INTRODUCTION
Mazaceae is a small family native to Asia and Oceania, with ca. 34 species (Deng et al., 2019). It was proposed within the Lamiales (Reveal, 2011) and was separated from Phrymaceae based on several molecular phylogenetic studies (Albach et al., 2005; Xia et al., 2009; Schäferhoff et al., 2010). Mazaceae include four genera, Mazus Loureiro, Lancea Hook.f. & Thomson, Dodartia L. and the recently described Puchiumazus Bo Li, D.G. Zhang & C.L. Xiang (Xiang et al., 2021).
Mazus, with ca. 30 species, is the richest genus of the family (Deng et al., 2019). It is distributed in eastern and southeastern Asia, Australia, and New Zealand (Barker, 1991; Hong et al., 1998). China is considered the center of diversification of the genus (Yang, 1979; Hsieh, 2000), with ca. 26 species and three varieties currently recognized (Hong et al., 1998; Deng et al., 2016). This genus is characterized by its herbaceous habit, flowers with zygomorphic corollas, with one 2-lobed upper lip and one 3-lobed lower lip, and fruit usually completely enclosed in the calyx when mature (Xiang et al., 2021).
Mazus pumilus (Burm.f.) Steenis is a species native to South and East Asia (Cao Shu, 1998), and has become established in America and Europe (Kew, 2021a). It grows in wet grasslands, along streams, trailsides, waste fields, wet places, and the edges of forests (Shahid et al., 2013). In Costa Rica and the United States, it has been reported as a naturalized species that appears in urban environments (Nishida et al., 2009; Pringle, 2018 Morales, 2020).
Nowadays, there is a homogenization of the world flora due to the cultivation and the accidentally introduction of foreign species (Mooney & Hobbs, 2000; Cárdenas et al., 2017). Some of these become invasive and cause problems related to species and population declines, habitat degradation, loss of the ecosystem functions, among others (Vitousek et al., 1997). In Colombia, there are reported 1046 exotic species (Bernal et al., 2016) of which only 35 are recognized as invasive (Cárdenas et al., 2017). Recording the exotic species is the first step for making analysis about their invasiveness potential and structuring management plans.
The synonyms of M. pumilus has been matter of controversy. Aiton (1812), Sweet (1825), Sweet (1826), Maximowicz (1875), Trautvetter (1884), and Hayata (1908) treated M. pumilus as Mazus rugosus Lour.; Aiton (1812) just cites as synonym to Lindernia japonica Thunb.; Sweet (1825) to L. japonica, Hornemannia bicolor Willd. and Gratiola goodenifolia Hornem. (sic); Sweet (1826) to L. japonica, Titmannia obovata Bunge and H. bicolor; Maximowicz (1875) to M. vandellioides Hance (sic), Vandellia obovata Walp. (sic), T. obovata and L. japonica; Trautvetter (1884) to L. japonica, Columnea tomentosa Roxb.? (sic), Stemodia tomentosa G. Don? (sic), H. bicolor, G. goodeniaefolia (sic), Trevirania gratiolae Roth., M. vandelliodes (sic), T. obovata and V. obovata; Hayata (1908) to M. vandellioides (sic), L. japonica and M. japonica (sic). Makino (1901), Matsumura (1912), Ohwi (1965), and Fu-Wu & Yue-Hong (2006) treated M. pumilus as M. japonicus; Makino (1901) cites as synonyms to M. rugosus, T. obovata, V. obovata, M. vandellioides (sic), H. bicolor, M. bicolor, G. goodeniaefolia (sic), C. tomentosa and S. tomentosa; Matsumura (1912) to M. rugosus; Ohwi (1965) is the only one who cites Lindernia japonica pro parte for M. japonicus and pro parte for M. miquelii Makino, and as synonyms to M. rugosus and T. obovata; finally Fu-Wu & Yue-Hong (2006) just cites to M. rugosus and L. japonica. None of these authors cited the location of the types or the reasons for which they included or not some synonyms. Steenis (1958) combined Lobelia pumila Burm.f. in Mazus pumilus (Burm.f.) Steenis, and mentioned that the synonyms of M. pumilus known until the date are L. japonica, M. rugosus, M. laevifolius, and M. vandellioides (sic); he only cited the ubication of the type for M. laevifolius. Cramer (1981) cited two synonyms for M. pumilus, L. japonica and M. rugosus, and mentioned that the types of these two names are unknown; Cheng-Yih (1984) cited L. japonica, M. rugosus, H. bicolor, T. obovata, V. obovata, M. vandellioides (sic), M. japonicus, M. bodinieri Bonati pro parte for M. pumilus and pro parte for M. spicatus Vaniot, and Lobelia esquiroli H. Lév. (sic); Smith (1991) to L. japonica and M. japonicus; Cao Shu (1998) to H. bicolor, L. japonica, M. rugosus, M. vandellioides, T. obovata, and V. obovata; Hsieh (2000) to M. rugosus and L. japonica, and Moreira & Bove (2008) to M. japonicus. None mentioned the types or their ubication or why they included or not some synonyms. In this way, there is no clarity which names are synonyms of M. pumilus, in which herbaria are deposited the types or if the types are lost or destroyed.
Herein, the family Mazaceae is reported for the first time for Colombia, with M. pumilus. Here we contribute to the knowledge of the Colombian exotic flora and with the clarification of the synonyms of M. pumilus and the ubication of their types.
MATERIALS AND METHODS
We made free tours in Bogotá D.C., between January and February of 2021 and collected the individuals in reproductive stage. The herbarium vouchers were deposited in UDBC (herbarium acronyms follow Thiers, 2021). To determine the generic identity of the species, we consulted the pertinent literature on the taxonomy of the Lamiales (Cao Shu, 1998; Deng et al., 2019; Kew, 2021b) and the species of this order reported for Colombia (Bernal et al., 2016). For the circumscription of Mazus, we followed Xiang et al. (2021).
For the elaboration of the description, we measured the organs with a digital caliper with an accuracy of 0.01 mm. For the review of the synonyms of M. pumilus we consulted in Biodiversity Heritage Library, Schoolar Google, Web of Sciencie and Scielo, the publications who included taxonomical treatments or mentioned names related with Mazus pumilus, then we analyzed the protologues of these names, available at Biodiversity Heritage Library (www.biodiversitylibrary.org), and saw the types in JSTORPLANTS (https://plants.jstor.org/). For the names that did not mention the type, we searched in which herbaria the authors deposited their types (Stafleu & Cowan, 1979) and asking to the curators to search exsiccata with collected by names author or with a label of the name. Finally, for the elaboration of the map, we use the records from our free tours and those of iNaturalist (Naturalista, 2021).
RESULTS AND DISCUSSION
Mazus pumilus (Burm.f.) Steenis, Nova Guinea 9 (1): 31. 1958. Basionym: Lobelia pumila Burm. f., Fl. Indica 187, pl. 60, f. 3. 1789. TYPE: Burman, Fl. Indica 187 Ic. pl. 60 f.3 (Lectotype designated by Cramer, 1981). Figs. 1, 2.
Mazus rugosus Lour., Fl. Cochinch. 385. 1790. TYPE: China, Cochinchina, J. Loureiro s.n. (holotype: BM-000997856!).
Trevirania gratiolae Roth, in Weber, Beitr. II. 123. 1810. TYPE: without data (holotype: M-0188280!).
Mazus laevifoliusBlume, Bijdr. Fl. Ned. Ind. 14: 753. 1826. TYPE: Indonesia, Java, Buitenzorg, w.d., w.c. (holotype: L-0003532!).

Fig. 1 Mazus pumilus. A, habit. B, root. C, lateral shoots. D, variation in the leaf morphology. E, upper leaf surface. F, lower leaf surface. Photographs: Andrés Fonseca-Cortés. Color version at http://www.ojs.darwin.edu.ar/index.php/ darwiniana/article/view/949/1224

Fig. 2 Mazus pumilus. A, inflorescence. B, flower in frontal view. C, flower side view. D, stamens, style and stigma. E, fruits. Photographs: Andrés Fonseca-Cortés. Color version at http://www.ojs.darwin.edu.ar/index.php/darwiniana/ article/view/949/1224
Mazus bicolor (Willd.) Benth., Numer. List. 3913. 1831. Basionym: Hornemannia bicolor Willd., Enum. Pl. [Willdenow] 2: 653 1809. TYPE: without data (holotype: B -W-11582 -01 0!).
Mazus vandellioides Hance, Ann. Bot. Syst. 3(2): 193. 1852. TYPE: China, Hong Kong, w.d. Hance s.n. (holotype: FI-063202).
Lobelia esquirolii H. Lév., Fl. Kouy-Tcheou: 58. 1914. TYPE: China, Kouy-Tcheou, mont du College, IV-1910, J. Esquirol 2062 (holotype: E-00284110!).
Mazus goodeniifolius (Hornem.) Pennell, J. Arnold Arbor. 24: 245. 1943. Basionym: Gratiola goodeniifolia Hornem., Enum. Pl. Hort. Hafn.19. 1807. TYPE: without data (holotype: C-10019014!).
Prostrate herbs, with one long tap root or with numerous roots, and one to many shoots. Leaves 0.4-2.4 × 0.4-0.8 cm, basal leaves usually rosulate, cauline leaves alternate; glabrous or with a few trichomes on the margin at the base, elliptic to obovate, base decurrent in the petiole, apex rounded, margin entire, crenulated or with a few teeth, pinnately nerved with 2-4 pairs of secondary veins. Inflorescence 2.5-6.0 cm long; peduncles of 0.5-0.7 cm long, terminal, racemose, glabrous or puberulous. Flowers 0.5-0.8 × 0.4-0.6 cm, bilabiate, calyx 0.4-0.5 × 0.2-0.3 cm, tube 0.1-0.2 cm long, glabrous or puberulent, green, with five lanceolate sepals, 0.2-0.3 × 0.1-0.2 cm, base of the sepals with a red gland; corolla gamopetalous, lilac outside, pale lavender inside with yellow macules, upper petals fused into an emarginate lip, lower petals fused into a trilobate lip, terminal lobe 0.1-0.2 × 0.1-0.2 cm, lateral lobes 0.3-0.4 × 0.2-0.3 cm, hairy, with two longitudinal ridges; stamens four, didynamous, filaments 0.2-0.3 cm long, anthers medifixed ca. 0.1 cm long; ovary superior, glabrous, bilocular; style 0.4-0.5 cm long; stigma flabelliform. Fruit a loculicidal capsule with numerous seeds.
Distribution. Mazus pumilus is native of Eastern Asia and Oceania (Cao Shu, 1998), and it has spread in many countries of America and Europe (Kew, 2021a). In Colombia (Fig. 3), this species has been collected in Bogotá D.C., and has been recorded in Medellín, Antioquia and in Granada municipality, Meta (Naturalista, 2021), growing between the pavement.
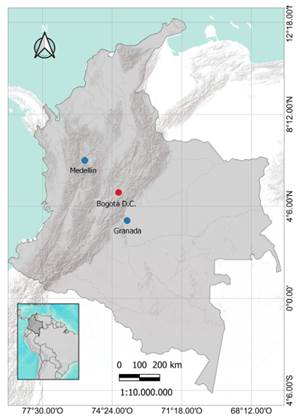
Fig. 3 Records of Mazus pumilus in Colombia, the red point correspond where this species were collected for the first time, the blue points were taken from iNaturalist (Naturalista, 2021). Map: Jairo A. Peña-Torres.
Etymology. Mazus from the Greek, “μαστός”, breast, alluding to the two ridges on the lower lip of corolla and pumilus from the Latin “pūmilus” that means dwarf, alluding to the small size of this species.
Uses. Pharmacological studies show that M. pumilus is a promising species for medicinal use and treatment, Priya & Rao (2016) report anticancer and antioxidant activity of various leaf extracts of M. pumilus. This taxon also possesses antibacterial and antifungal properties (Safdar et al., 2017). Ishtiaq et al. (2019) demonstrated the anti-nociceptive, anti-inflammatory and hepatoprotective effects of the methanol extract of M. pumilus. Additionally, this species has wide medicinal uses in the local and popular tradition.
In India, mainly in the Jammu and Kashmir states, indigenous local communities use the leaves of M. pumilus to treat epilepsy (Sharma et al., 2013). In urban regions of India, the whole herb is utilized as an antifebrile, emmenagogue and aperitive (Mishra et al., 2015) and the infusion is consumed as tonic (Vardhana, 2008).
Examined material
COLOMBIA. Cundinamarca. Bogotá D.C., Engativá, Unicentro de Occidente, 4°43’22.1”N 74°06’ 5.3” W, 2600 m, 27- I-2021, A. Fonseca-Cortés 1338 (UDBC); Bogotá D.C., Engativá, Ciudadela Colsubsidio, 4°43’14.4” N 74°06’57.0” W, 2600 m, 24-II-2021, A. Fonseca-Cortés & J. Peña-Torres 1450 (UDBC); Barrios Unidos, Museo de Los Niños, 4°39’42.38” N 74°05’18.65” W, 2600 m, 24-II-2021, A. Fonseca-Cortés & J. Peña-Torres 1451 (UDBC); Bogotá D.C., Barrios Unidos, Parque del Salitre, 4°39’58.93” N 74°05’19.35” W, 2600 m, 24-II-2021, A. Fonseca-Cortés & J. Peña-Torres 1452 (UDBC).
COMMENTS
Burman (1768) in his description of Lobelia pumila just cites an illustration (Tab 60 f.3) present in the same publication, and he does not mention any exsiccata. Steenis (1958) mentions that the type of Lobelia pumila is at G. There are two exsiccata at G of the collection of Burman, G-00096392 and G-00096393, the former has a label of “TYPE” and the latter of “TYPE DUPLICATE”. However, there is no clarity if that labels were put by Steenis or not. Cramer (1981) states the illustration mentioned by Burman (1768) as the type. Therefore, Stennis (1958) did not lectotypified L. pumila (article 7.11, Turland et al., 2018), but Cramer (1981) did (article 9.12, Turland et al., 2018).
After the search in the herbaria, we found all the types of the synonyms listed in the introduction except for Tittmannia obovata, for which we cannot contact the curator of the herbarium in which it is probably deposited (LE) (Stafleu & Cowan, 1979).

Fig. 4 Genus and species morphologically similar to M. pumilus. A, Mazus pumilus. B, Cymbalaria muralis. C, Nuttallanthus sp. Color version at http://www.ojs.darwin.edu.ar/index.php/darwiniana/article/view/949/1224
The analysis of the types and of the protologues allow us to confirm that the names cited in the different treatments listed in the introduction, with the exception of Columnea tomentosa, Stemodia tomentosa, Lindernia japonica, Mazus bodinieri and Tittmannia obovata, are synonyms of M. pumilus. The holotype of M. laevifolius is at L (L-0003532); the holotype of M. rugosus is at BM (BM-000997856); the holotype M. vandellioides is at FI (FI-063202); the holotype of Hornemannia bicolor is at B (B -W-11582 -010), the holotype of Lobelia esquirolii is at E (E-00284110), the holotype of Trevirania gratiolae is at M (M-0188280!) and the holotype of Gratiola goodenifolia is at C (C-10019014).
There are two collections at UPS collected by Thunberg and determined as L. japonica by him, UPS-14334 (which has written Lindernia japonica α) and corresponds to M. miquelii Makino, and UPS-14335 (which has written Lindernia japonica β) and corresponds to M. pumilus. Of the treatments of Mazus, only Ohwi (1965) treats the problem and mention L. japonica pro parte for M. miquelii and L. japonica pro parte for M. japonicus. However, none has purposely or not tried to lectotypified this name. The original description of L. japonica does not cite the α or β, but mentions “ramis… erectiusculis… pollicaribus usque spithamaeis” (Thunberg, 1784); which means, branches… erect... of one to seven inches, this length is only present in M. miquelii.
In this sense, here We propose UPS-14334 as the lectotype for L. japonica, due to their description fits better with M. miquelii.
When Bunge (1831) described Tittmannia obovata, mentioned “subssesilibus, glandulosus- pubescentibus”, M. pumilus does not present glandular indumentum (Pringle, 2018). Additionally, we can’t analyze the type of this name. Therefore, we could not verify this name as a synonym of M. pumilus.
Walpers (1844) mentions “V. (TITTMANNIA Bnge) OBOVATA Wlprs. Mss.”. Maximowicz (1875), Cao Shu (1998), IPNI (2021) and Tropicos (2021) cite this name as Vandellia obovata Walp. However, Walpers (1844) did a combination for the name Tittmannia obovata Bunge. For this reason, this name should be written as Vandellia obovata (Bunge) Walp., and not as Vandellia obovata Walp., following the article 41.1 (Turland, et al., 2021).
Although Hornemann (1807) wrote Gratiola goodenifolia and Blume (1826) wrote Mazus laevifolia, these names should be written as Gratiola goodeniifolia and Mazus laevifolius following the article 60.10 and 23.5 respectively (Turnland et al., 2018).
Despite Cheng-Yih (1984) listed to M. bodinieriBonati pro parte as a synonym of M. pumilus, the analysis of the syntypes listed by Bonati (1908) and disponible at JSTORPLANTS (P. bodinieri, 1593, is not present in this plataform neither in the virtual collections of P), shows that this name is actually a synonym of M. spicatus, as reported by Cao Shu (1998). As no author has designated a lectotype for M. bodinieri, here we propose to H. Wilson 931 (E-00284117), following the article 9.12 (Turland et al., 2018).
The holotype of C. tomentosa (LINN- HS1102-6) presents bigger plants, wider leaves and serrate margins, characters not seen in M. pumilus, for this reason we do not include it in the synonymy.
Mazus pumilus could be confused with Cymbalaria muralis G. Gaertn., B. Mey. & Schreb (Plantaginaceae) (Fig. 4b), and the genus Nuttallanthus D.A. Sutton (Plantaginaceae) (Fig. 4c), but it is clearly distinguished from these by the characters listed in the Table 1.
In Bogotá there are many plant nurseries, which usually import species from other countries. These plants come with soil, which usually carries seeds of other foreign species (Cárdenas et al., 2017). Due to the lack of control, many exotic species have gotten in Colombia (Cárdenas et al., 2017). In this way, M. pumilus probably arrived with imported species, and then become established in the urban zones, as reported in Costa Rica (Nishida et al., 2009; Morales, 2020) and the United States (Pringle, 2018).
Mazus pumilus has been reported as an annual species (Barker, 1991; Shahid et al., 2013; Pringle, 2018). However, we saw great patches covered by this species. Although its inflorescence is terminal, this species can produce many lateral shoots (Fig. 1c) and two or more inflorescences (Fig. 2a), which fits with the concept of perennial species (Albani & Coupland, 2010). In this sense, M. pumilus can be an annual or perennial species, and the expression of one of these habits is probably related with the environmental conditions, as reported in Mimulus gutattus DC. (Phrymaceae) (Baker & Diggle, 2011).












 uBio
uBio 


