INTRODUCTION
Zephyranthes Herb. is a genus of perennial bulbous plants belonging to the Amaryllidaceae family, which stands out for its high ornamental potential and, at the same time, as a producer of phytochemicals. The species of this genus are of American origin but have been cultured and naturalized as ornamentals in various countries (Meerow et al., 1999; Tapia-Campos et al., 2012; Katoch and Singh, 2015). Phytochemical research of the genus began around 1940 with the report of the presence of alkaloids, such as lycorine, and is currently one of the areas of greatest scientific interest in these bulbous plants due to the pharmacological, antimicrobial, antifungal, acetylcholinesterase and cytotoxic properties of their active principles (Greahouse, 1941; Katoch and Singh, 2015). Taxonomically, Zephyranthes belongs to the tribe Hippeastreae (Amaryllidaceae) and its species inhabit tropical and subtropical regions of America (Meerow et al., 2000; Tapia-Campos et al., 2012), and although several efforts have been made to understand its evolutionary complexity, it is still a controversial clade, due to interspecific cross-linked hybridization revealed by molecular data and phylogenetic analyzes (García et al., 2014, 2019).
Cytogenetically, Zephyranthes exhibits a wide range of chromosome numbers ranging from 2n=2x=10 to 2n=96, diploid and polyploid species, polyploid complexes, and the presence of aneuploid-polyploid polymorphisms with varied karyotypic formulas (Raina and Khoshoo, 1971; Bhattacharyya, 1972; Greizerstein and Naranjo, 1987; Daviña and Fernandez, 1989; Daviña, 2001; Felix et al., 2011a; Daviña and Honfi, 2018). Furthermore, there are at least three basic numbers x=5, 6 and 7 whose diploids are found in the subtropical zone of South America (Daviña et al., 2019). Fluorescent chromosome banding techniques allow longitudinal differentiation of chromosomal regions (Honfi et al., 2017). In plants, the specific identification of constitutive heterochromatin regions with a sequential triple staining with chromomycin, distamycin and 4’-6-diamidino-2-phenylindole (CMA/DA/DAPI) (Daviña, 2001) has been used infrequently in the clade Hippeastreae and there are some antecedents in the genera Zephyranthes (Daviña, 2001; Felix et al., 2011b) and Habranthus Herb. (Barros e Silva and Guerra, 2010).
Zephyranthes citrina Baker is a species native to the Gulf of Mexico, described for the first time in 1882, when it was spread to South America and is currently used ornamentally for the aesthetic qualities of its perigonium, particularly for the intense yellow coloration of its tepals (Hume, 1935; Tapia-Campos et al., 2012). In a genus where white and pink shades are the most widespread, the intense yellow tepals are of great interest and value in breeding. Likewise, various phytochemicals have been found in this species, some of them of pharmacological importance (Boit et al., 1957; Herrera et al., 2001; Kohelova et al., 2021). Recently, 27 different alkaloids have been detected in this species, among them, seven were unknown to science. Some of these alkaloids have shown biological activity associated with Alzheimer’s disease and cytotoxic activity linked to oncological diseases (Prakash and Vedanayaki, 2019; Kohelova, 2021; Kohelova et al., 2021). Within the framework of the characterization of the phytogenetic resources of the Amaryllidaceae family of ornamental and phytochemical interest, the objective of this work was to describe the species chromosomally and to detect karyotypic markers that easily identify it.
MATERIALS AND METHODS
Within the framework of scientific cooperation between CIATEJ (Mexico) and UNaM (Argentina), we studied individuals from a population of Zephyranthes citrina (Daviña 681) cultivated in Posadas, Misiones, Argentina, whose control specimen is deposited in the herbarium of the Universidad Nacional de Misiones (MNES) (Figure 1).
Standard cytological techniques
The protocols used by Daviña (2001) were applied and the number of chromosomes in mitotic cells was determined using the meristems of the root tips pretreated with a 0.002M saturated solution of 8-hydroxyquinoline for 8 h at room temperature. They were fixed in absolute ethanol:glacial acetic acid in a 3:1 ratio and stored in the same fixative at about 4° C. The conventional Feulgen
staining was then performed, which consists of an acid hydrolysis of the rootlets in 1N HCl for 10 min at 60° C and a subsequent staining with basic fuchsin (Schiff’s reagent) in a dark chamber for at least 20 min. The meristematic zones were macerated in 2% acetic orcein and subsequently squashed.
Molecular chromosome techniques
Pretreated and fixed roots were used as described above in standard staining techniques and also following the protocol suggested by Schwarzacher et al. (1980), which consists of macerating the root tips in an enzymatic solution (2% cellulase, 1% pectinase, in 0.01 M citrate buffer, pH 4.8) and squashing in 45% acetic acid. The coverslip was removed with liquid nitrogen and air dried for 1 d at room temperature before use.
A triple sequential CMA/DA/DAPI staining was performed. For the CMA (chromomycin A3) bands, the procedure developed by Schweizer (1976) was followed. The slides were incubated in CMA staining solution (McIlvaine buffer pH 7, 10 mM Cl2Mg, 0.12 mg/ml chromomycin A3) for 2 h in the dark at room temperature, washed and air dried, and mounted in a solution 1:1 glycerol:McIlvaine buffer with 5 mM Cl2Mg. Next, they were stained with distamycin A (DA) drops dissolved in McIlvaine buffer pH 7. They were incubated in DA solution at room temperature in the dark in a humid chamber for 15 to 30 min. Subsequently, they were washed and dried. Finally, for the DAPI (4’-6-diamidino-2-phenylindole) bands, the method suggested by Schweizer (1976) was used. The slides were incubated in DAPI staining solution (McIlvaine buffer pH 7, 1-2 μg/ml DAPI) for 30-45 min in the dark at room temperature, washed and air dried, and mounted in the same solution as above.
Karyotype analysis
Chromosomes were observed and photographed with a Leica DML binocular epifluorescence microscope equipped with a DF C310 FX video equipment. 10 optimal metaphases were analyzed, and the nomenclature proposed by Levan et al. (1964) was used to classify chromosomes according to the centromeric index (i=s*100/c, where s=length of the short arm and c=total length of the chromosome). In addition, the total length of the chromosome complement (TCL) and the arm ratio (r=l/s) were calculated. In the idiograms, the chromosomes were grouped according to their morphology and within each group they were ordered by decreasing size. As it is a polyploid species, the idiogram was made considering all the chromosomes. The satellites were classified according to the nomenclature suggested by Battaglia (1955, 1999) with which microsatellites were distinguished from macrosatellites, since the former have diameters less than half the diameter of the chromosome. The value of the length of the satellites was included within the total length of the arm to which they were associated.
RESULTS
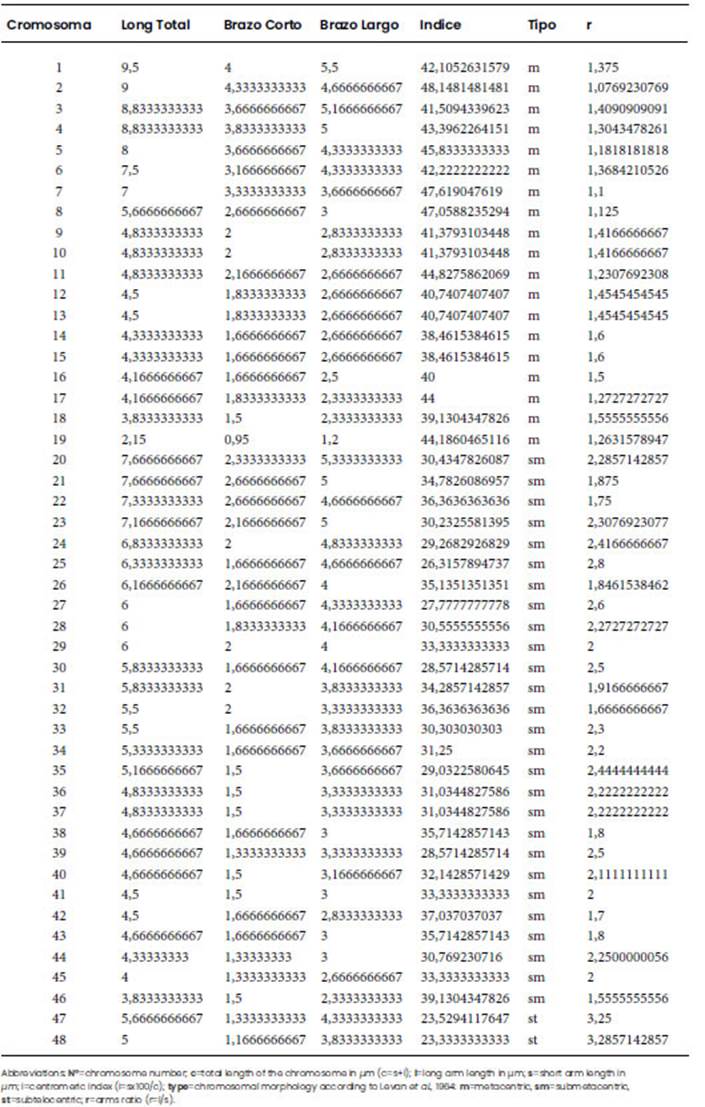
Mitotic metaphases revealed the octoploid condition of Z. citrina with 2n=8x=48 chromosomes and a basic number of x=6 (Figure 2A). The karyotypic formula was 20 m + 26 sm + 2 st, (Figure 3) and the total complement length was 271.31 μm (Table 1). Satellites were observed in the short arm of metacentric chromosomes 8, 9, 11 and 12, all located terminally. In the case of chromosomes 8 and 9 they were macrosatellites, while those of chromosomes 11 and 12 were microsatellites. The mean centromeric index (i) was 36.04 and the mean chromosome length was 5.65 μm. The CMA/DA/DAPI triple fluorescent staining pattern showed constitutive GC (guanine-cytosine)-rich heterochromatin bands (Figure 2B, C). The terminal bands located on the short arm of chromosomes 8 and 9 (m) revealed the presence of a type of constitutive heterochromatin CMA+/DAPI0, whose size includes the satellite and was 1.6 μm. On chromosomes 11 and 12 (m), a CMA+/DAPI- fluorescent band, rich in GC, 0.3 μm long, was identified on the short arm. The amount of constitutive GC-rich heterochromatin corresponded to 0.7% of the polyploid genome. The karyotype comprised chromosome pairs with terminal constitutive heterochromatin bands that included satellites and heteromorphic clusters indicating that it was an allooctoploid (Figure 3).
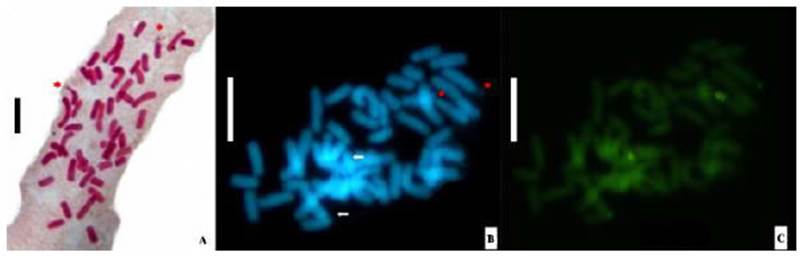
Figure 2 Mitotic metaphase of Z. citrina: A- conventional staining, 2n=8x=48, asterisks indicate the satellites of chromosomes 8 and 9 (m). B-C-Sequential banding CMA/DA/DAPI; arrows indicate sites DAPI - (bands CMA+/DAPIo) on chromosomes 8 and 9 (m); asterisk bands indicate CMA+/DAPI- on metacentric chromosomes 11 and 12 (m). Bars =10 μm.
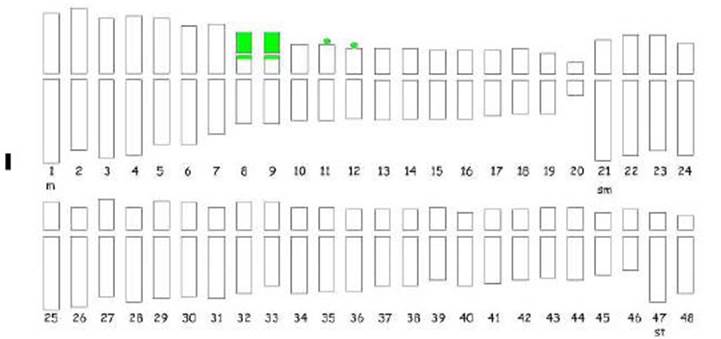
Figure 3 Idiogram of the complete chromosome set of Z. citrina 2n=48 (20 m + 26 sm + 2 st). Heterochromatic bands (in green) CMA+/DAPIo on chromosomes 8, 9, CMA+/DAPI- on chromosomes 11 and 12 (m), in the short arm. Bars =1 μm.
DISCUSSION
Zephyranhtes citrina is an octoploid species that belongs to the group of species with a basic number x=6, which is the most frequent of the genus; this group contains, in addition to diploids, the largest number of polyploid and aneuploid species?. The detected number agrees with those reported by Soontornchainaksang and Chaiyasut (1996), Bobby et al. (2003) and Raina and Khoshoo (1972a) (as Z. sulphurea) and differs from 2n=47 registered by Gonzalez et al. (1980) in provenances from Cuba. This is the first description of constitutive heterochromatin for the species. So far, there are only two antecedents in the Hippeastreae clade on the presence of DAPI+ bands, which correspond to Habranthus robustus Herb., a diploid with 2n=2x=12 (= Zephyranthes robustus, sensuGarcía et al., 2019) and Habranthus brachyandrus (Baker) Seally, a tetraploid with 2n=4x=24 (=Zephyranhtes brachyandra, sensu García et al., 2019) (Barros e Silva and Guerra, 2010; Felix et al., 2011b). The other species of Habranthus and Zephyranthes, with known constitutive heterochromatin patterns, present banding patterns with regions rich in GC (Daviña, 2001; Barros e Silva and Guerra, 2010; Felix et al., 2011b), as well as the type of pattern detected for Z. citrina in this work.
The origin of the polyploids in the Hippeastreae clade remains uncertain in many cases. The main reason is due to the few registered meiotic studies, since both microsporogenesis and megasporogenesis are processes that occur when the flower bud is still inside the bulb without showing external signs of such events and therefore, numerous bulbs must be sacrificed with no guarantees of finding the coveted meiotic stages. Reported male meiosis in both cultivars and natural species have revealed high percentages of bivalents and regular meiosis; multivalent and irregular meiosis to meiotic aberrations such as bridges, lagging chromosomes, micronuclei, among others (Coe, 1953; Sharma and Ghosh, 1954; Tandom and Mathur, 1965; Yokouchi, 1965; Raina and Khoshoo, 1972b; Daviña and Fernandez, 1989; Thoibi Devi and Borua, 1997; Daviña, 2001). For these reasons and based on the described characteristics of the karyotype, Z. citrina is considered to be allopolyploid, which may be clarified with future meiotic studies.
Natural and synthetic hybrids of Zephyranthes have been reported, resulting from intra- and inter-specific crosses, designed to expand options for growers (Raina and Khoshoo, 1972a; Chowdhury and Hubstenberger, 2006; David, 2011). Chowdhury and Hubstenberger (2006) highlight seven barriers to the formation of hybrids in Zephyranthes, among which chromosome number and ploidy level are preponderant due to the existing chromosome variety in the genus. Another crucial aspect is the existence of reproduction by apomixis and pseudogamy in species of the genus, reproductive events that constitute barriers to obtain simple hybrids (Raina and Khoshoo, 1972a; Chowdhury and Hubstenberger, 2006; Crane, 2019). Obtaining hybrids implies the identification of possible progenitors suitable for crossbreeding plans, both to increase aesthetic and ornamental varieties and to obtain new phytochemical combinations.
At least two successful hybrid lineages are known from crosses with Z. citrina. One of them is of interspecific origin and the other is intergeneric. Among the interspecific hybrids with fertile progeny, the tri-hybrid “Best Pink Trihybrid” stands out, product of the cross [(Z. candida x Z. citrina) x Z. macrosiphon] (Chowdhury and Hubstenberger, 2006), where Z. citrina was used as a pollen donor because it is an apomictic species (Howard, 1996). On the other hand, Zephyranthes ajax is a commercial hybrid with pale yellow tepals resulting from a cross between Z. citrina x Z. candida, a somatic chromosome number of 2n=42 and variable ploidy in the endosperm of its progeny (Tandon and Kapoor, 1962). These endosperm characteristics with mitotic aberrations and variable ploidy was also observed in Z. citrina (Bobby et al., 2003). In the lineage of hybrids of intergeneric origin, there is a hybrid known as Cooperanthes “Percy” (also Zephyranthes x Percyi) that was introduced by Traub in 1954 by crossing Z. citrina and Cooperia drummondii Herb. (David, 2011).
Having cytogenetic markers provides a useful tool to detect in early stages if the hybridization was successful, before the first flowering period of the obtained progeny. It is evident that Z. citrina is a species of high value as a parent, of interest in crossbreeding plans due to the qualities of its corolla and the fact that cytological markers for the species have been detected in this work. These results characterize Z. citrina as an octoploid and contribute to the knowledge of its cytogenomic structure. Future crosses using this species as a male parent will allow the initiation of new hybrid lineages of ornamental and/or phytochemical interest, which will be able to multiply massively. Protocols for mass multiplication of bulbs (Rodriguez Mata et al., 2018) and in vitro culture protocols, adjusted to obtain plants without ploidy alteration and with an efficiency of 85% in the acclimatization stage, are available (Syeed et al., 2021).












 uBio
uBio