INTRODUCTION
The tribe Eupatorieae (Asteraceae) includes almost 190 genera and 2200-2500 species with a predominantly Neotropical distribution. Species richness is particularly high in Brazil, tropical Andes and Mexico (King & Robinson, 1987; Hind & Robinson, 2007; Panero & Crozier, 2016; Rivera et al., 2016a; Roque et al., 2017; Gonzáles et al., 2018, Grossi et al., 2020). According to the most recent phylogeny (Rivera et al., 2016b), Eupatorieae have several evolutionary lineages that are taxonomically classified in 20 subtribes, among them Praxelinae with approximately 190 species grouped in seven genera: Chromolaena DC., Eitenia R.M. King & H. Rob., Eupatoriopsis Hieron., Lomatozona Baker, Osmiopsis R.M. King & H. Rob., Praxeliopsis G.M. Barroso, and Praxelis Cass. (King & Robinson, 1987; Hind & Robinson, 2007). This subtribe inhabits from southern North America up to central Argentina and Uruguay. Chromolaena and Praxelis are the southernmost genera, reaching the mountain ranges of the Pampas region (Cabrera, 1941; 1963; Cabrera et al., 1999; Freire & Ariza Espinar, 2014). Species of these genera were, throughout most of the history of the Eupatorieae, classified totally or partially into the large genus Eupatorium L. (Candolle, 1836; Baker, 1876; Robinson, 1923; Cabrera & Vittet, 1954). As a result of a series of studies published in the 1970s and 1980s, this genus was split into ca. 100 genera, which were described as new, raised from infrageneric groups or reinstalled to genus level (King & Robinson, 1987). This taxonomical point of view was completely accepted for the southern South America floras in the early twenty-first century (Troncoso Oliveira, 2010; Freire & Ariza Espinar, 2014).
The South American genus Praxelis, with 19 species, is one of the most widely distributed of the subtribe, ranging from Colombia, Venezuela and the Guianas up to central Argentina and southernmost Brazil (Dillon & Hensold, 1993; Badillo, 2001; Funk et al., 2007; Freire & Ariza Espinar, 2014; Abreu & Esteves, 2017; Ávila et al., 2016; Bernal et al., 2019; Christ & Ritter, 2019). It has not been recorded to this day in Chile, Ecuador, or Uruguay. It is defined by its pappus with numerous scabrid bristles, deciduous phyllaries, capitula with conical receptacles and asymmetrical carpopodium. These structures have been recognized as important evolutionary characters in Eupatorieae (Robinson et al., 2009; Grossi et al., 2020).
Knowledge about the flora of southern South America varies according to the country or taxonomic group considered. In the case of Uruguay, several efforts have been made in recent years to catalogue the biodiversity of Asteraceae (Sancho et al., 2006; Hurrell, 2013; Trujillo et al., 2014; Valtierra & Bonifacino, 2014) and particularly of Eupatorieae (Pérez Sobrino, 2016; Rodríguez-Cravero et al., 2017; 2019). Currently, Praxelinae is represented in Uruguay by six species of Chromolaena: C. caaguazuensis (Hieron.) R.M. King & H. Rob., C. hirsuta (Hook. & Arn.) R.M. King & H. Rob., C. ivifolia (L.) R.M. King & H. Rob, C. laevigata (Lam.) R.M. King & H. Rob., C. squarrulosa (Hook. & Arn.) R.M. King & H. Rob., and C. verbenacea (DC.) R.M. King & H. Rob. (Freire, 2008; Flora del Cono Sur, 2020). During the revision of species of Praxelis in southern South America, populations of P. clematidea R.M. King & H. Rob. were recorded in Uruguay. The goals of this work are to report the genus and the species for the Uruguayan flora for the first time and to establish its differences with morphologically similar species within Praxelinae in Uruguay and surrounding areas of Argentina and Brazil.
MATERIAL AND METHODS
We analyzed specimens and photographs from the following herbaria: BA, BAA, CORD, ECT, LIL, LP, MBM, MVJB, MVFA, MVM, and SI (Thiers, 2020). Descriptions are based on herbarium specimens and field observations. Several field trips were made throughout Uruguay and eastern and northeastern Argentina, during the years 2017, 2019, and 2020. Specimens collected were deposited in BA, LP, MVFA and/or MVM.
A detailed analysis of the species was carried out considering macro and micromorphological characters, especially those previously used for delimiting genera of Praxelinae and species of Praxelis: leaf, involucre, phyllary, cypsela, pappus, and trichomes (King & Robinson, 1987; Grossi et al., 2020). Leaves and capitula were rehydrated, treated with a clearing process and stained with 2% safranin (Zarlavsky, 2014). Samples were examined with a Nikon Eclipse E200 microscope. For scanning electron microscopy (SEM) studies, dry materials (cypsela, pappus and receptacle) were placed directly on the stubs, coated with palladium/ gold, and observed in a Philips XL-30SEM at the Argentinian Museum of Natural Sciences.
The species was illustrated by M. Alejandra Migoya (FCAyF, UNLP), with sketches of the capitulum and phyllaries made by Diego G. Gutiérrez.
RESULTS
Praxelis clematidea R.M. King & H. Rob., Phytologia 20: 194. 1970. Eupatorium urticifolium L. f. var. clematideum Hieron. ex Kuntze, Revis. Gen. Pl. 3: 148. 1898, nom. illeg. Eupatorium urticifolium L. f. var. clematideum Chodat, Bull. Herb. Boissier 2: 711. 1903, nom. illeg. Eupatorium catarium Veldkamp, Gard. Bull. Singapore 51: 121. 1999, nov. nom. Eupatorium clematideum Griseb., Abh. Königl. Ges. Wiss. Göttingen 19: 172. 1879, nom. illeg., not (Wall. ex DC.) Sch. Bip. TYPE: Argentina, Córdoba, “pr. Córdoba”, P. G. Lorentz 81 (lectotype GOET 0001494 [photo!], designated by Freire & Ariza Espinar, Flora Argentina 7 (1): 404. 2014); isolectotype GOET 001493 [photo!]). Figs. 1 and 2.
Annual or perennial herbs, or rarely subshrubs, somewhat woody at base, up to 0.5-1 m tall. Stems erect or ascendant, generally laxly foliose, cylindrical, striated hirsute, rarely glabrescent. Leaves opposite, petiolate, petioles up to 2 cm long, hirsute; leaf blades 2-7 x 1-5 cm, ovate or ovate- rhombic, apex acute, basis attenuate or cuneate, margins serrate, membranous, adaxial surfaces hirsute with uniseriate simple hairs, abaxial surfaces densely hirsute with uniseriate simple hairs and glandular hairs; actinodromous. Capitulescence of densely many-headed corymbiform cymes. Capitula subsessile, very short peduncles up to 0.7 cm long, hirsute with uniseriate simple hairs. Involucres 5.5-9 x 3.5-9 mm, campanulate, phyllaries imbricate 18-24, in 3-5 series, increasing gradually in length, outers 3.6-9 x 0.4-2 mm, ovate, apices strongly acuminate, margins entire, pubescent mainly at the apex, inners 5-8 x 0.8-2 mm, narrowly oblong, apices acute or acuminate, sometimes attenuate, margins entire, glabrescent, all 3-5-nerved dorsally and deciduous. Receptacles highly conical, glabrous. Florets 30-50, bisexual, corollas 3-4.5 x 0.4-0.7 mm, tubular-funnelform, lilac, whitish at maturity, shortly 5-lobed, lobes 0.3-0.7 mm long, glandular or eglandular, papillose on inner surface; styles 4-7.5 mm long, basis not enlarged, glabrous, style branches 1.5-4 mm long, linear, densely long-papillose; stamens with anther apical appendages ovate, collars balusterform, basis rounded. Cypselae 1.7-2.4 x 0.4-0.8 mm, obovate, obcompressed, 2-4-ribbed, setuliferous mainly on the ribs, carpopodium distinct, asymmetrical, laterally inserted in the receptacle. Pappus of 16-30 bristles, 2-4 mm long, scabrid persistent white.
Common names. Two common names have been reported in Argentina: “carapari” (Novara, 2009) and “langosta” (English locust; Cantero et al., 2019) but they seem to be scarcely used. In China, it is known as 假臭草: Jia Chou Cao (eFloras, 2020).
Distribution and habitat. Praxelis clematidea (Figs. 1 and 2) is a widely spread native species in South America inhabiting Argentina, Bolivia, Brazil, Paraguay, and Peru (Dillon & Hensold, 1993; Cabrera et al., 1996; Freire, 2008; Troncoso Oliveira, 2010; Robinson, 2014; Abreu, 2015; Ávila et al., 2016), reaching with this new record northwestern Uruguay, in the departments of Artigas and Salto (Fig. 3). It occurs in subtropical forests and rainforests, shrublands and grasslands. In Uruguay, it is found on roadsides as a ruderal plant (Fig. 2B) in the Cuenca Sedimentaria del Oeste ecoregion (Brazeiro, 2015). In its native range, P. clematidea is commonly found on roadsides and as a weed in cultivated areas (Marzocca, 1976; De Egea et al., 2016).
Observations. Within Praxelinae, Chromolaena and Praxelis are the two southernmost distributed genera of the subtribe. They share several morphological characters like numerous bristles of pappus with long equal or subequal elements and cypselae with usually more than two ribs (Fig. 4 A-B). However, Praxelis is morphologically well circumscribed by its 3-5-seriate and campanulate involucre, highly conical receptacles and cypselae with asymmetrical carpopodium laterally inserted to the receptacle (Fig. 4 C-D).
Praxelis clematidea is a plant of ethnobotanical interest (Barboza et al., 2006) with antibacterial effect against both Gram + and Gram - bacteria, antimycotic effect (Filho et al., 2013; 2016; Li et al., 2017), anti-neuroinflammatory agent (Xiao et al., 2020) and significant gastroprotective activity (Falcão et al., 2013). Other possible applications include phytoremediation (Fu et al., 2017; Wei et al., 2018).
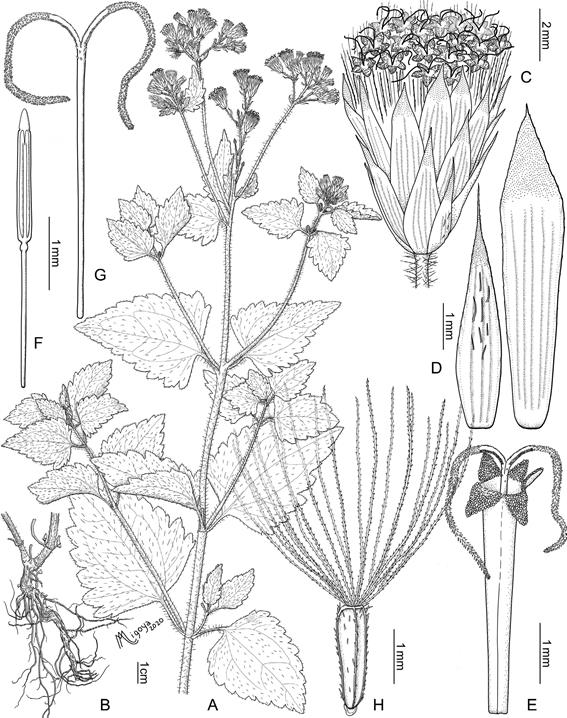
Fig. 1 Praxelis clematidea. A, plant. B, detail of basal stems and roots. C, capitulum. D, phyllaries (outer on the left, inner on the right). E, floret without cypsela and pappus. F, stamen. G, style. H, cypsela with pappus. Illustration: A. Migoya. Material from Gutiérrez 1707 (BA).

Fig. 2 Praxelis clematidea. A, plant in its habitat. B, population. C, leaf. D, capitulescence. E, capitulum in superficial view. F, mature capitula showing the pappus. Photos by V. G. Salgado and D. G. Gutiérrez. Color version at http://www.ojs.darwin.edu.ar/index.php/darwiniana/article/view/912/1193

Fig. 3 Distribution of Praxelis clematidea in Uruguay and surrounding areas of Argentina and Brazil.
Specimens examined
ARGENTINA. Corrientes. Depto. Empedrado, Ruta Nacional 12 y río San Lorenzo, 27-III-1976, Schinini 12833 (SI). Depto. General Paz, General Paz, I-1966, Krapovickas 11830 (LP). Depto. Goya, 28-I-1971, Krapovickas 17827 (LP); Ruta Nacional 12, puente sobre el río Corrientes, 25 km N de Esquina, 20-II-2020, Gutiérrez et al. 1605 (BA). Depto. Itatí, Ruta Nacional 12, 73 km E de Corrientes, 21-II-2020, Gutiérrez et al. 1619 (BA). Depto. Ituzaingó, Ituzaingó, 5 km de Ruta Nacional 12, 5-II-1976, Romanczuk 789 (SI). Depto. Paso de los Libres, Parada Pucheta, 22-XII-1946, Huidobro 3857 (LIL). Depto. San Martín, Ruta Provincial 114, 20 km O de La Cruz, camino a Colonia Pellegrini, 25-II-2020, Gutiérrez et al. 1677 (BA). Depto. San Cosme, Isla Toledo, Río Paraná, 25-VII-1944, Ybarrola s.n. (LIL). Depto. San Miguel, Ruta Provincial 118, 10 km N de San Miguel, 21-II-2020, Gutiérrez et al. 1628 (BA). Depto. San Roque, Chavarria, cercanías, 28-III-1945, Ybarrola 2786 (LIL). Depto. Santo Tomé, Santo Tomé, 24-II-2020, Gutiérrez et al. 1675 (BA). Entre Ríos. Depto. Federación, Colonia Flores, II-1949, Martínez Crovetto 4986 (SI). Misiones. Depto. Concepción, 8 km E de Concepción de la Sierra, 24-II-2020, Gutiérrez et al. 1661 (BA). Depto. Eldorado, Puerto de Eldorado, 17-IV-1979, Gómez 91111 (SI).
BRASIL. Rio Grande do Sul. Capão da Canoa, Capão Novo, 7-III-1997, Ribas 1848 (MBM); Osório, Trevo entre as rodovias Estrada do Mar e RS030, 18- IV-2015, Gonzatti & Valduga 1769 (ECT).
URUGUAY. Artigas. Margen de la Ruta 3, km 613, 800 m s.m., Arroyo Itacumbú, 13-I-2020
González s.n. (MVM 23485). Salto. Ruta 3, 6 km O de Monteclaro y 12 km NO de Palomas, 27-II-2020, Gutiérrez et al. 1707 (BA, MVM); Ruta 3, 7 km O de Belén, 27-II-2020, Gutiérrez et al. 1701 (BA, MVM).

Fig. 4 Reproductive microcharacters in Praxelis. A, bristles of pappus and cypsela. B, part of a scabrid bristle of the pappus. C, conical receptacle and upper part of peduncle. D, lower part of a cypsela showing the asymmetrical carpopodium and lateral insertion of the fruit. Material from Gutierrez et al. 1508 (BA).
Key to species of Praxelis in Uruguay and surrounding areas of Argentina and Brazil
1. Solitary capitulum, long pedunculate, or capitula in scapiform terminal branches ................................................... 2
1. Capitula grouped in corymbiform cymes .................................................................................................................. 3
2(1). Plants unbranched, hirsute, leaves semi-rosulate, phyllaries pubescent ............................................... P. missiona
2. Plants branched, glabrous or glabrescent, leaves opposite, phyllaries glabrous ..................................... P. kleinioides
3(1). Herbs up to 0.35 m tall, laxly pubescent, cymes lax, phyllaries 3-nerved ................................................ P. ostenii
3. Herbs 0.5-1 m tall, densely pubescent, cymes denses; phylllaries 3-5-nerved ....................................... P. clematidea
DISCUSSION AND CONCLUSIONS
Currently, P. clematidea is considered a high risk invader (USDA, 2014). At the end of the 20th century, it was detected as an invasive plant almost simultaneously in Hong Kong and Australia (Corlett & Shaw, 1995; Waterhouse, 2003). In the past twenty years, it has spread through China and other Asian countries such as Indonesia, Singapore, Taiwan and Thailand (Wang et al., 2006; Wu et al., 2010; Changtragoon et al., 2012; Chen et al., 2018; Tjitrosoedirdjo & Wahyuni, 2018) and reached North America in southeastern United States of America (Abbott et al., 2008). In general, its presence was detected only after it was well-established, because of its morphological similarity with other weeds of Eupatorieae such as Chromolaena odorata (L.) R.M. King & H. Rob. and Ageratum conyzoides L. (Waterhouse, 2003; USDA, 2014; Chen et al., 2018). The species has apparently been eradicated in the Republic of Palau, an island located in the western Pacific Ocean, where a few plants were found in isolated patches in 2007 (Space et al., 2008). Even though P. clematidea is often found invading natural environments like woodlands, it shows preference for disturbed areas such as roadsides (Chen et al., 2007; USDA, 2014), similar to the habitats where it was found in northwestern Uruguay and other areas of its original distribution.
With the addition of P. clematidea, the tribe Eupatorieae is now represented by 21 genera and 53 species in Uruguay (Rodríguez-Cravero et al., 2019; Flora del Cono Sur, 2020). The absence of this species in the Uruguayan floras of beginnings of the 1900s (Arechavaleta, 1906; Herter, 1930), later studies on the tribe (King & Robinson, 1987) and regional catalogues or floras (Freire, 2008; Flora del Cono Sur, 2020), in addition to its invasive behavior, might indicate that its presence is relatively recent in the country. However, its Argentinian occurrences near the Uruguayan border and wind dispersed fruit, the absence of a comprehensive flora for the country, and morphological similarities between P. clematidea and other species (that caused it to be unnoticed in early stages of invasion all over the World), raises the possibility that it was, until now, overlooked. To date, the available information is not conclusive and phylogeographic studies, for example, could be helpful to understand its evolutionary history in this area.












 uBio
uBio 

