., 2004; Jara Seguel and Zúñiga, 2004; Baeza et al., 2010a; Jara Seguel et al., 2010; 2021). The karyotype morphology for species of the families Alstroemeriaceae, Asteraceae, and Amaryllidaceae has been the most intensively researched in Chile, with various species and subspecies restudied using fluorescent methods (FISH, DAPI, CMA3).
Alstroemeriaceae harbors ca. 204 species distributed in Central and South America, as well as in Oceania. In Chile, this family comprises 41 species within the genera Alstroemeria, Bomarea and Luzuriaga. Karyotype morphology has been described for 18 Chilean species (Jara Seguel and Urrutia Estrada, 2021; Jara Seguel et. al., 2021). Alstroemeriaceae are known worldwide for their ornamental appeal due to their fleshy flowers and fruits, among which Alstroemeria is the most valued genus for which artificial interspecific hybrids have been produced.
Species identification in the Alstroemeria genus has been controversial and has been based mainly on morphological characteristics (Bayer, 1987; Muñoz Schick and Moreira Muñoz, 2003; Finot et al., 2018a; 2018b). Karyotype studies have served to elucidate the taxonomy of the genus in addition to molecular and morphological analyses. For instance, thanks to karyotype morphology it was possible to confirm Alstroemeria graminea Phil. within Alstroemeria (Jara Seguel et al., 2004), thus rejecting its inclusion within the monotypic genus Taltalia as proposed by Bayer (1998). A similar situation was described in the case of Bomarea ovallei (Phil.) Ravenna, for which karyotype morphology supplied additional cytogenetic data to morphology that allowed it to be classified within Bomarea, thus deleting the monotypic Leontochir (Hofreiter, 2006; Palma Rojas et al., 2007). In addition, all Alstroemeria species are characterized by the presence of an asymmetrical and bimodal karyotype 2n=16. Almost five species that inhabit arid and semiarid zones of Chile have the most asymmetrical and bimodal karyotype within the genus, whereas A. ligtu and A. presliana inhabiting humid zones in southern Chile, has more uniform chromosome sizes (Jara Seguel et al., 2021). Intraspecific variation in karyotype morphology has also been found within the complex A. hookeri Sweet subsp. hookeri which has been studied in two localities, one inhabiting the coast near the Nahuelbuta mountain range and the other the Central Valley (Baeza et al., 2010b). A particular situation has been recently published comparing A. pulchra Sims. subsp. pulchra and A. pulchra subsp. lavandulacea Ehr. Bayer chromosomes (Baeza et al., 2018). In these subspecies, both components of the homologous chromosome pair 1 -a large metacentric typical of the Alstroemeria genus- showed a notable heteromorphism in size. However, the cytological explanation given for that chromosome heteromorphism is preliminary and its frequency in the populations of both subspecies was not mentioned. It would be useful to carry out meiotic studies analyzing bivalent configuration and to conduct sequential FISH banding or chromosome painting on these subspecies in order to elucidate mechanisms of chromosome change, and more specifically on those containing pair 1. In the case of the Bomarea genus the karyotypes are less asymmetrical and uniform in chromosome length than in Alstroemeria and Luzuriaga (Palma Rojas et al., 2007; Jara Seguel et al., 2010; 2021). Alstroemeria and Bomarea grow in a wide range of environments along the length of Chile, ranging from arid and semiarid zones in the north to humid zones in the south (Muñoz Schick and Moreira Muñoz, 2003). Luzuriaga shows asymmetric and bimodal karyotypes, all 2n=20, for three intercontinental species (Chilean and New Zealand species; Jara Seguel et al., 2010). The three Chilean Luzuriaga species inhabit humid environments in the forest community of southern Chile, extending from 34°S to 53°S in Patagonia.
In the case of the cosmopolitan family Asteraceae, 927 species are found in the Chilean flora (Marticorena, 1990) and 18 species have been described in terms of karyotype morphology. To date, karyotype evolutionary trends among intracontinental and/or intercontinental taxa have been interpreted for the genera Chaetanthera, Haplopappus, Hypochaeris, Grindelia, and Taraxacum using different methods (Weiss Schneeweiss et al., 2003; Baeza and Schrader, 2005a; 2005b; 2005c; Baeza et al., 2006; Baeza and Torres Díaz, 2006, Baeza et al., 2013). For example, for New World members of Grindelia and Haplopappus their evolution has not been accompanied by large karyotype changes, although small chromosomal rearrangements have been described and differences are highlighted based on number and asymmetry level (Baeza and Schroder, 2005b). In the case of Hypochaeris, general uniformity of their karyotypes and a stable chromosome number 2n=8 have been described for South American species including Chilean taxa, though differences in the location of secondary constriction and chromosome size have also been reported (Weiss Schneeweiss et al., 2003). Secondary constrictions and Nucleolar Organizing Region (NOR) location are well differentiated characteristics among four groups of species within the Hypochaeris genus (Weiss Schneeweiss et al., 2003). Another less studied genus is Chaetanthera, which is native to South America and for which eight Chilean diploid species (2n=20, 22, 24, and 28) have been studied. These showed symmetric karyotypes with predominantly metacentric and submetacentric chromosomes (Baeza and Schrader, 2005c; Baeza et al., 2012), with one species [Ch. renifolia (J. Remy) Cabrera] being tetraploid (2n=44) presenting a highly asymmetric and bimodal karyotype with predominance of subtelocentric and telocentric chromosomes (Baeza et al., 2010c). Amaryllidaceae is represented in Chile by 114 species and 18 genera. Up to now, karyotype morphology has only been described for 42 species belonging to the genera Famatina, Gethyum, Gilliseia, Ipheion, Latace, Leucocoryne, Miersia, Miltinea, Myostema, Phycella, Rhodophiala, Placea, Rodholirium, Solaria, Speea, Traubia, and Tristagma. Seven genera of Amaryllidaceae show asymmetric karyotypes and similar chromosome morphology among species with scant variations (Baeza et al., 2004; Baeza and Schrader, 2004; 2005a; 2005c; Baeza and Torres Díaz, 2006; Baeza et al., 2007b; 2008; 2012; Cisternas et al., 2010; Sassone et al., 2018). Leucocoryne species have symmetric karyotypes and predominantly metacentric chromosomes, although telo- or subtelocentric chromosomes, and exceptionally submetacentric chromosomes are also present. It is worth noting that the total haploid set length (THL) estimated for Leucocoryne taxa (ranging from 77.5 to 147.5 μm in diploids 2n=10, and from 98.7 to 315.5 μm in tetraploids 2n=18) is higher than the values previously documented for a number of Alstroemeria taxa (2n=16, THL ranging from 53.9 to 112 μm) which have been described as having the largest genome sizes (C-values) within monocots (Jara Arancio et al., 2012).
A number of hypotheses have been proposed to explain the origin and adaptive significance of the karyotype asymmetry and bimodality (Stebbins, 1971; Vosa, 2005). Karyotype bimodality is a feature that has been related to a specialized type of nuclear architecture that can be independent of the genetic status (White, 1973). According to our dataset for Chilean plants, karyotype asymmetry with or without bimodality versus karyotype symmetry are present in species of various unrelated families inhabiting different environments, whether arid or humid. So it is possible that rather than representing an evolutionary adaptation to environmental conditions as described above, these karyotype structures could be reflecting a certain degree of karyotype conservation retained from their respective ancestors. Nevertheless, minor or major modifications of such structures are related to adaptive changes during their evolution. In Alstroemeria species, changes in asymmetry and bimodality are correlated with variations in total chromosome length and DNA C-values, each of them related to a type of environment in particular (Buitendijk and Ramanna, 1996; Buitendijk et al., 1997; Jara Seguel et al., 2004; 2021). Sanso (2002) offers a preliminary explanation to this question in his studies of Alstroemeria species, suggesting that these characteristics may be due to karyotypic orthoselection or karyotype conservation.
Within the Chilean gymnosperms only the karyotype morphology of Araucaria araucana has been described (Hodcent, 1968; Bandel, 1970; Cardemil et al., 1984). In general, the authors agree on the presence of 26 chromosomes of metacentric and submetacentric morphologies, symmetric and with wide variations in size, with large and small chromosomes within bimodal karyotypes. In addition, these karyotype characteristics of A. araucana are similar to those described for 14 species of the family Araucariaceae found along a distributional range in the southern hemisphere (Hizume and Kan, 2015). Gymnosperms live principally in the Andean and Nahuelbuta mountain slopes at altitudes between 1,000 and 1,600 m.a.s.l., although some small populations of F. cupressoides inhabit the Central Valley of southern Chile.
Classic banding and fluorescent methods
Different banding methods have been performed in Chilean angiosperms (Jara Seguel and Urrutia 2012, Jara Seguel et al., 2021). To date, ten species of the Alstroemeriaceae family belonging to Alstroemeria and Bomarea have been studied by means of classic C-banding. In Alstroemeria species, the haploid relative length values of the C-bands vary between 2.0 and 6.5% (Buitendijk and Ramanna, 1996; Jara Seguel et al., 2004), while for Bomarea ovallei (Syn. Leontochir ovallei) this parameter reaches 20% (Jara Seguel et al., 2005). Within the genus Alstroemeria, considerable intraspecific and interspecific variation in C-band relative length and chromosome location of constitutive heterochromatin have been observed (Jara Seguel et al., 2021). These features have proved to be additional to heterozygosity in the size and location of C-bands among homologous chromosome pairs in a number of species (Buitendijk and Ramanna, 1996). The presence of large C-bands has been co-related with large chromosome size and high nuclear DNA content, and these features have been associated with geographical distribution and climate on a latitudinal gradient in line with increases in total chromosome length (Buitendijk and Ramanna, 1996; Buitendijk et al., 1997; Jara Seguel et al., 2004; 2021). C-banding has been an important tool to describe genome complexity in a number of Alstroemeriaceae species. For this reason, and due to the use of conventional microscopy and low-cost reagents, the C-band technique is affordable for any laboratory and could be performed in more angiosperm families, thus providing further valuable data on genome structure and dynamics. This information may be fundamental for the application of other modern molecular techniques such as cloning, sequencing and in situ hybridization of either C-heterochromatin regions or ribosomal cistrons, all focused on gaining more in-depth understanding of the phylogenetic relationships among species. In the case of the Ag-NOR method, only Rhodophiala laeta (Amaryllidaceae) has been studied, for which active rDNA sites have been located in chromosomes (Jara Seguel et al., 2012).
The application of fluorescent banding such as DAPI, CMA3 and FISH has been a key step in the study of genome characteristics in Chilean species belonging to the genera Alstroemeria, Chaetanthera, Grindelia, Haplopappus, Hypochaeris, Leucocoryne, Nolana, Placea, and Rhodophiala (Kamstra et al., 1999; Weiss Schneeweiss et al., 2003; Zhou et al., 2003; Baeza et al., 2004; Baeza and Schrader, 2004; 2005a; 2005b; 2005c; Baeza et al., 2007a; Chacón, 2012a; Souza et al., 2015; Lujea and Chiarini, 2017). To this end, the Antarctic species Deschampsia antarctica has also been studied, resulting in a total sum of 24 studied species. These modern methods have revealed interesting details on chromosome structure, supporting species relationships for the majority of the genera. Interestingly, mechanisms of duplication of 5S rDNA sites exhibited wide variability in the chromosome distribution of a number of Leucocoryne species (Souza et al., 2015). Hypotheses on genome evolution within Leucocoryne and Alstroemeria have been proposed and have also extended to phylogenetic reconstructions (e.g., Alstroemeria, Chacón et al. 2012; Leucocoryne, Souza et al., 2015).
Chilean gymnosperms have been scantly studied using banding methods. In the sole case of Araucaria araucana, fluorescent banding using chromomycin A3 (CMA) and 4′, 6-diamidino-2-phenylindole (DAPI) has been reported to date (Hizume and Kan, 2015). Large and thick CMA-bands at the proximal region or secondary constriction of two long metacentric chromosomes have been described as a remarkable genome characteristic in A. araucana. In addition, several weak DAPI-bands have been observed at the interstitial and/or centromeric regions of some chromosomes. For this reason, most chromosome pairs were identified by means of chromosome shape and fluorescent banding pattern. The application of the Ag-NOR technique in Araucaria araucana has been crucial for the identification of the chromosome location of active rDNA (Cardemil et al., 1984).
In general, the scarce available data obtained for Chilean plants suggests that fluorescent chromosome banding is focused on physical chromosome gene mapping of specific sequences or DNA fragments. This opens the way to comprehensive studies on genome affinities and dynamics (e.g., meiotic chromosome behavior, chromosome rearrangement, rDNA location), where promising advances in genome structure and functionality can be achieved. However, much remains to be done in the future for a large number of Chilean species, in particular for those with unknown karyotypes, e.g. the allotetraploid Fitzroya cupressoides, a long-lived species that can reach up to 4,000 years old.
Nuclear DNA content
Nuclear DNA content, C-value and genome size are recognized as a strong unifying element in biology, having practical and predictive applications across many fields of science such as ecology, biogeography, physiology and embryology (Bennett and Leitch,1997; 2005b; Gregory, 2005; Kraaijeveld, 2010; Greilhuber et al., 2010; Grover and Wendel, 2010; Leitch and Leitch, 2012; Pellicer et al., 2018). Many authors have documented data on C-values including local and global floras of different continents, with ca. 12,000 species described (Leitch et al., 2017; Pellicer et al., 2018). In addition, phylogenetic reconstructions have facilitated the understanding of trends on genome size evolution in land plants, suggesting that each major group has been subject to different evolutionary or selective forces (Leitch et al., 2005; Smarda et al., 2014; Carta and Peruzzi 2016) resulting in a remarkable diversity (Pellicer et al., 2018).
The C-values of only 22 Chilean angiosperm species have been studied to date, representing less than 0.5% of the total studied worldwide (ca. 10,768 species, Leitch et al., 2017). Within Alstroemeriaceae, seven species belonging to the genus Alstroemeria have been among the most studied, for which C-values ranged between 19.9 pg in A. pulchra subsp. pulchra and 34.7 pg in A. ligtu L. subsp. ligtu (Buitendijk et al., 1997), which is additional to data available for Bomarea salsilla Mirb. (C=10.3 pg) and Luzuriaga radicans Ruiz & Pav. (C=6.6 pg) (Smarda et al., 2014). It is worth noting that C-values of Alstroemeria species fall within the largest genome sizes of the Plantae kingdom (Sanso and Hunziker, 1998), although they are lower than the maximum 1C=152.2 pg described for monocots (Leitch and Leitch, 2012). Other studied genera are Krameria (Krameriaceae, Palma Rojas et al., 2017), Leucocoryne and Tristagma (Amaryllidaceae, Sassone et al., 2018). Leucocoryne shows C-values around 28-30 pg, although somatic values vary; for example, in diploid species 2C-values are around 56-60 pg, in triploids they are close to 86 pg, whereas in tetraploids the range spans from 115.6 to 121.8pg. In the case of Tristagma, 2C-values range from 33 to 35.5 pg in diploid species and close to 66.48 pg in tetraploids (Sassone et al., 2018). On the other hand, Lapageria rosea has an intermediate C-value of 6.8 pg (Bennett and Leitch, 2005a), whereas small C-values (Prosopis 1C=0.4 pg, Berberidopsis 1C=0.3 pg, and Fuchsia 1C=1.46 pg) (Bukhari, 1997; Bennett and Leitch, 2005a; Talluri and Murray, 2009) have been described for the remaining Chilean genera studied to date. These values are lower than the average 1C=5.9 pg estimated for angiosperms (Leitch et al., 2005; Leitch and Leitch, 2012).
Studies on C-values for Chilean gymnosperms are scarce, with data only available for the diploids Araucaria araucana (C=22.7-25.8 pg, Price et al., 1973; Zonneveld, 2012), and Austrocedrus chilensis (C=21.7 pg, Price et al., 1973), in addition to the allotetraploid Fitzroya cupressoides (C=10.2 pg, Price et al., 1973). Note that this C-value of F. cupressoides is inconsistent with the possible genome duplication regarding the C-value of their putative parents A. chilensis and P. uvirerum, which, although not yet studied, are estimated to have a C-value close to 21 pg with 22 chromosomes. This situation suggests that more complete studies are required to understand the polyploidization process in F. cupressoides. Worldwide, C-values are known for ca. 421 gymnosperm species (ca. 25% of the total) with a mean C-value of 18.8 pg ranging between 7.2 and 36 pg. In general, the C-value variation in gymnosperms is low, being of conserved lineages due to their relatively narrow range of chromosome numbers, constancy in chromosome numbers and karyotypes within genera and families, rare polyploidy, and displaying the lowest recombination rates reported so far in any eukaryotic lineage (Pellicer et al., 2018).
C-values have been scarcely studied for Chilean pteridophytes and bryophytes. At present only the horsetail fern Equisetum bogotense has an estimated C-value of 21.3 pg (Christenhusz et al. 2021). Given the high incidence of polyploids (octoploids up to 2n=480) or the presence of diploid species with high chromosome number (2n=66, 82, 216) within pteridophytes, C-value estimation might elucidate interesting aspects on genome structure and complexity. A similar situation may occur within the bryophytes, with high polyploidy (up to octoploidy) being frequent within Chilean taxa, as has been described for species from the northern hemisphere (Kuta and Przywara, 1997). Therefore, efforts should be focused on the use of methods for estimating C-values via flow cytometry (Voglmayr, 2000; Bennet and Leitch, 2001; Obermayer et al., 2002; Hanson and Leitch, 2002) or image microdensitometry (Voglmayr, 2000; Palma Rojas et al., 2017), thus increasing the data sets available worldwide for these diverse plant groups (Leitch et al., 2017). At a global level, C-values have been estimated for 303 species of pteridophytes and 309 species of bryophytes (Pellicer et al., 2018). Mean C-values for pteridophytes vary between 5.15 pg (in diploids) and 4.59 pg (in polyploids), whereas the mean C-value in bryophytes has been estimated at 0.49 pg (Bennett and Leitch, 2001).
Prospects
The high floristic endemism recognized so far for continental and insular Chile (45% in the continent and 64-87% in insular areas) has a significant world biodiversity heritage value. This characteristic has been favorable for the inclusion of large extensions of the Chilean territory -continental and insular- within worldwide biodiversity hotspots. However, as shown in this review, the scarce knowledge on cytogenetic characteristics suggests that a series of aspects related to the genome structure, dynamics and evolution of Chilean land plants are unknown for ca. 91.8% of the species, including a basic feature such as chromosome number. The knowledge gap is alarmingly deeper for many species, as neither chromosome morphology and mapping nor nuclear DNA content have been studied to date. Nonetheless, these knowledge gaps are a challenge for the future of cytogenetics in Chile. It is important to note that many native plant species have been studied in parallel using modern molecular methods such as DNA fingerprinting and gene sequencing or DNA barcoding, although no reports have been made in terms of the determination of total number of species and their taxonomic representation to date. Currently, important advances in genome knowledge on land plants from other geographical areas around the world have facilitated the understanding of different levels of genome structure, complexity and dynamics, even in the field of ecology. Many of the Chilean plants still not included in that valuable knowledge could be of relevant importance to assess their adaptive capacity prior to the climate crisis that has been affecting Chile and other regions of the continent over the present decade. For these reasons, a collaborative effort between cytogeneticists and, if possible, a peer undertaking with molecular geneticists and evolutionary ecologists could help to develop an integrated strategy to accelerate progress and evaluate plant response to different ecological settings. This endeavor would obviously require funding and the onus is on state institutions to channel the necessary financial resources to accomplish this key task. In addition, training of more specialists in cytogenetics is necessary to address as many taxonomic plant groups as possible. The cytogenetic information provided here for Chilean native plants -from continental, insular and Antarctic zones- constitutes an important contribution to knowledge of the South American and worldwide flora, given the high endemism that not only encompasses its own genomes, but also highlights their close relationship with other floras such as those in Oceania, part of North America, and Central America, bringing them together as part of a floral kingdom which has evolved for millions of years resisting dramatic geological and climate changes such as have occurred in the Southern cone.












 uBio
uBio