Degeneration joint change is a characteristic part of the aging process and occupational stress (Burkhardt & Grupe, 2018; Chen et al., 2017; Listi & Manhein, 2012; López-Barrales et al., 2015; Mays & Dungworth, 2009; Ortner, 2003; Schaffer et al., 2010; Watkins, 2012; Weiss & Jurmain, 2007). In adulthood, and increasingly during the post-reproductive interval, a combination of progressive, deteriorative loss of bony tissue leads to porosity whereas reactive bone formation in the articular surfaces of the joints causes marginal ostephytic lipping (Knüsel et al., 1997; Larsen, 2015; Snodgrass, 2004). Once the joint capsule has suffered collapse and the mating articular pairs establish contact, the subsequent friction between elements results in eburnation (Molnar et al., 2011; Waldron, 2009; Yustos et al., 2021). In addition to age, other factors such as sex, genetics, obesity, genetic predisposition and occupational stress may increase the severity of degenerative joint changes (Chen et al., 2017; Jurmain, 1977; McGonagle et al., 2010; Ortner, 2003; Weiss & Jurmain, 2007). Furthermore, stress suffered earlier in life, such as disease or trauma to the joint can develop severe degenerative changes (Ortner, 2003; Gibbon et al, 2014; Watkins, 2012). Acute stress from vehicular accidents and contact sports such as rugby can rupture the intervertebral joints of the thoracic spine resulting in Schmorl’s nodes on the centrum surface and other degenerative markers (Űstűndağ, 2009). Degenerative joint changes commonly affect the wrist (Roberts et al., 2006), knees (Plomp et al., 2015), temporomandibular joint (Suby & Giberto, 2019), upper limbs (Palmer et al., 2016), spine (Bridges, 1994; Knüsel et al., 1997; Listi & Manhein, 2012; Quispe & Williams, 2019; Snodgrass, 2004; Űstűndağ, 2009; Williamson, 2015), shoulders (Brennaman et al., 2017), hips and elbows (Watkins, 2012; Woo & Sciulli, 2013). As individuals age they will commonly experience pain, inflammation and loss of mobility in the affected areas (Goldring & Goldring, 2006).
While it is accepted that degenerative joint changes manifests in eburnation and osteophyte formation, whether porosity falls under the diagnostic term “osteoarthritis” and is the result of the same processes has been called into question (Austin, 2017; Becker, & Goldstein, 2018; Rothschild, 1997). Two or more of these features has to be present to diagnose osteoarthritis and only eburnation is pathognomonic of this joint disease. It must be borne in mind that porosity in the absence of eburnation and lipping is not necessarily evidence of osteoarthritis (Austin, 2017; Becker & Goldstein, 2018; Gibbon et al., 2014; Molnar et al., 2011; Palmer et al., 2016; Roberts et al., 2006; Rothschild, 1997).
Furthermore, some variability exists in the pathology of the different areas affected. For instance, the elbows and knees tend to exhibit deep grooves coupled with eburnation (Ortner, 2003), whereas other areas primarily present polished surfaces, including the humeral head (Woo & Sciulli, 2013) and the metacarpal articular surfaces (Molnar et al., 2011). Similarly, the vertebral column may present extensive lipping but eburnation is often less extreme with polish and very weak grooves, perhaps from constraints in mobility. In the spine, degenerative joint changes primarily affect the subchondral bone of the intervertebral discs and the apophyseal joints (Maat et al., 1995; Ortner, 2003; Woo & Pak, 2014). Due to a decrease in cellular activity that is essential to the maintenance of disc cartilage, degeneration is considered to begin with the intervertebral disc (Benoist, 2003; Űstűndağ, 2009; Zehra et al., 2015). Apophyseal joints act to counter and support the posterior load-bearing muscles of the back and are principally associated with eburnation, porosity, and marginal lipping. The subchondral bone (vertebral body) is characterized primarily by erosion and osteophyte development (Calce et al., 2017; Ortner, 2003). The lower thoracic shows a higher severity than the upper thoracic, whereas the region least affected is the seventh thoracic to the upper thoracic (Larsen, 2015).
Age differences
Most individuals over the age of 50 years across cultures and geographic regions will experience degenerative join changes associated with osteoarthritis (Felson, 2003; Molnar et al., 2011). Some suggest that over the life course, articular cartilage will be subject to deterioration due to extended periods of mechanical use (Bettié et al., 2009; Loeser, 2009, 2010). During the aging process, progressive change in articular cartilage accumulate in the extracellular matrix (Loeser, 2010). Degenerative changes may rapidly progress in older individuals because of the decreased ability of articular cartilage to withstand the pressures of mechanical loading (Molnar et al., 2011; Ortner, 2003; Waldron, 2009). Outliers who suffered more severely than others, perhaps from a decreased resistance to stress from the accumulation of physical damage, chronic illness or genetic predisposition, increase in number as a function of advancing age (Marklein et al., 2016).
Sex differences
There is much variation existing in the expression of degenerative joint changes in adult females and males. As a result, there are conflicting views on the relationship between sex differences and degeneration during the life course. According to Larsen (2015) when significant differences can be observed for degenerative joint changes associated with osteoarthritis, males tend to express a higher frequency than females (Molnar et al., 2011). Similarly, Woo & Pak (2014) noted that males showed a greater severity than females. Rogers & Dieppe (2003) argue that while this is true for men before the age of 50 years, after that age women show greater prevalence of degenerative joint changes involved in the expression of osteoarthritis (Nevitt & Felson, 1996; Rogers & Dieppe, 2003). This could possibly be related to declining levels of estrogen that women experience following menopause. Quispe & Williams (2019) found that males present a greater severity of degenerative markers than females for most indicators. However, older adult females, aged 50-55 years, exhibit significantly greater eburnation than their male counterparts, although this was not true of the elderly (Quispe & Williams, 2019). Meanwhile, Snodgrass (2004) found both sexes exhibit the same progression of vertebral degeneration with respect to age, although females are more variable. Srikanth et al. (2005) analyzed 34 studies on sex differences and degeneration to determine whether there were an increased severity in males or females. Although only four studies on the spine were investigated, none yielded significant difference between males and females (Srikanth et al., 2005).
Given these disparate results, it is possible that females and males may or may not differ in the degeneration of the thoracic skeleton with respect to age. The two sexes are compared at two post-reproductive age cohorts, separated by twenty years. The twenty-year interval between the two age cohorts conservatively estimates the amount of time needed for significant skeletal degeneration to occur based on the length of the human maturation sequence (Quispe & Williams, 2019). The current study on the thoracic spine serves as a continuation of Quispe & Williams (2019) who focused on the cervical vertebrae. Therefore, the same sample size and composition -both sexes at 50-55 and 70-75 years- and the use of the Buikstra & Ubelaker (1994), scoring system, is replicated in this study.
MATERIALS AND METHODS
To examine whether sex differences in degenerative markers exists with respect to age, thoracic vertebrae were examined from complete skeletons of the William M. Bass collection of the University of Tennessee, Knoxville, where an extensive collection of human remains with recorded sex, age and causes of death is curated (Shirley et al., 2011). The socioeconomic status of 60% of donors can be categorized as low to middle income and 40% were laborers with a high school diploma (Wilson et. al., 2007). The donor self-reported parameters included working-class, with a living stature between five-feet two inches and six-feet. Specific stature parameters decreased extrinsic factors that could potentially affect the expression of degenerative markers. Using these selection criteria, 99 individuals from the Bass collection were examined (Table 1). The sample included females and males between the ages of 50-55 years, and females and males between the ages of 70-75 years, replicating the research design of Quispe & Williams (2019). The total sample included 52 females and 47 males, of whom 53 were older adults and 46 were elderly individuals (Table 1). Each thoracic vertebra from T1 to T12 was examined. The superior and inferior surfaces of the body were scored for lipping, porosity, and eburnation.
TABLE 1 Number of individuals included in the study by age category and sex
| Category | Age in years | Female | Male | Total |
| Older adult | 50-55 | 28 | 25 | 53 |
| Elderly adult | 70-75 | 24 | 22 | 46 |
| Total | 52 | 47 | 99 |
There are multiple interpretations available for the diagnosis of osteoarthritis. Most acknowledge eburnation as its most severe manifestation and indeed pathognomonic of osteoarthritis, although many researchers also attribute this pathology to joint surfaces that exhibit at least two degenerative changes including porosity, osteophytosis or joint contour modifications (Austin, 2017; Becker & Goldstein, 2018; Gibbon et al., 2014; Molnar et al., 2011; Palmer et al., 2016; Plomp et al., 2015; Roberts et al., 2006; Suby & Giberto, 2019; Űstűndağ, 2009). Yet there is variation in the way in which these diagnostic principles are applied. Whereas some collapse the categories of eburnation and other degenerative joint changes under osteoarthritis (Austin, 2017; Becker & Goldstein, 2018; Gibbon et al., 2014; Palmer et al., 2016; Űstűndağ, 2009), others tabulate these markers separately (Henderson et al., 2013; Molnar et al., 2011; Plomp et al., 2015; Roberts et al., 2006; Suby & Giberto, 2019; Watkins, 2012; Woo & Sciulli, 2013; Yustos et al., 2021; Zoëga & Murphy, 2016). Given these conflicting diagnostic criteria, the Standards (Buikstra & Ubelaker, 1994) were chosen to rank degenerative joint changes independently as others have done (Henderson et al., 2013; Watkins, 2012; Williamson, 2015; Woo & Sciulli, 2013; Yustos et al., 2021; Zoëga & Murphy, 2016).
Following Buikstra & Ubelaker (1994) and Quispe & Williams (2019), the degree of lipping was observed macroscopically and scored from 1- 4, 1) barely observable, 2) sharp ridge, often times curled and coupled with spicules, 3) pronounced spicule formation, and 4) ankylosis or fusion (Table 2; Fig. 1). Porosity was ascertained macroscopically using scores from 1-3 and included 1) observable pinpoint perforations, 2) presence of larger coalesced perforations, and 3) both pinpoint and coalesced expressed (Fig. 2) (Buikstra & Ubelaker, 1994; Quispe & Williams, 2019). Eburnation was scored from 1-3 via macroscopic examination and included 1) unobservable or questionably present, 2) polish visible, and 3) polish present coupled with grooves (Fig. 3) (Buikstra & Ubelaker, 1994; Quispe & Williams, 2019). In addition, the extent of lipping, porosity and eburnation were also recorded from 1-3, including 1) 1/3, 2) 1/3 to 2/3, and 3) >2/3 of the surface affected (Table 2) (Buikstra & Ubelaker, 1994; Quispe & Williams, 2019). A scoring error study conducted at Georgia State University yielded no significant difference between scores for eight vertebrae conducted as two scoring attempts separated by two weeks.
TABLE 2 Description of vertebral osteoarthritic features scored using Buikstra and Ubelaker (1994)
| Feature | Score | Extent | Description |
| Marginal lipping | 1 | <1/3 | Barely observable |
| 2 | 1/3-2/3 | Sharp ridges, often curled and with some spicules formation | |
| 3 | >2/3 | Pronounced formation of spicules | |
| 4 | Ankylosis or fusion | ||
| Porosity | 1 | <1/3 | Observable pinpoint perforations |
| 2 | 1/3-2/3 | Presence of larger coalesced perforations | |
| 3 | >2/3 | Both pinpoint and coalesced expressed | |
| Eburnation | 1 | <1/3 | Unobservable or questionably present |
| 2 | 1/3-2/3 | Polish visible | |
| 3 | >2/3 | Polish present coupled with grooves |
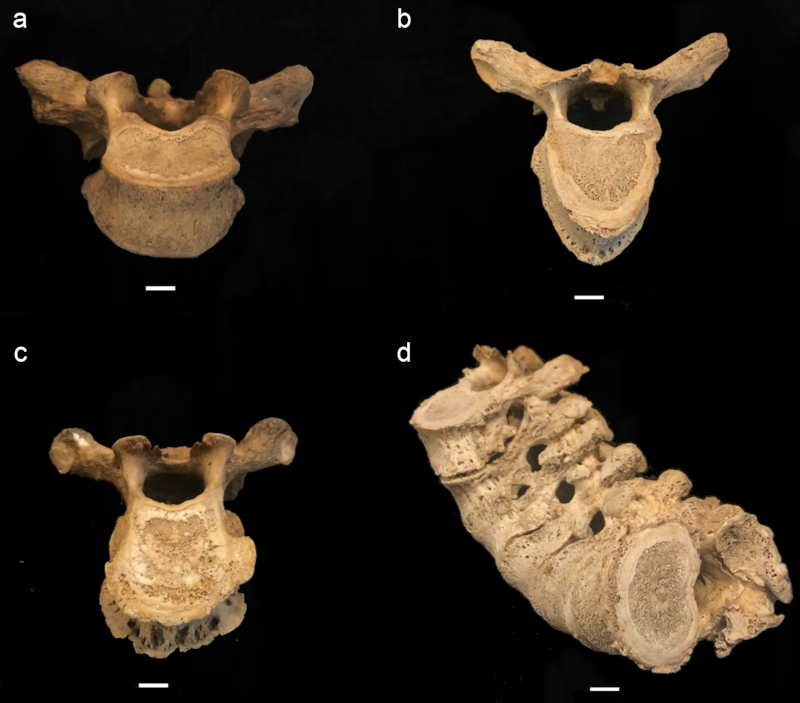
FIGURE 1 Scoring system for lipping; (a) barely observable, (b) sharp ridge, often times curled and coupled with spicules, (c) pronounced spicule formation, and (d) ankylosis or fusion; scale: 1 cm.
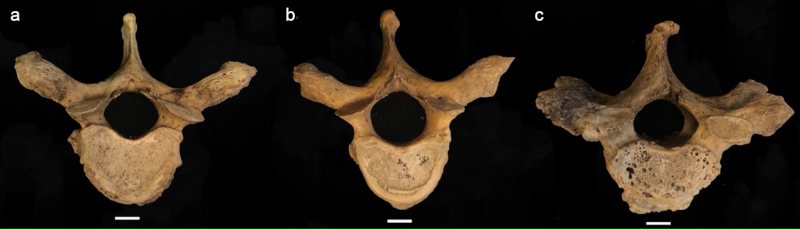
FIGURE 2 Scoring system for surface porosity; (a) observable pinpoint perforations, (b) presence of larger coalesced perforations, and (c) both pinpoint and coalesced expressed; scale: 1 cm.
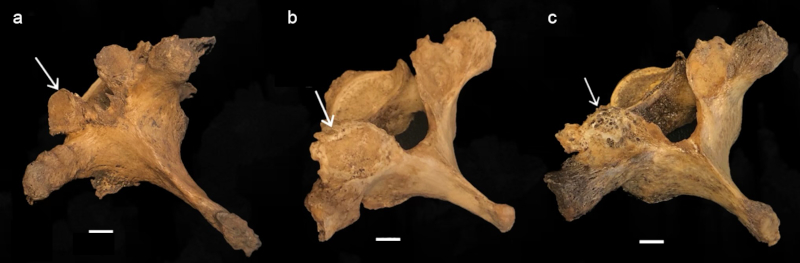
FIGURE 3 Scoring system for eburnation, identified with white arrows; (a) unobservable or questionably present, (b) polish visible, and (c) polish with groove(s); scale: 1 cm.
Lipping, porosity and eburnation signal osteoarthritis but reflect the various responses of the body to the narrowing or obliteration of interior joint spaces (Robson-Brown et al., 2008; Zehra et al., 2015). The scoring system used here preserves the independence of each degenerative marker. Although none of them specifically, such as eburnation, or in combination are used to diagnosis osteoarthritis, all of these degenerative markers are involved in the expression of this pathology.
The Mann-Whitney U statistic was included to test for differences within and across age cohorts. These tests included whether the sexes manifest thoracic degeneration differently by comparing older adults to elderly within each sex. Whether females and males differed as older adults and as elderly individuals was also explored. To compare thoracic degeneration across categories, 95% confidence intervals and mean values for all vertebrae scored for the four age/sex groupings are presented for each degenerative marker.
Ordinal regression was included to address the degree to which eburnation, widely considered to be diagnostic of osteoarthritis, compares to the other degenerative markers (Woo & Pak, 2014). The resulting goodness-of-fit chi-square statistic was examined to evaluate the significance of the relationship between degenerative markers. Ordinal regression allowed for a comparison of all thoracic vertebrae elements pooled together to identify significant associations, or lack thereof, between degenerative markers. Marginal lipping, porosity and eburnation were compared with one another and each of these with their respective extent of degenerative joint changes on the superior and inferior surface.
RESULTS
Older adult males have higher or equal means than their female counterparts for all features excepting eburnation and extent of eburnation where females show greater or equal values (Table 3). Similarly, the means for elderly males are higher than those of elderly females with the exception of porosity, eburnation and extent of eburnation where the converse is present (Table 3). In fact, the means for elderly males surpass those of all other age and sex groups with the exception of porosity, eburnation and extent of eburnation where elderly females are higher (Fig. 4). The means for lipping and extent of lipping are higher than for porosity and eburnation (Fig. 4). The standard deviation is highest for extent of marginal lipping across age and sex categories (Table 3).
TABLE 3 Descriptive statistics for each sex and age category
| Female | Male | |||||||||
| Older adults | N | Min | Max | Mean | SD | N | Min | Max | Mean | SD |
| (50-55 years) | ||||||||||
| Marginal lipping superior | 334 | 1 | 4 | 1,580 | 0,688 | 300 | 1 | 3 | 1,670 | 0,691 |
| Marginal lipping inferior | 336 | 1 | 4 | 1,590 | 0,685 | 300 | 1 | 3 | 1,740 | 0,704 |
| Extent of marginal lipping superior | 336 | 1 | 3 | 1,930 | 0,878 | 300 | 1 | 3 | 2,030 | 0,884 |
| Extent of marginal lipping inferior | 336 | 1 | 4 | 1,920 | 0,914 | 300 | 1 | 3 | 1,920 | 0,873 |
| Porosity superior | 336 | 1 | 2 | 1,020 | 0,133 | 300 | 1 | 3 | 1,060 | 0,244 |
| Porosity inferior | 336 | 1 | 2 | 1,040 | 0,186 | 300 | 1 | 2 | 1,060 | 0,238 |
| Porosity extent superior | 336 | 1 | 3 | 1,610 | 0,787 | 300 | 1 | 3 | 1,810 | 0,844 |
| Porosity extent inferior | 336 | 1 | 3 | 1,580 | 0,795 | 300 | 1 | 3 | 1,710 | 0,817 |
| Eburnation superior | 336 | 1 | 3 | 1,040 | 0,221 | 300 | 1 | 2 | 1,040 | 0,204 |
| Eburnation inferior | 336 | 1 | 3 | 1,040 | 0,215 | 300 | 1 | 2 | 1,020 | 0,140 |
| Eburnation extent superior | 336 | 1 | 3 | 1,070 | 0,306 | 300 | 1 | 2 | 1,010 | 0,100 |
| Eburnation extent inferior | 336 | 1 | 3 | 1,090 | 0,384 | 300 | 1 | 2 | 1,020 | 0,151 |
| Female | Male | |||||||||
| Elderly | N | Min | Max | Mean | SD | N | Min | Max | Mean | SD |
| (70-76 years) | ||||||||||
| Marginal lipping superior | 285 | 1 | 3 | 1,690 | 0,636 | 254 | 1 | 4 | 1,980 | 0,736 |
| Marginal lipping inferior | 285 | 1 | 3 | 1,770 | 0,647 | 255 | 1 | 4 | 2,080 | 0,740 |
| Extent of marginal lipping superior | 285 | 1 | 4 | 1,930 | 0,893 | 254 | 1 | 3 | 2,170 | 0,880 |
| Extent of marginal lipping inferior | 285 | 1 | 3 | 2,000 | 0,878 | 255 | 1 | 3 | 2,230 | 0,859 |
| Porosity superior | 285 | 1 | 3 | 1,160 | 0,457 | 254 | 1 | 3 | 1,140 | 0,392 |
| Porosity inferior | 285 | 1 | 3 | 1,220 | 0,500 | 255 | 1 | 3 | 1,240 | 0,539 |
| Porosity extent superior | 285 | 1 | 3 | 1,730 | 0,853 | 254 | 1 | 4 | 1,790 | 0,885 |
| Porosity extent inferior | 285 | 1 | 3 | 1,680 | 0,848 | 255 | 1 | 3 | 1,680 | 0,836 |
| Eburnation superior | 285 | 1 | 3 | 1,150 | 0,402 | 254 | 1 | 3 | 1,110 | 0,350 |
| Eburnation inferior | 285 | 1 | 3 | 1,080 | 0,293 | 255 | 1 | 2 | 1,050 | 0,228 |
| Eburnation extent superior | 285 | 1 | 3 | 1,120 | 0,428 | 254 | 1 | 3 | 1,100 | 0,394 |
| Eburnation extent inferior | 285 | 1 | 3 | 1,060 | 0,297 | 255 | 1 | 3 | 1,110 | 0,430 |
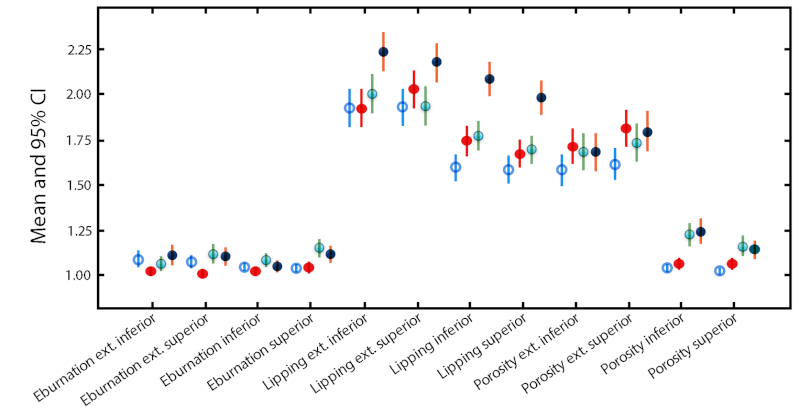
FIGURE 4 Means (circles) and 95% confidence intervals (lines) of all vertebrae scores for each age/sex category; older adult females = unfilled blue circles; older adult males = solid red circles; elderly females = green circles with blue shade; elderly males = solid dark blue circles.
Differences with respect to age within each sex
Elderly adult females are significantly distinct from older adults in porosity of the superior and inferior T8, superior T9 and T10 and inferior T6 and T12 (Table 4). Elderly females also show significantly greater severity than older female adults for marginal lipping, where inferior T1 and T8 and superior T9 are significantly distinct between the two groups. The same pattern is apparent for the extent of marginal lipping where inferior T2, T3 and T11 differ, and for eburnation for the superior surfaces of T9, T10 and T12, where elderly females show significantly greater severity (Table 4). In contrast, older adult females are significantly elevated in the severity of the extent of eburnation for T1 inferior (p=0.031) compared to elderly female individuals (Table 4).
TABLE 4 Results of Mann-Whitney U tests for differences between older adults and the elderly within each sex
| Females only | Marginal lipping | Extent of marginal lipping | Porosity | Extent of porosity | Eburnation | Extent of eburnation |
| T1 inferior | 0.026 O<E | 0.031 E<O | ||||
| T2 inferior | 0.004 O<E | |||||
| T3 superior | 0.036 O<E | |||||
| T3 inferior | 0.040 O<E | 0.021 O<E | ||||
| T6 inferior | 0.024 O<E | |||||
| T8 superior | 0.011 O<E | |||||
| T8 inferior | 0.036 O<E | 0.001 O<E | ||||
| T9 superior | 0.030 O<E | 0.005 O<E | 0.012 O<E | |||
| T10 superior | 0.010 O<E | 0.051 O<E | ||||
| T11 inferior | 0.022 E<O | |||||
| T12 superior | 0.051 O<E | |||||
| T12 inferior | 0.051 O>E | |||||
| Males only | ||||||
| T1 inferior | 0.003 O<E | |||||
| T3 inferior | 0.015 O<E | |||||
| T4 superior | 0.001 O<E | |||||
| T4 inferior | 0.009 O<E | |||||
| T5 superior | 0.009 O<E | 0.024 O<E | ||||
| T5 inferior | 0.019 O<E | 0.011 O<E | ||||
| T6 superior | 0.004 O<E | 0.049 O<E | ||||
| T6 inferior | 0.003 O<E | 0.040 E<O | ||||
| T8 inferior | 0.020 O<E | |||||
| T9 inferior | 0.029 E<O | |||||
| T12 inferior | 0.046 O<E |
Elderly males are by far the most distinctive from their older adult counterparts in marginal lipping, where both superior and inferior surfaces of T4, T5 and T6, as well as inferior T3 and T12 are significantly different (Table 4). Elderly males compared to older male adults are also elevated in the severity of porosity where superior T6 and inferior T5, T8 and T9 differ significantly. In contrast, older male adults show greater severity of porosity in inferior T9 (p=0.029) as well as in the extent of porosity for inferior T6 (p=0.040). The elderly differ far more from older adults in the inferior vertebrae surfaces compared to the superior ones for both sexes, although the difference is more pronounced in males.
Sex differences within age categories
Males exhibit significantly greater severity of marginal lipping compared to females for inferior T1, T8 and T10 as well as superior T1 and T3 (Table 5). The same pattern holds for extent of marginal lipping for inferior T2 and T3 as well as for porosity on superior T1, and extent of porosity in superior and inferior T3 (Table 5). In contrast, females show a nearly significantly greater extent of eburnation compared to males for T12 superior (p=0.052).
TABLE 5 Results of Mann-Whitney U tests for differences between females and males within older adults and elderly individuals
| Older adults (50-55 years) | Marginal lipping | Extent of marginal lipping | Porosity | Extent of porosity | Eburnation | Extent of eburnation |
| T1 superior | 0.029 F<M | 0.006 F<M | ||||
| T1 inferior | 0.028 F<M | |||||
| T2 inferior | 0.022 F<M | |||||
| T3 superior | 0.046 F<M | 0.003 F<M | ||||
| T3 inferior | 0.050 F<M | 0.003 F<M | ||||
| T9 inferior | 0.040 F<M | |||||
| T10 inferior | 0.040 F<M | |||||
| T12 superior | 0.052 M<F | |||||
| T12 inferior | 0.052 M<F | |||||
| Elderly adults (70-76 years) | ||||||
| T1 superior | 0.041 F<M | 0.011 F<M | ||||
| T2 inferior | 0.043 F<M | |||||
| T4 superior | 0.008 F<M | |||||
| T5 superior | 0.019 F<M | |||||
| T6 inferior | 0.022 F<M | |||||
| T7 inferior | 0.029 F<M | |||||
| T9 superior | 0.028 M<F | |||||
| T9 inferior | 0.028 F<M | |||||
| T11 inferior | 0.029 F<M |
Elderly males exhibit a pronounced severity of marginal lipping compared to females of the same age category for superior T4 and T5 as well as inferior T2, T6, T7, T9 and T11 (Table 5). Superior T1 also differs between elderly males and females for extent of marginal lipping and extent of porosity. In contrast, extension of eburnation for T9 superior is significantly elevated (p=0.028) in elderly females compared to males (Table 5). Similar to when age differences were compared within each sex, the inferior surface exhibits greater severity than the inferior surface when females and males are compared within age categories (Table 5).
When the 95% confidence intervals are compared across age and sex categories, notable differences include the distinctly higher marginal lipping and extent of marginal lipping scores for elderly males. Older adult males are relatively elevated in extent of porosity for both inferior and superior surfaces compared to other age/sex categories although the elderly of both sexes surpass older adults in porosity. For extent of eburnation inferiorly, older adult females and elderly males are distinct from older adult males, while superiorly, all age/sex categories are elevated compared to older adult males. The elderly, and particularly females, surpass older adults of both sexes in scores for eburnation superiorly (Fig. 4).
Most of the comparisons between degenerative joint changes are not significant. However, the degenerative markers that do demonstrate statistical significance are instructive (Table 6). Marginal lipping versus extent of marginal lipping on the inferior surface is significantly related in older adults (p=0.013), elderly individuals (p=0.014) and males (p=<0.001). In addition, porosity and extent of porosity on the superior surface are significantly associated in elderly adults (p=0.002) as well as females (p=0.005). Additional comparisons demonstrate significance (Table 6). One of these is porosity and extent of porosity on the inferior surface in elderly adults (p=0.004) and in males (p=0.017). Another is marginal lipping and porosity on the superior surface in older adults (p=0.017). Eburnation is not significantly associated with any of the other variables, including extent of eburnation, suggesting an independent expression of this degenerative marker.
TABLE 6 Chi-square results from ordinal regression of degenerative markers for older and elderly adults and the two sexes
| Older adults (n = 636) | Elderly adults (n = 540) | Females (n = 629) | Males (n = 555) | |||||||||
| X 2 | df | p | X 2 | df | p | X 2 | df | p | X 2 | df | p | |
| Lipping vs. extent of lipping inferior | 16 | 6 | 0,013 | 12,6 | 4 | 0,014 | 21,7 | 4 | <0.001 | |||
| Porosity vs. extent of porosity superior | 15,1 | 3 | 0,002 | 10,6 | 2 | 0,005 | ||||||
| Porosity vs. extent of porosity inferior | 11,2 | 2 | 0,004 | 8,1 | 2 | 0,017 | ||||||
| Lipping vs. porosity superior | 8,1 | 2 | 0,017 | |||||||||
DISCUSSION
Walking erect places great strain on the thoracic vertebrae and has for millions of years (Cook et al., 1983) as well as more recently in prehistory (Trinkaus, 1985). Given this antiquity of vertebral degenerative markers it should be no surprise that deterioration of the spine is quite common in extant humans by 40 years (Bridges, 1994; Ortner, 2003; Quispe & Williams, 2019), but its expression is minor compared to individuals of more advanced ages (López-Barrales et al., 2015; Mays & Dungworth, 2009; Snodgrass, 2004; Űstűndağ, 2009; Yustos et al., 2021). Degeneration of the skeleton accelerates after 50 years and virtually everyone has some form of osteoarthritis by 65 years of age (Solomon, 2001).
The thoracic vertebrae are believed to exhibit less extreme degenerative joint changes of the centrum than the lumbar and cervical regions because of the restricted movement potential of the thorax (Bridges, 1994; Larsen, 2015). In contrast, the apophyseal joints across the vertebral column are similar in degree of degenerative changes associated with osteoarthritis (Bridges, 1994). In this study, the upper (T1-T3) and lower (T9-T12) thoracic regions appear to be affected by degenerative joint changes more severely than the middle (T4-T8) vertebrae (Tables 4 and 5). This is also the case for prehistoric Amerindians from Alabama where the greatest frequency of osteoarthritis occurred on T7-T10 and T12, with a peak at T9/T10 (Bridges, 1994). Other studies have identified the lower thoracic as particularly vulnerable to degenerative joint changes (Űstűndağ, 2009; Yustos et al., 2021); however, the upper thoracic is also cited as a location of extensive degenerative joint changes associated with osteoarthritis (Jurmain, 1990). Larsen (2015) suggests the lower thoracic vertebrae bear the greatest degree of mechanical loading. This observation is corroborated by the significant differences within age (Table 4) and sex (Table 5) categories in this region. The greater severity of degenerative joint changes observed in the lower thoracic vertebrae may be due to the amount of weight it must support and a reduced threshold for mechanical loads compared to the mid and upper thoracic regions.
Although there is no specific activity or labor linked to any unambiguous manifestation of degenerative joint changes, there is some degree of correspondence between repetitive strain, particularly involving mechanical loading and severity (Larsen, 2015; Ortner, 2003; Űstűndağ, 2009), as well as genetic predisposition (Battié et al., 2009). Specific occupational stressors nevertheless appear to operate on the vertebral skeleton over the life course, such as habitual horseback riding or 19th century industrial labor in London (Larsen, 2015), fishing in arctic waters (Zoëga & Murphy, 2016) or labor as an enslaved person (Watkins, 2012). The individuals in our sample from the University of Tennessee William M. Bass Osteology Collection where donated primarily from laborer occupations and from low and middle income service workers (Wilson et al., 2007). Some of the degenerative features observed could have stemmed directly from manual labor, and in the case of many males, and perhaps some females, probably derived from the transmission of heavy loads from the arms to the lower appendicular skeleton via the thoracic spine. The significant differences between the sexes in both age categories (Table 5) provide evidence that the occupational stress from labor in low- and medium skilled work was at least in part gendered. In contrast, López-Barrales et al. (2015) found no statistically significant sex differences in vertebral osteoarthritis in prehistoric Andean peoples from Chile. However, prehistoric Tiwanaku females, in contrast to males, exhibit evidence of gendered repetitive movements in patterns of osteoarthritis (Becker & Goldstein, 2018) whereas males of Iron Age Zambia present greater spinal osteophytosis than do females (Gibbon et al., 2014). It appears the degeneration of the spine is particularly influenced by occupational stress associated with gendered differences in labor profiles.
However, males generally weigh more and have more massive skeletons than females and the greater body weight alone over decades would tend to usher in a mechanical response of the thoracic amphiarthrodial joints from the effects of gravity and daily life. In fact, most studies show a greater prevalence of degenerative joint changes in males compared to females (Molnar et al., 2011), particularly when the sexual division of labor becomes intensified, as for example, after the adoption of agriculture (Larsen, 2015). In this study, males overall presented a greater expression of degenerative markers than females. This was especially observable in margin lipping (Tables 3, 5 and 6; Fig. 4). It is therefore curious as to why older and elderly female adults, with presumably lower body weights, show significantly greater eburnation and extent of eburnation compared to males within each age category, at least for some thoracic vertebrae (Tables 4 and 5). Eburnation arises after the cartilaginous layer between mating pairs of a joint recedes or is dislodged, but the joint continues to operate producing bone-to-bone contact, which polishes and grooves the articulation (Molnar et al., 2011; Ortner, 2003) and may be expressed independent of porosity or marginal lipping (Table 6). Hormonal disruptions associated with menopause may at least partly explain the greater severity of eburnation in females (Larsen, 2015). In addition to the highly eburnated thoracic elements from females noted in this study, eburnation of the cervical vertebrae is also significantly elevated in older adult females compared to males (Quispe & Williams, 2019).
Degenerative changes of the thoracic spine differ from those of the mobile joints of the skeletal system in the presence of ankylosis, which is more common in the vertebral column than in diarthrodial articulations (Larsen, 2015). The smaller inferior surface bears a relatively greater mechanical load compared to the superior articular mate below it, which may increase the growth of osteophytic lipping and eventually ankylosis. The larger number of thoracic vertebrae affected on the inferior compared to the superior surface also could be related to the unique shapes of the vertebral bodies and the placement of the articular facets.
Spinal curvature also may influence degeneration, and in particular, the increasing kyphosis of the mid-thoracic region as a function of age (Toh et al., 2001). Already by age 50 years, substantial kyphosis of the thoracic skeletal has occurred, particularly in males who generally must carry a heavier head, upper arms and torso than females. These differences are merely accentuated with age and lead to extensive lipping and eburnation of the upper and lower thoracic vertebrae. Although a higher degree of degenerative joint changes would be expected to occur where the spinal curvature is the greatest since these are areas of intense stress compared to the vertebrae aligned more directly in the line of gravity, this is not always the case (Bridges, 1994). However, extensive lipping of the thoracic vertebrae is a risk factor for fractures in males older than 50 years, particularly within the mid-thoracic curvature (Pariente et al., 2017), and may help to counteract thoracic kyphosis (Toh et al., 2001). One might also expect a greater expression of degenerative markers as a function of placement, whereby larger inferior vertebrae withstand more weight than smaller superior ones. Although the lumbar skeleton generally exhibits a greater degree of degenerative change compared to the vertebrae positioned more superiorly, there is no direct order of increasing severity as a function of placement. However, the lower thoracic in this study do seem to exhibit a greater severity of degenerative change than superior elements confirming to some extent that position does influence the degree of expression, although other factors may be present. These include changes in spinal curvature anticipating the lordosis of the lumbar vertebrae that may place unique stresses on the lower thoracic skeleton.
CONCLUSION
One goal of this study was to examine whether older adults differed from the elderly within males and within females. Another was to directly compare the sexes within age categories, corresponding to older adults (50-55 years) and the elderly (70-75 years). Age and sex differences exist for degenerative markers of the thoracic region. When only females are considered, elderly individuals exhibit a higher severity than older adults with the exception of the extent of eburnation in T1 and the extent of lipping in T11 on the inferior surface. For males, a greater expression of osteoarthritis is observed for elderly individuals with the exception of porosity in inferior T9 and extent of porosity in inferior T6. Lipping is particular excessive in elderly males throughout the thoracic skeleton.
When comparing older adult of both sexes, males present a greater severity of lipping, extent of lipping, porosity and extent of porosity while females show a greater extent of eburnation. These significant values are restricted to the upper and lower thoracic region. Among elderly adults, males express a higher degree of lipping, extent of lipping and extent of porosity, whereas females exhibit a greater degree of eburnation. Contrary to Rogers & Dieppe’s (2003) predictions, elderly males exhibit a significantly higher severity of lipping throughout the thoracic region compared to elderly females.
Elderly adults of both sexes manifest a greater severity of degenerative markers than their older adult counterparts. Males also express a greater degree of severity than females, excepting eburnation and extent of eburnation for some vertebrae. These exceptions could be a result of differences in hormone profiles associated with reproductive cessation as well as gendered occupational stress.












 uBio
uBio 


