Avian botulism is a common cause of death for waterbirds worldwide. It occurs when the neurotox- in produced by the bacterium Clostridium botulinum is ingested. Under anaerobic conditions, the presence of suitable nutrients, and favorable temperature, the botulinal toxin (neurotoxin) is synthesized and released (Brand et al. 1983). There are eight types of botulinal toxins, named from A to G, and among them, type C is the one that most often affects birds (Rocke and Bollinger 2008). This neurotoxin pro duces a general flaccid paralysis by interfering with acetylcholine release at the neuromuscular junction, affecting skeletal muscles, usually the wings first, and progressively affecting the lower limbs and neck (Nei- manis et al. 2007). Large outbreaks of type C botulism have been documented in gulls (Macdonald and Stan- dring 1978, Gophen et al. 1991, Neimanis et al. 2007) and in other aquatic species such as grebes, ducks, herons, shorebirds, geese, coots (Baldone et al. 2006, Shin et al. 2010, Defilippo et al. 2013, Wlodarczyk et al. 2014), and also mammals such as coypus (Defilip po et al. 2013).
Here, we describe the first confirmed outbreak of avian botulism in two islands located at the Nahuel Huapi lake (Nahuel Huapi National Park, Rio Negro province, Argentina), where Kelp Gulls (Larus domin- icanus) breed (Fig. 1).
The Nahuel Huapi lake, located in northwestern Patagonia, is one of the largest (ca. 560 km2) and deepest (~ 400 m) lakes of Argentina. The lake is with- in the Nahuel Huapi National Park (> 700 000 ha). It is an oligotrophic lake with a large shoreline that al- ternates between rocky and sandy coasts. There is lit- tle aquatic vegetation and the forest usually extends to the shoreline. The city of San Carlos de Bariloche and other small towns are found along its coastline. The temperature in the area fluctuates between -2°C in winter and 23°C in summer (Paruelo et al. 1998, Mermoz et al. 2000). Many water birds inhabit the area (53 species from nine families), some of which also breed in the area, such as the Ashy-headed Goose (Chloephaga poliocephala), the Flying Steamer Duck (Tachyeres patachonicus), the Red-gartered and the White-wiged Coot (Fúlica armillata and F. leucop- tera), and the Great and Silvery Grebes (Podiceps major and P occipitalis), and ducks from the Anatidae family (Ojeda et al. 2016). The Kelp Gull also nests in some of the islands found in the lake (Frixione et al. 2012, N. Rosciano pers. obs.).
During the breeding season of 2019-2020, we were conducting fieldwork at the Kelp Gull colonies (Fig. 1). On 17 January 2020, when most chicks had fledged, we found several adult gull carcasses in the colony, nine of them on ventral recumbency and with green stained ventral feathers. We also observed two adult gulls sitting on the ground, unable to stand or fly even when we approached them, and two juveniles of Ashy-headed Goose with difficulties to walk, stum- bling themselves and using their wings to move for- ward when approached.
Based on the clinical presentation and previous experience of one of the authors (S. Seijas), a botulism outbreak was suspected. On January 27 2020, we or- ganized fieldwork to collect samples for toxicological analysis. By that time, the sick birds were dead and new birds were found sick. We collected blood from six Kelp Gulls showing clinical signs and pieces of in- testine from three Kelp Gulls and one Ashy-headed Goose fresh carcass (following Uhart and Zaccagnini 1999, Villar et al. 1999).
All samples were kept refrigerated in the field in a portable cooler until arrival at the laboratory. Plas ma and red blood cells were separated (5 minutes, 4000 rpm, 10°C) and preserved in a -20°C freezer. All samples were analyzed at the Servicio de Bacteriología Sanitaria (SBS) - Instituto Nacional de Enfermedades In fecciosas ANLIS Dr. C. Malbrán. From the total of nine birds sampled (eight gulls and one goose), only six were suitable to perform the toxicological tests (three blood and three intestine samples); the other three were discarded because there was not enough volume or the quality was not appropriate for the analysis.
The plasma fractions of the collected blood were used to detect the presence of botulinum toxin by di- rect inoculation on albino mice (18-20 g), following the technique described by Villar et al. (1999). Feces were extracted from the collected intestine samples and cultured in Tarozzi medium (BBL Cooked Meat Medium - BD) at 37°C for 5 days; then centrifuged and filtered (millipore filter 0.45 pm) and inoculated to al bino mice (18-20 g). One out of six samples resulted positive for botulinum toxin type C (the Ashy-headed Goose lower intestine tissue).
Five individuals of Kelp Gull and one individual of Ashy-headed Goose with clinical signs were captured for rehabilitation. They were treated symptomatical- ly with fluids, assisted feeding and supported care. Only one Kelp Gull and the Ashy-headed Goose recov- ered after a period of 4 days; both were returned to the colony on 5 February. During this new visit to the colony, we found seven adults of Kelp Gull with sim ilar clinical signs as in January. Further visits to the colony were not possible that summer due to logistic constraints.
During each of the visits to the colony, we made field interventions to manage the spread of the out- break by collecting carcasses in different stages of decomposition and burying the larvae dropped from the carcasses after collection to prevent other birds scavenging on them; later, back in land, we incinerat- ed the carcasses as pathologic waste (Reed and Rocke 1992).
Overall, during the outbreak (approximately 20 days since the first birds with symptoms were found to the last day we attended the colonies), a total of 12 adults and three juvenile Kelp Gulls were alive and with clinical symptoms, while 22 adults and two ju- venile Kelp Gulls were found dead. These numbers represent approximately 2.5% of the adult and 1.4% of the juvenile Kelp Gulls’ population accounted for in the colonies during that summer. In addition, on the same islands, we found a total of three juvenile geese with clinical signs and the carcasses of two adult Ashy-headed Goose.
Diagnosis of botulism was based on clinical evi- dence and confirmed by the demonstration of toxin in the feces of affected birds. The clinical signs observed in the birds during the sampling period and the posi- tive result for botulism type C supports the diagnosis of avian botulism type C in the Ashy-headed Goose and the Kelp Gulls. An epidemiological link was as- sumed for all birds that presented similar symptoms, and therefore it was presupposed that they were also affected by the same toxin since all the birds co-exist- ed at the same place and time with the bird that was diagnosed positive.
Clinical signs of affected geese and gulls were consistent with those described in literature (e.g. Nei- manis et al. 2007). Affected birds displayed a range of progressively flaccid paralysis, with mildly affected birds incapable of flight, but able to stand and run, while moderately affected birds were in sternal re- cumbency with drooping wings. With the exception of moribund birds which were dull and poorly respon- sive, all birds were alert, responsive, and aggressive and had mobile heads. Staining of the ventral feathers with green feces was frequent.
This is the first confirmed case of botulism, caused by a type C botulinum toxin, in the Nahuel Huapi lake and the National Park area. It represents an important finding for the study area since previous cases of birds showing similar symptoms were ob- served in other lakes of the region in summer 2019: three Ashy-headed Goose and three Black-faced Ibis (Theristicus melanopis) in Gutierrez, Correntoso and Guillelmo lakes. For all these individuals it was not possible to confirm the presence of a toxin (S. Seijas, unpublished data).
Birds affected by botulism may have low amounts of toxin in their blood and thus this disease may not be detectable. Therefore, it is advisable to test a large number of birds with different degrees of the condi- tion (Rocke and Bollinger 2008). In other studies, the toxin was detected by serum inoculation in mice in the range of 50-70% (Neimanis et al. 2007 and refer- ences therein). Additionally, other tissue proved to be good for toxin detection (e.g. Rose 1934) and, in our case, the intestine samples allowed us to extract the feces and to culture them to detect the presence of the toxin. The level of sensitivity of the toxin detection in mouse inoculation is also highly specific and it is not sensitive to low levels of toxin that are often present in clinically affected birds. Therefore, with the small sample size we had in this study, there were small chances of obtaining a positive result.
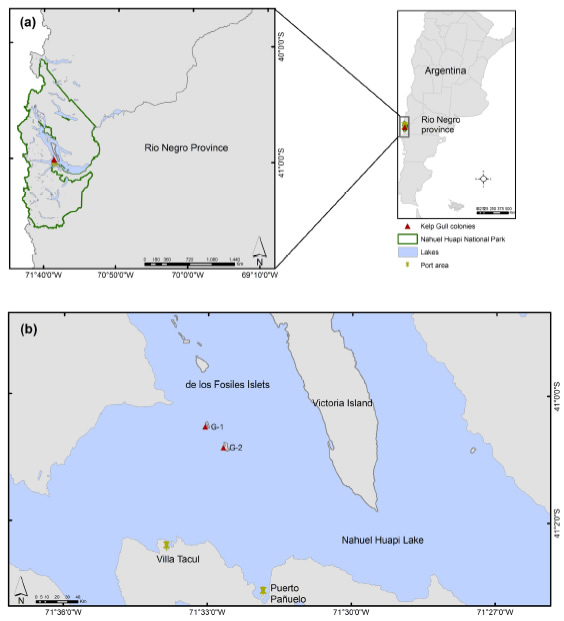
Figure 1: (a) Map and location of the Nahuel Huapi National Park, encompassing the Nahuel Huapi lake, on the west of Rio Negro province, in Patagonia Argentina. (b) The red triangles indicate the location of the Kelp Gull colonies on the Nahuel Huapi lake, where the botulism outbreak occurred.
The reasons for the reported outbreak, as well as its extension, remain unknown. The source of infec- tion usually occurs in aquatic environments, when favorable conditions such as high temperatures and anaerobiosis induce C. botulinum bacteria to produce toxic spores (Rocke and Bollinger 2008). The spores can be found in freshwater habitat, sediments, aquat- ic invertebrates, mollusks, and crustaceans, but also in certain vertebrates’ tissues (e.g. bacteria colonize the intestinal tract of individuals; Reed and Rocke 1992, Latorre 2010). Outbreak detection coincided with a period of maximum temperatures recorded during that summer in the Nahuel Huapi lake area (maximum average: 23°C) and also with the lowest water levels of the lake in the season. Water ponds on the island may be a suitable environment and source for toxigenesis. Deposition and accumulation in these ponds of carcasses of small aquatic or terrestrial in- vertebrates that held the toxin within the bacterial cells may have acted as the source of the outbreak (Wobeser 2007). Although other birds may have been exposed to the toxin in different areas of the Nahuel Huapi lake system, once the outbreak began, possibly the carcasses of affected birds became an important source of toxin through larvae and other scaveng- ing invertebrates (Reed and Rocke 1992, Wobeser 1997). The colonial habits of Kelp Gulls, combined with their generalist and opportunistic foraging be- haviors, may have favored an rapid spread of the toxin, thus affecting an important number of birds (Macdonald and Standring 1978, Gophen et al. 1991, Neimanis et al. 2007).
Diseases such as botulism are usually lethal for wildlife and have significant effects on bird popula- tions. The extent of the outbreak reported here re- mains unknown, but the impact on the Kelp Gull col- ony did not seem to be of great concern (~2.3% of the population), compared to events reported in other ar- eas of the world (Macdonald and Standring 1978, Go phen et al. 1991, Neimanis et al. 2007, Veloso Soares 2014). However, specific research on the source of the toxin and on the initiating conditions of these out- breaks would be of great importance to understand the potential threats to the environment and to differ ent bird populations that live in the study site (Neima nis et al. 2007, Shin et al. 2010, Defilippo et al. 2013). This would be key in the Nahuel Huapi National Park area, since aquatic birds such as the Ashy-headed and the Upland Goose (Chloephaga picta) are both consid- ered as threatened species in Argentina (MADyS and AA 2017). Early detection of potential outbreaks can trigger protocols for carcass management and ade- quate sampling methodology across different areas of the lake. It is important to highlight that the botulinal neurotoxin type C affects only birds and it is not toxi- genic for humans (Rocke and Bollinger 2008). Howev er, future outbreaks should not be overlooked, since the accumulation of intoxicated carcasses near water supplies and coastal areas can lead to contamination and could affect many species. This is important for the Nahuel Huapi lake and National Park areas that are intensively visited by tourists and residents for recreational activities, especially during warm sum- mer days.
Finally, we recommend the implementation of regular monitoring programs for these colonies to control potential outbreaks (e.g., collecting and incin- erating or burying carcasses that may be the source of contamination found at the colonies to prevent other animals from accessing the toxin) and to record the impact on the bird population. These actions will be key to understanding the ecological factors associated with this kind of outbreaks in the Nahuel Huapi lake and National Park area, while mitigating their conse- quences on the ecosystem, particularly on waterbirds.
acknowledgments
We appreciate the help from J. Nielsen, J. Miranda and E. Margarido from the Conservation Department of the Nahuel Huapi National Park, who responded with celerity to the logistic needs presented during the outbreak. Dr. S. Barandarian and vet. M. Ibañez Molina for providing contacts and technical support. Vet. M. Wainer for assistance in the birds’ rehabili- tation process. Universidad Nacional del Comahue (CRUB-UNComa) provided the boat for transport to the islands, and N. de la Cruz and S. Dominik pro vided the mooring. CAU-CAU Company contributed financially to the project. This research was conduct- ed under permit #1588 from the National Park, and the outbreak was managed under the Nahuel Huapi National Park Emergency Contingency Plan, file EX - 2020 -08461368-APN-DGA#APNAC. We thank A. Quaglia, M. Saggesse, an anonymous reviewer and the associate editor D. Canal for their valuable comments and suggestions that helped improve the manuscript.












 uBio
uBio 

