Nikolai Kulchitsky first identified the enterochromaffin cell in 1897 in the intestinal mucosa of cats and dogs, nevertheless lung neuroendocrine cells (LNC) were first discovered by Feyrter in 19381,2. Normal lung tissue contains neuroendocrine cells along the bronchial and bronchiolar epitelium. LNC can be divided into isolated cells along the tracheal mucosa, accounting for 0.41% of the epithelial cells or clusters, also called neuroendocrine bodies, composed of 4 to 10 cells3.
Lung tissue inflammation, injury and exposure to toxic products are associated with reactive hyperplasia of LNC with subsequent regenerative repair, fibrosis and chronic inflammation. However, LNC hyperplasia can occur with out underlying disease. This proliferation of neuroendo crine cells is the so called phenomenon diffuse idiopathic pulmonary neuroendocrine cell hyperplasia (DIPNECH). DIPNECH is a rare idiopathic disease, associated with occlusive bronchitis, first reported by Aguayo in 19924.
In the World Health Organization classification of tu mors of the lungs, published in 2015, all neuroendocrine tumors are grouped together in the same category. Nev ertheless, high grade small cell lung cancer and large cell neuroendocrine carcinoma are differentiated from atypical carcinoids, low grade typical carcinoids and DIPNECH5.
A 5.7% of patients with carcinoids have DIPNECH as a comorbidity and 47.4% of patients with DIPNECH have typical carcinoids, whereas 15.8% have atypical carci noids, suggesting that DIPNECH develops into a tumorlet and eventually a carcinoid tumor. Nowadays DIPNECH is considered a pre-invasive lesion6.
Case report
A 64-year-old woman with smoking history of 20 years dis covered a painless nodule in her right axillary region. Axillary ultrasound revealed enlarged lymph nodes with irregular margins and loss of the usual cortico-medullary differentiation.
Mammography and mammary ultrasound showed grouped coarse heterogeneous calcifications on the upper outer quad rant of the right breast and two poorly circumscribed and defined nodular formations with deposits of calcium, respectively. The patient underwent a core biopsy of the breast nodule. The histopathological result was concordant with breast invasive ductal carcinoma of no special type (NST), negative for hormonal receptors (estrogen and progesterone) and HER2 positive, Ki67 of 40%.
A thoracoabdominal staging CT showed a 2.27 cm part-solid nodule (solid part < 5mm) with air bronchogram in the upper segment of the superior right lobe and a soft tissue formation in the upper outer quadrant of the right breast of 2.3 × 3cm and a 2 × 1.7cm axillary lymph node (Fig. 1).
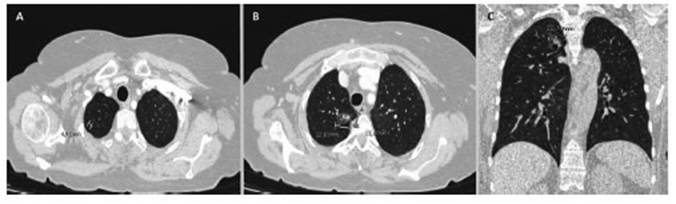
Fig. 1 Pre-operative Chest CT. A: Axial view. 0.46cm ground glass nodule in the apical segment of the right upper lobe. B: Axial view. 2.27 cm subsolid nodular opacity with air bronchogram in the upper segment of the superior right lobe. C: Coronal view
Breast cancer metastases tend to be solid, well circum scribed, rounded lesions, more often in the periphery of the lung. Because of the imagenological characteristics (ground glass opacity), the lung nodule was interpreted as inflamma tory vs. a primary lung cancer (adenocarcinoma). The breast cancer was staged as T2cN2cM0 according to the TNM sys tem. The patient underwent neoadjuvant chemotherapy with cyclophosphamide, doxorubicin and paclitaxel prior toaxillary lymphadenectomy with quadrantectomy, followed bylocore gional radiotherapy to the right breast area and the right su praclavicular fossa and monoclonal therapy with trastuzumab and pertuzumab.
Within a year, anew thoracic CT scan showed persistence of the lung nodule with no changes according to maximum diameter, but with solid portion of 6mm. Due to persistence in time and enlargement of the solid portion, the nodule was interpreted as a lung primary tumor. We decided to perform an excisional biopsy with intraoperative pathological analysis, which informed positive for neoplastic cells, possible adeno carcinoma. Due to the nodule characteristics and its location, the decision was toper form a right upper lobectomy with systematic nodal sampling after confirmation of the malignancy of the nodular lesion.
At cross section, macroscopic observation showed a con solidated area of grayish white. The diameters were 2cm and 1.3 cm respectively. At 4cm of the previous mentioned lesion, a 0.4cm area of increased consistency was found.
Microscopic observation revealed acinar adenocarcinoma in the bigger lesion. Absence of mediastinal nodal metastasis. The small lesion was on the basis of chronic inflammation and fibrosis. Immunohistochemical staining was positive for synap tophys in and chromogranin. (Fig. 2). The lung adenocarcinoma was staged as T1N0M0.
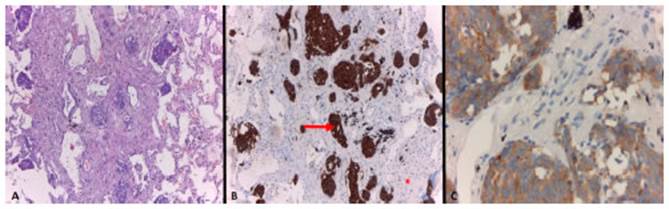
Fig. 2 Histological examination of resected lung tissue. A: Adenocarcinoma. Well differentiated glandular epithelial proliferation with stromal invasion. H&E staining x10. B: Arrow indicated oval and short fusiform cell hyperplasia displayed in nodular arrangement, on the basis of chronic inflammation and fibrosis (asterisk). Neuroendocrine cells strongly positive for chromogranin immunohistochemical staining, x10. C: Tumorlet positive for synaptophys inimmunohistochemical staining, x40
Discussion
Proliferation of LNC may be confined to the mucosa of the airway or invade it locally, beyond the basement mem brane to form tumorlets, benign neoplasms (lesions ≤ 0.5cm) or develop into carcinoid tumors (lesions >0.5cm).
Tumorlets are prevalent among middle aged women, between 50 and 70 years old, often diagnosed with a pre viously diagnosed cancer, particularly breast carcinoma. Regarding clinical significance, tumorlets are usually asymptomatic and lack evidence of severely restrictive or obstructive lung disease, despite they are surrounded by inflammatory and reactive tissue with fibrosis, even when they are multiple7.
These lesions are usually seen as small, smooth and spherical nodules on CT scan, with surrounded areas of scarring and bronchiectasis. They are usually informed as indeterminate because of the size and morphology8. As said before, tumorlets are often seen in patients with a previously diagnosed cancer. In case of a patient with multiple pulmonary nodules and a previous diagnosed malignancy, a misdiagnosis of metastatic disease can occur if lung biopsies are not performed.
In our case, the combination of the pathology report mentioning the lung nodule and the tumorlet in the resec tion specimen with the CT showing a tiny ground glass nodule in the same region, constitute evidence that they are the same entity (Fig 1). Retrospectively, we believe that is highly likely that the nodular ground glass opacity seen on the CT scan is actually the tumorlet found at the histopathologic examination.
Tumorlets share morphologic and immunohistochemi cal features with other neuroendocrine tumors. LNC have round, oval, or spindle nuclei with salt-and-pepper chro matin and clear or eosinophilic cytoplasm. Hyperplastic LNC spread thru the basement membrane of the peribron chiolar tissues forming tumorlets with nesting pattern and desmoplastic stromal reaction. Diagnosis of NLC should be performed by immunostaining with chromogranin, synaptophysin and CD569.
The relationship between pulmonary tumorlets and carcinoid tumors has not been definitively established. As previously mentioned, it is suggested that DIPNECH de velops into tumorlets and eventually into carcinoid tumors. The mechanism for any such progression may involve critical genetic alterations, such as allelic imbalance of the int-2 gene, causing progressive growth deregulation10.
Since isolated tumorlets are incidental finding at histo pathologic examination of lung parenchyma after surgery, no further treatment is necessary, not being the case when associated with DIPNECH. Although they tend to be benign and indolent, there are reports of tumorlets with cellular atypia, progression into invasive neuroendocrine tumors and lymph node metastases11,12. Surveillance imaging with CT scan should be performed according the base pathology, as tumorlets are usually findings after lung resection for other reasons.
This case report reveals the importance of consider ing carcinoid tumorlets as differential diagnosis of small pulmonary nodules visible on CT, as they usually mani fest as a sub centimeter pulmonary nodules, tending to be misdiagnosed as primary lung tumors or metastases.
The presence of these lesions in association with other tumors are rare, most cases being breast or lung cancer13. Although they tend to be benign and indolent neoplasms, some lesions may progress into invasive neuroendocrine tumors.
The patient is in good condition, still receiving trastu zumab/pertuzumab therapy, currently under surveillance without any signs of tumor recurrence.














