Introduction
The intensification of agriculture has made crops more vulnerable to pests. Agricultural practices such as multiple crops per growing season, reduced fallow and monocultures have contributed to creating favourable conditions for the emergence of pests and to reducing natural barriers to their spread10. Ecosystem will be inevitably affected by the action of pesticides, due to their toxicity, persistence and bioaccumulation.
The herbicide 2,4-D (2,4-dichlorophenoxyacetic acid) is one of the oldest synthetic pesticides. It was released in the forties. Being currently one of the most used agrochemicals in agriculture19. It is within the group of phenoxy or phenoxyacetic or chlorophenolic herbicides. It is found within the “hormonal herbicides” because it acts similarly to the natural hormone auxin, or indol-3 -acetic acid (AIA)3. 2,4-D has been classified slightly and moderately dangerous (class II and III)28.
Fish are particularly targeted for contamination, for this reason, they are used as a model for the assessment of pollution in aquatic ecosystems21. Laboratory tests with fish showed that several substances were potentially genotoxic17, while others proved harmless4.
Due to the speed, sensitivity and reliability of the technique of micronucleus and nuclear morphological alterations (MN and NMA) in peripheral blood of fish, it is intensively used for the evaluation of contaminants in aquatic environment.
For this reason, the aim of the present work was to evaluate the possible impact of a contamination with 2.4- D, in sublethal concentrations in Piaractus mesopotam icus, through the frequency of micronuclei and alterations in the shape of the nuclei in erythrocytes of peripheral blood.
Materials and Methods
The trials were conducted with juvenile specimens of P. mesopotamicus commonly known as “pacu”. The fish were acclimatized for 30 days prior to treatment. Two trials were conducted, one with the herbicide 2,4- D pure (P) and another with a commercial formulation, 2,4- D Sumargo amine (FC).
In the experiments 6 aquariums of 20 liters were used with 2 specimens in each one. The duration of the trial was 70 days. Two groups were considered (treated and control). Different concentrations (1 ppm, 1.8 ppm, 3.2 ppm, 5.6 ppm and 10 ppm) of the herbicide were placed in the treated group and only water from artesian wells was added in the control.
After 70 days, the animals were anesthetized with xylocaine to extract blood from the caudal vein. For the analysis of micronuclei in peripheral erythrocytes, a drop of blood was placed on a slide for smears. The smears were fixed in ethanol for 10 minutes and left to air dry at room temperature; they were then stained with 5% Giemsa in Sorenson buffer (pH 6.9) for 20 minutes26.
The analysis of the smears was carried out by randomly sweeping the smears to count 4000 cells per individual. The MN and NMA were classified with the suggested criteria for fishs5. The program Infostat version 2013e 9 was used for statistical analysis. The data were submitted to the variance analysis and Tukey’s statistical test.
Results
In the 22 P. mesopotamicus specimens studied, an average of 16 erythrocytes per field was observed, with a total of 4000 cells per individual. In order to determine the frequency of micronuclei and nuclear morphological alterations, the shape and structure of the cells were taken into consideration.
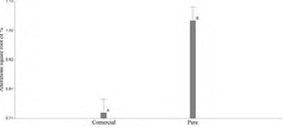
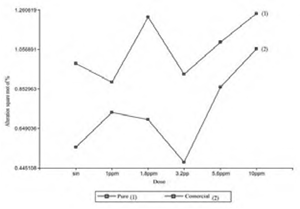
The following alterations were identified: MN: micronuclei; BN: binuclei; LOB: nuclear lobulations (lobed nuclei and blebbed nuclei); MUN: nuclear notches (notched nuclei); VAN: vacuoles (vacuolated nuclei) (Figure 1). In each bioassay, 8,000 cells were counted for the control group and 36,000 for the treated group, evaluating a total of 16,000 erythrocytes (controls) and 72,000 (treated) for presentation (P) and (FC). In individuals exposed to the pure herbicide, 81 (1%) cells with MN and AMN were evidenced; while in those treated, 416 (1.15%) were identified. On the other hand, P. mesopotamicus specimens in contact with the commercial formulation showed 25 (0.31%) cells with MN and NMA, and those treated showed 241 (0.66%) red blood cells with abnormalities (Figure 2).

Figure 1 Erythrocytes of Piaractus mesopotamicus. a) Normal. b) Micronucleous. c) Binuclei. d) Nuclear lobulations. e) Nuclear notches. f) Vacuolated nuclei.
The study of the data obtained revealed that the control individuals treated with pure 2.4-D presented higher amounts of MN and NMA compared to the specimens exposed to the commercial formulation. When contrasting the control and treated group within each experience the differences are 0.15% (2.4-D pure) and 0.35% (2.4-D amine Sumagro).
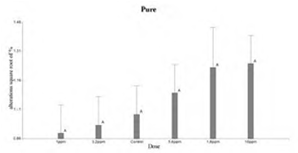
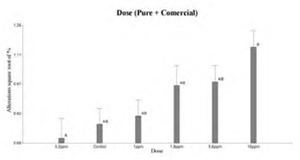
Figure 3 shows that the doses used of pure 2.4-D were not statistically significant with respect to control. Whereas, in Figure 4 the 10 ppm dose of the commercial formulation was significant with the control. When comparing the 2.4-D pure and FC treatments, Tukey’s test showed significant differences in the behavior of both trials (Figure 5 and Figure 6). On the other hand, comparisons according to the dose of the herbicide, did not show differences between controls and treated (Figure 7).
Discussion
When comparing the data obtained in both trials, pure 2.4-D showed a higher amount of MN and AMN in the control and treated groups. This difference was probably due to the fact that both experiences were not carried out simultaneously in time. This led to the assumption that in the trial with pure 2.4-D other factors inherent to the treatment itself, such as temperature, oxygen level, etc, could be interacting. The impact of external factors such as temperature were demonstrated and evaluated in genotoxicity tests in fish18.
The situation in this work where only P. mesopotamicus specimens subjected to the 10 ppm concentration of the herbicide 2.4-D amina Sumagro showed a significant difference with the control could be due to the fact that besides the active principle (2.4-D), these may have other compounds as byproducts of their industrial synthesis such as polychlorinated dibenzodioxins15, nitrosamines13 and other chlorinated phenols that may express clastogenic activity13.
On the other hand, manufactured products contain adjuvants (solvents, thinners, dispersants, emulsifiers, enhancers) in unknown proportions 30. There are works in which it is evidenced that commercial formulations both in vivo and in vitro induce a greater genotoxic activity than pure drugs29.
On Oncorhynchus mykiss exposed to the commercial formulation of 2.4-D 480 for 24 and 96 hours revealed significant differences in the frequency of micronucleated erythrocytes in control and treated individuals exposed to 10 ppm of the herbicide. No discrepancies were observed in concentrations of 2 ppm and 20 ppm regarding the control; although dilutions of 0.8 ppm and 5 ppm showed differences at 96 h and 24 h, respectively.
According to the author, this may be due to factors such as inhibition of hematopoiesis or the action of DNA repair enzymes20. In the same way, specimens of Cnesterodon decemaculatus contacted at doses 252 mg/l, 504 mg/l and 756 mg/l of the commercial formulation DMA (58.4% 2,4D) showed at 48 h and 96 h an increase in the frequency of MN and NMA, evidencing significant effects and for the 3 doses used 25.
In relation to the pure 2.4-D trial, no statistically significant differences were observed between the control and treated individuals. This differs from what was found in specimens of Clarias batrachus with the pure herbicide 2,4-D at doses of 25 ppm, 50 ppm and 75 ppm during 48, 72 and 96 hours 2. On the other hand, similar toxicity test was performed in Channapunctatus which evaluated the genotoxicity of pure 2,4-D with the same concentrations and time11. In both studies, significant differences were observed in the amount of micronuclei and nuclear morphological alterations in the erythrocytes of the treated individuals with respect to the controls. The data reveal the existence of a positive correlation between the amount of micronuclei and nuclear deformations depending on the time and dose used. The results of these authors showed significant differences even in the lowest dose 25 ppm, which exceeds the concentration of 10 ppm of the present work. This difference probably led to not finding significant values in our dilutions.
It should be noted that despite the similarity in concentrations and exposure times cited in Clarias batrachus 2 and Channa punctata 11 , it was observed that the alterations differ in number, this could be due to the sensitivity of the bioindicator (species). Such an assumption leads to the consideration that the response of P. mesopotamicus to the herbicide may have been due to its rusticity. The way species react to different xenobiotics seems to play a fundamental role in the evaluation of contaminants.
Comparative studies of the frequency of micronuclei and nuclear abnormalities in erythrocytes in three fish species C. carpio, Astyanax eigenmanniorum and Cheirodon interruptus, from Villa Dalcar lagoon (Río Cuarto, Córdoba, Argentina) showed presence of MN and NMA, being more frequent in A. eigenmanniorum and less in C. carpio 23. The sensitivity in the species could have certain relation with the rusticity of each one of them16.
Thus, it was also shown that Salmo trutta (brown trout), Salmo trutta (european eel) and Phoxinus phoxinus (european minnow) species exposed to cyclophosphamide, cadmium and colchicine for 72 hours show differences in response to genotoxic agents. In specimens of S. trutta all three chemical compounds induced MN. Phoxinus phoxinus evidenced micronuclei only at cadmium exposure. However, Anguilla anguilla did not present significantly MN in any of the treatments24.
It is known that MN formation occurs during cell division as a result of two events: chromosomal breakage and/or mitotic spindle dysfunction, which can lead to incomplete distribution of chromosomes in daughter cells during mitosis1. The frequency of MN in peripheral erythrocytes is then the result of the dynamic balance between the formation of micronucleated cells and their elimination, where a modification of the basal MN frequency would reveal an alteration in one or both processes 22.
Entry of erythrocytes into circulating blood may be caused either by new cell production by the cephalic kidney, or under stressful conditions, by the release of cells stored in the spleen. Splenic contraction has been shown in fish in response to exercise and hypoxia 14.
Elimination may be by apoptosis 7or splenic removal which has been demonstrated in mammals6, although the latter would not affect to a large extent, as the elimination mechanism involves spleen sinusoids and fish have a non-sinusal structure27. The balance between the processes that are promoting MN frequency may vary over time27. Previous studies have suggested that exposure to contaminants inhibits erythropoiesis8.
Consequently, fewer erythroid cells are subjected to differentiation into potentially micronucleated and altered erythrocytes. Then the formation of MN and NMA decreases, leading to a change in balance. As a result, the peak of micronucleated erythrocytes is observed between 1 and 5 days after exposure to contaminants 1, 27. This duration is short compared to the average life span of circulating erythrocytes estimated at 51 days for Carassius auratus langsdorfii 12.
Since in this work the fish remained under the exposure of the herbicide for 70 days with herbicide renewal every two days, this time allowed all circulating erythrocytes to be under the influence of the agrochemical. Therefore, it would be expected that the presence of MN and NMA should be greater in both bioassays, however this increase was only evident in fish treated with FC at a dose of 10 ppm.
Therefore, the data from this experience allow us to conclude that only the juveniles of Piaractus mesopotamicus put in contact with the 10 ppm concentration of the herbicide 2,4-D amine Sumagro showed a significant increase in the frequency of erythrocytes with micronuclei and nuclear morphological alterations with respect to the controls. The results differ from those existing in the literature, possibly due to different causes: 1) the concentrations used were higher than those of the present work; 2) the sensitivity expressed by the different species exposed to 2,4-D; and 3) exposure time to the contaminant agent and the variability in the response in acute and chronic trials.