Introduction
Besides being the main nutrient-fixing organ in plants, roots are responsible for soil water and nutrient uptake. Their absorption capacity is directly related to their developmental stage. Branching and penetration are root prime morphological characteristics supporting plant development in moisture deficient soils. A greater or lesser root development determines water absorption rates, and influences important physiological processes such as photosyn thesis, respiration, and cellular elongation, among other metabolic activities 1,12,18.
Roots play a vital role in crop yield. The mentioned water and nutrient uptake directly depends on transport efficiency of root cell plasma membranes, root architecture, nutrient transport regulation, and root growth 14,19. In this sense, Jackson et al. (2008) have recognized the close relationship between root architecture (branching and penetration patterns) and soil water and mineral uptake. They also agree with Jenik et al. (1964), who classified the root system into a) ‘macrorhizae’, including long-lived support roots, which carry out most support, transport, and storage functions; and b) ‘brachyrhizae’ or feeder roots, generally thin, short-lived and carrying out absorption and metabolic functions (rhizosynthesis). Holdaway et al. (2011) argued that roots are denser when subjected to moisture and nutrient restrictions. In agreement, a slightly higher proportion of fine roots was found in sites with low nutrient content and penetration restrictions. It has also been reported that tree root production generally follows aerial growth patterns, tending to stabilize with canopy closing 7,38.
In oil palms (Elaeis guineensis Jacq.), the highest proportion of absorbing roots is located in the superficial horizons, at depths greater than 45 cm 4. Fine roots are generally found between 0 and 20 cm of soil depth, while thin and medium roots are found between 20 and 45 cm. From that depth downwards, the proportion of roots per unit volume of soil decreases sharply 3,6,33. Oil palm root system is characterized by stable and continuous growth. From germination and during the first year, fine roots grow 1 cm day-1. When the plant reaches adulthood (>10 years), fine rooting drops to 0.1 cm day-1(13. Vertical growth patterns of oil palms show a constant direction, conditioned by gravity, whereas horizontal growth shows either upward or downward direction. This is partly determined by emer gence times and environmental conditions such as light, temperature, pH, and oxygen avail ability, among others 14. Subsoil root growth and development may also be affected by mechanical or chemical properties such as acidity and high water table 17,31.
Root activity is based on biomass and root length 26,37, which indirectly reflect plant absorbing capacity 18,33,35. Root length density (RLD), i.e. root length (cm) and diameter (fine, thin, medium) in a specific area, is associated with soil aeration and water/nutrient absorption and transport 28. Root vertical distribution allows quick responses to seasonal changes affecting water and nutrient availability.
Unlike the reasonably well studied production rate of aerial biomass, information on root biomass remains rather limited, mainly due to methodological issues 21. Therefore, the present study aimed to describe vertical distribution of different types of oil palm roots and its association with soil bulk density in a haplic Luvisol soil, a deep and imperfectly drained soil with a short waterlogging period.
Materials and methods
Fieldwork was carried out in a 9 year old commercial oil palm (Elaeis guineensis Jacq.) plantation, with an area of 1-30-00 ha, in the ejido Filadelfia (17°37’10’’ N and 91°55’50’’ W), municipality of Palenque, Chiapas, Mexico. The cultivated genotype was hybrid Deli x Avros. The plantation was established at 9 x 9 m (planted in a triangular pitch arrangement). Mean annual precipitation and temperature are 2500 mm and 27 °C, respectively, and average altitude of 29 meters above sea level 9.
The soil was described and classified after Cuanalo (1990). A 1 kg sample was taken from each horizon for analysis at the Laboratory of Water, Soil and Plants of the Graduate College, Tabasco Campus. The following parameters were determined in all soil samples: organic matter (OM) using Walkley and Black method; pH by water potentiometry (1:2), and texture by Bouyoucos. Ca2+, Mg2+, K+, Na+ were extracted using 1 N ammonium acetate at pH 7.0 and quantified by spectrophotometry. Extractable P was obtained by the Olsen method, and total N by the semi-micro Kjeldahl method. All analyzes were performed as proposed by the Official Mexican Standard (2002). The soil was classified according to the World Reference Base for Soil Resources based on physicochemical analysis results and soil profile descriptions 36.
For root and soil bulk density, three wells were dug to a depth of 1.5 m at representative points of the studied area, in between plants, equidistant from the nearest palms. Samples were taken from each of the four cardinal sides (north-south-east-west) of each soil profile, every 10 cm, using the cylinder method 33. Fifteen 270.5 cm3 samples, were taken from each cardinal side, 60 per profile. Root vertical distribution was observed on these samples.
For each sample, roots were carefully washed, measured (length and diameter) and oven-dried at 70° C to constant weight. Later, they were weighed with precision balance (± 0.0001 g). According to diameter, roots were classified as: fine (<1 mm), thin (1-3 mm) and medium (3-10 mm) 5. Root length density (RLD) was calculated after Moreno et al. (2005):
RLD = Root Length/Soil Volume
where:
root length = expressed in km and soil volume in m3.
Soil samples used to estimate bulk density were dried in a forced circulation oven (Venticell) at 60°C to constant weight, for approximately 72 hours. Estimation was made according to:
DA= Soil weight/soil volume
where:
soil weight = expressed in g and soil volume in cm3.
The resulting data were subjected to ANOVA, multiple mean comparisons (Tukey α= 0.5), and correlation analysis using the SAS statistical package (2004), version 9.1.
Results and discussion
Soil classification and description
The studied soil was a haplic Luvisol (LVha) with a B argic horizon. Sandy, clayey and loamy textured. According to the NOM-021-RECNAT-2000, mildly acidic in the first 30 cm depth and moderately alkaline at 30-60 cm depth (Table 1, page 160).
Table 1: Tabla 1: Physicochemical properties of a Haplic luvisol cultivated with oil palm in Ejido Filadelfia, Palenque, Chiapas, Mexico. Propiedades fisicoquímicas del Luvisol háplico cultivado con palma de aceite en el Ejido Filadelfia, Palenque, Chiapas, México.
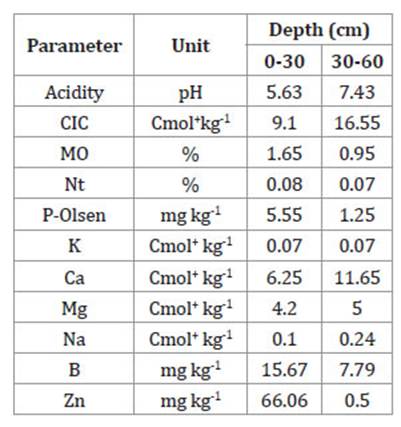
At both depth levels, total N content is low, while P content is medium in the first 30 cm and low in the 30-60 cm level. K shows very low values at both depths. Exchangeable Ca and Mg show medium and high concentrations, respectively, while cation exchange capacity (CIC) is low in the first 30 cm of soil depth, reaching medium level at 30 to 60 cm depth.
Physiographically, Haplic Luvisols are located in flat areas with slightly convex slopes (less than 0.5%). Their parent material corresponds to alluvial sediments influenced by marsh sediments, both from the Pleistocene. They are deep soils with a thin A horizon, loamy-sand textured and dark-brown colored. Imperfectly drained, but with a waterlogging period shorter than that of gleyic Luvisols. In these soils, distance to the water table and humidity regime are more important than low permeability 26. Haplic luvisols found in Savannah of Palenque are usually covered by forests and agricultural lands 26. This region hosts major rubber (Hevea brasiliensis L.) and oil palm (Elaeis guineensis Jacq.) plantations, grazing lands, and, during a certain period each year, watermelon (Citrullus lanatus Thunb.), Haplic luvisols are optimal soils for oil palm cultivation due to their depth (> 150cm), over 50% base saturation, and pH of 5.5-6.6 16. In the present study, deficiencies in primary macronutrients were detected. Other important soil parameters, such as pH and CEC, indi cated that oil palms can grow and develop without toxicity problems and that chemical fertilizers correcting the mentioned deficiencies can be retained in the exchange complex 16,20,30. Soil organic matter was low at the two sampled depths (0-30 and 30-60 cm). Although low organic matter is characteristic of oil palm plantations in the area, this could be a limiting factor, considering the role of OM in soil physical, chemical, and biological fertility. Regarding physical factors, and given soil structure, oil palm roots were not able to fully breathe and grow.
Vertical root distribution
Figure 1 (page 161) shows that root length density (RLD) decreased as soil depth increased. The highest number of oil palm roots (73%) was found in the first 30 cm, with 78% of fine roots, in agreement with Barrios et al. (2003).
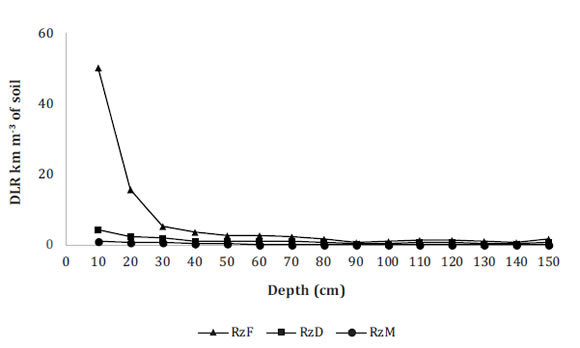
Roots: RzF = fine; RzD = thin; RzM = medium.
Raíces: RzF = finas, RzD = delgadas, RzM = medias
Figure 1: Figura 1: Root length density (km m-3) of oil palms cultivated in a haplic Luvisol in Palenque, Mexico. Densidad de longitud de raíces (km m-3) de palma de aceite cultivada en un Luvisol háplico de Palenque, México.
Although most oil palm roots are found in surface horizons, roots can still be found at depths of up to 1.5-5 m 37.
Fine roots resulted the most abundant through the entire soil profile, evidencing good absorption and metabolic activity 10,37. The largest number (341) of fine roots was found in the first 10 cm, followed by thin roots 11 and medium roots (Figure 2, page 161).
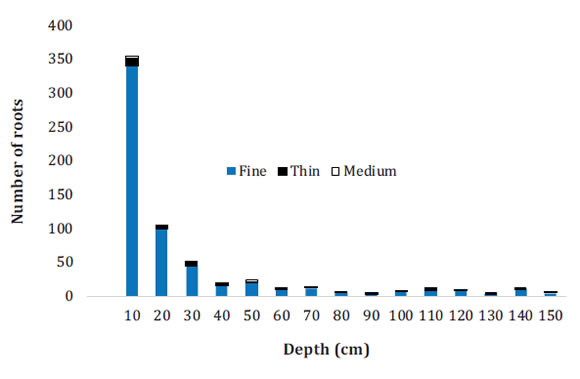
Roots: RzF = fine; RzD = thin; RzM = medium.
Raíces: RzF = finas, RzD = delgadas, RzM = medias.
Figure 2: Figura 2: Distribution of the number of oil palm roots growing in a haplic Luvisol in Palenque, Mexico. Distribución de número de raíces de palma de aceite cultivada en un Luvisol háplico de Palenque, México.
Table 2 shows comparison of means for RLD, for p ≤ 0.05. In the first 10 cm depth numerous, mainly fine roots were observed.
Table 2: Tabla 2: Root Length Density (RLD km m-3) according to diameter, depth and bulk density in an oil palm plantation on haplic Luvisol, Palenque, Mexico. Densidad de Longitud de Raíces (DLR km m-3) según diámetro, profundidad y densidad aparente en una plantación de palma de aceite cultivada en un Luvisol háplico de Palenque, México.
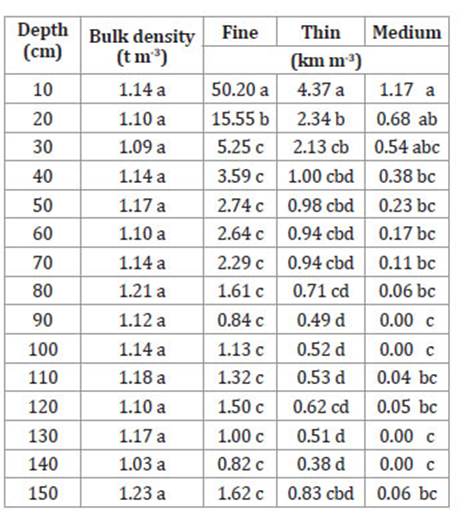
N = 12. Different letters within columns indicate significant differences (Tukey α = 0.05).
N = 12. Letras diferentes dentro de columnas indican diferencias significativas (Tukey α = 0,05).
Significant statistical differences in RLD values were found between the first 10 cm and the other depths for each type of roots (fine, thin and medium). As depth increased, RLD values became more homogeneous. These results differ from previous studiesfinding most roots at greater depths, in the first 45 cm 6,33, or deeper 2,23, even for other tree species 18,22,25. Bulk density did not show significant differences among depths (Table 2), nor a relationship with DLR. The African palm, due to its high capacity for radical exploration at high depths, may contribute to high soil bulk density levels through root decomposition 15,29.
Significant differences were found between number of fine (NFR) and thin roots (NTR) in the first 10 cm, and deeper. However, no significant differences in number of medium roots (NMR) (Table 3, page 163) could be observed.
Table 3: Tabla 3: Number of roots (NR) by depth, diameter and bulk density in oil palms grown in a haplic Luvisol soil in Palenque, Mexico. Número de raíces (NR) por profundidad, diámetro y densidad aparente en palma de aceite cultivada sobre un suelo Luvisol háplico de Palenque, México.
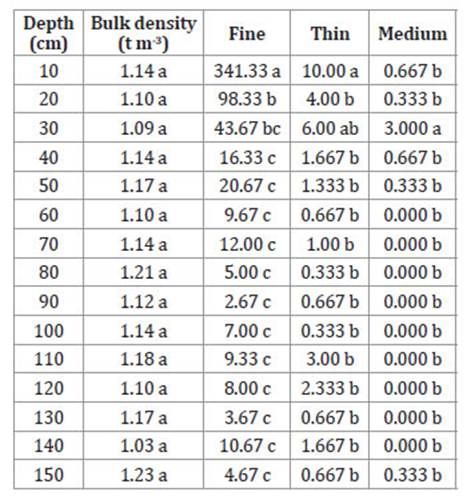
N = 12. Different letters within columns indicate significant differences (Tukey α = 0.05).
N = 12. Letras diferentes dentro de columnas indican diferencias significativas (Tukey α = 0,05).
The first 50 cm of soil depth contained 88% of total fine roots, 67% of total thin roots, and 94% of total medium roots. The number of medium roots decreased between 60 and 140 cm, showing up again at 150 cm.
The results showed that fine roots were distributed throughout the entire soil profile, evidencing high nutrient-absorption and metabolic activities, resulting in good palm oil yields, despite soil limitations. In Indonesia, where this palm is cultivated on millions of hectares, biomass yield is of great importance for carbon capture and degraded soils resto ration 34.
Conclusions
The studied soil was classified as a haplic Luvisol. The highest number of oil palm roots was found in the first 30 cm of soil (73%), most of them being fine roots (78%). The first 50 cm contained 88, 67 and 94% of total fine, thin, and medium roots, respectively. Further study is required to assess the behavior of root length density over the years, for 15, 20, 25 and 30 years old plants.














