INTRODUCTION
Asthma is a heterogeneous, inflammatory airway disease characterized by recurrent episodes of bronchospasm, bronchial hyperreactivity, and increased bronchial secretions.1-3 It affects ap proximately 300 million people around the world; in Latin America, there is great heterogeneity re garding its prevalence depending on each country, ranging from 5 % to 24 %, as is the case in Costa Rica.4 In Argentina, it is estimated that between 6.4 % and 9.36 % of the population has asthma, according to different studies.5-6
The knowledge of the disease has made signifi cant advancements in recent years, especially in terms of diagnosis and treatment. Paradoxically, even though morbidity and mortality and hospi talizations have been reduced through preventive anti-inflammatory treatment, there are still epide miological indicators of poor control, and in some countries, there are still surprisingly high percent ages of hospitalizations and mortality.1-3,5,7,8
This new knowledge also generates topics of discussion and points of interest that require local perspectives. There are already several recent in ternational guidelines and one local guideline from a few years ago that cover the diagnosis and treat ment of asthma broadly. The objective is to address key questions with the highest level of evidence, focusing on topics related to asthma management, in order to create a practical and easily readable tool with recommendations that provide original contributions aimed at the interests of physicians dedicated to asthma.1-3,9-10
This document was prepared by a panel of specialists from the Association of Allergy and Clinical Immunology (AAAeIC) and the Argentin ian Association of Respiratory Medicine (AAMR), with special emphasis on establishing scientific evidence-based recommendations for the diagnosis and treatment of asthma in adults, adapted to the local context.
MATERIALS AND METHODS
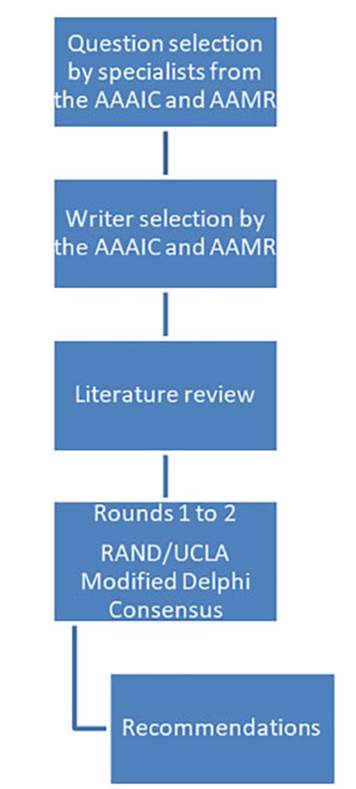
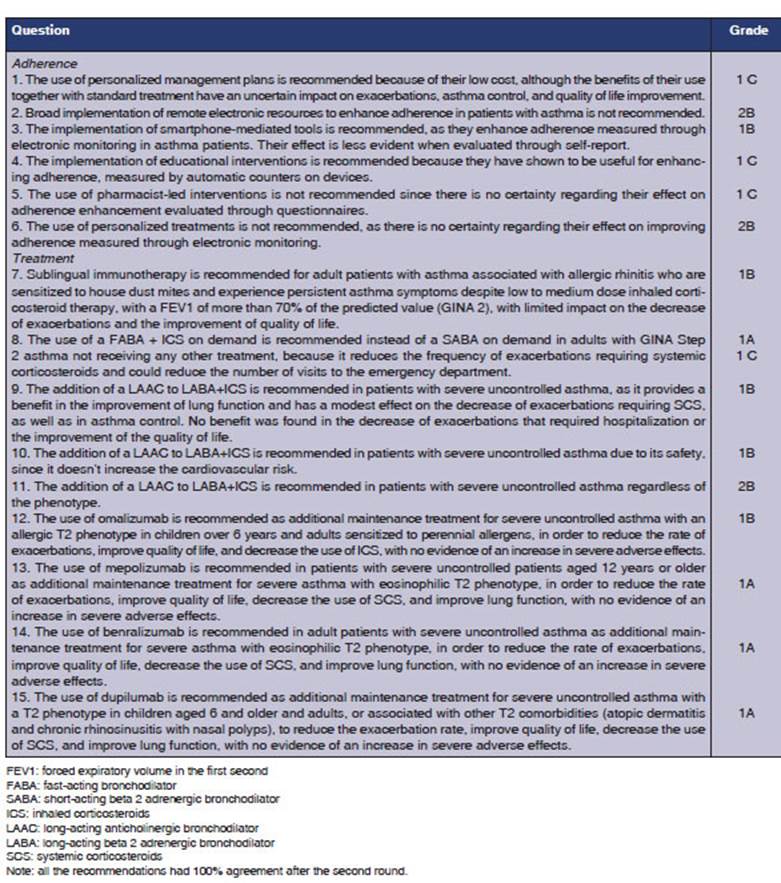
The convergence of both societies allowed for the analysis of common areas. Representatives with extensive experience in asthma management were selected from each society to choose questions that addressed common discussion points and were clinically relevant in our country regarding the diagnosis and treatment of asthma in adults. Twenty-eight specialists proposed twenty-two questions, assigning a score based on both conditions. These questions were then ranked from highest to lowest score, and the top 15 were analyzed (Table 1). For the purpose of preparing the manuscript, the questions were grouped based on the topics they covered (seven questions on adherence and eight on treatment), so that each question could be discussed and analyzed and the corresponding recommendations could be established (Figure 1).
Literature searches were conducted in the MEDLINE, EMBA SE, Cochrane, SciELO, and Lilacs databases until October 31, 2022, using search terms relevant to the respective questions. WE used the GRADE system (Grading of Recommendations, Assessment, Development, and Evaluation) of scientific evidence level of the publications of the American College of Chest Physicians (ACCP), and of the recently published level of recommendation.11 The levels of scientific evidence were characterized as A (strong evidence), B (moderate evidence), and C (low or very low evidence) according to the study design, the consistency of the results, and the clarity of the evidence to answer clinical questions. This system was chosen for its simplicity, transparency, explicitness, and consistency with the current methodological approach for the development of evidence-based good clinical practice. The recommendations were labeled according to the balance between risk, benefit, social and epide miological importance, and, in some cases, cost. Recommendations can be level 1 (mandatory) or level 2 (doubtful). For example, a 1A recommendation is a mandatory recommendation with strong scientific evidence, while a 2C recommendation is one with low scientific evidence, and is considered doubtful11.
The recommendations in response to each question were subjected to the agreement of a panel of nine specialists (four allergists and five pulmonologists) using the RAND/ UCLA modified Delphi consensus methodology.12-13 Agree ment on a recommendation was reached if a 75 % consensus was achieved. Each question and its recommendation were discussed in virtual meetings by the panel of specialists. If an agreement was not reached in the first round, a second round was conducted after a review of the literature and proposals one week later. All the recommendations reached a consensus exceeding 75 % within two rounds.
QUESTIONS AND RECOMMENDATIONS
About adherence
The World Health Organization (WHO) defines adherence as “the extent to which a patient’s use of medication corresponds to the prescribed regimen”.14 Patient behavior regarding treatment adherence is complex and diverse. It is widely recognized that non-adherence is very common in patients with asthma (30-70 %) and is motivated by numerous factors.1-3,9-10 The concept of poor ad herence primarily applies to the underutilization of daily preventive treatment.15-21 It is evident that poor adherence leads to an increase in morbidity, mortality, and use of healthcare resources. The psychosocial factors of the patient, inherent to the disease itself, the doctor-patient relationship, and access to medications have been extensively determined in the studies15-21. In the different defi nitions of “poorly controlled asthma,” assessing adherence problems and addressing them before labeling a patient as having severe asthma is a mandatory step in the recommendations of differ ent international and national guidelines.1-3,8-10 In clinical studies, it is necessary to ensure treatment adherence more than 80 % of the time, and this is achieved not only through patient self-reporting of medication intake but also by counting the doses of the drugs under investigation and using electronic dosing devices during each visit or through telemedicine. Therefore, it is likely that better asthma control can be achieved in these patients solely by improving adherence.21
Educating asthmatic patients is essential and recommended in every A evidence guideline as regards its benefits in reducing morbidity and improving adherence to treatment and follow-up.1-3,9,22-23
Due to its relevance, the initial questions about different interventions to enhance adherence and their respective recommendations were grouped together.
1. Question: What is the efficacy of adding a written treatment plan to the routine care for adult outpatients with asthma?
Justification:
All the international and local guidelines agree on the need for providing an asthma patient with a written treatment plan, despite its challenging implementation1-3,8-9,22-23 and the limited scientific evidence regarding the positive effects of having this tool in asthma management.
On one hand, having a written plan contrib utes to the inclusion of the patient as an active participant in their treatment and as the central figure in their own condition. Thus, it is expected that knowledge of the treatment and action plan allows the patient to have better control over the disease, providing them with more tools to promptly request a consultation if necessary. Additionally, a written plan raises awareness of the disease among the patients, reducing the undesirable effects caused by the low risk per ception associated with asthma. At this point, the importance of a written treatment plan is significant, as individuals diagnosed with asthma often underestimate their symptoms and the potentially fatal outcomes, especially in cases of poorly controlled asthma. 6-8
On the other hand, the practical implementation of a written treatment plan is considered highly feasible due to its low cost, and is also beneficial for healthcare personnel involved with asthma patients and caregivers of high-risk asthmatic groups.1-3,8-9,22-24
Lastly, a tool like the written treatment plan can help enhance adherence to asthma treatments, which is generally low, and its improvement is top priority.
Recommendation
The use of personalized management plans is recommended because of their low cost, although the benefits of using them in combination with standard treatment have an uncertain impact on exacerbations, asthma control, and improvement in quality of life (1C).
2. Question Should remote electronic re sources be used instead of the routine care for adult outpatients with asthma?
Justification
There is significant variability in the scientific evidence regarding the use of remote electronic resources for enhancing treatment adherence in asthma patients. The uncertainty arises from methodological heterogeneity in the studied popu lations and measurement methods, both of which affect the variability of the effect size.25
More specifically, there is high certainty for treatment adherence evaluated through electronic monitoring, low certainty for adherence evaluated through self-report25, and very low certainty for the adherence evaluated through pharmacy aerosol refill. So, evidence results are inconsistent and inconclusive regarding the actual effectiveness of using remote electronic resources to enhance adherence to asthma treatment.
Recommendation
Broad implementation of remote elec tronic resources to enhance adherence in patients with asthma is not recommended (2B).
3. Question. Should smartphone-based interventions be used instead of the routine care for adult patients with asthma?
Justification
The certainty about the positive relationship be tween the use of mobile phone-mediated tools and the improvement in adherence is high. However, certainty is moderate for the adherence evaluated through self-report.26
Recommendation
The implementation of mobile phone-mediated tools is recommended as they enhance adherence when measured through electronic monitoring in asthma patients. Their effect is less evident when evaluated through self-report (1B).
4. Question. Should education be used as an intervention to promote adherence instead of the routine care for adult outpa tients with asthma?
Justification
As previously mentioned, education is essential in the treatment and follow-up of asthma pa tients.1-3,9,22-23,27 In fact, the set of educational con tent directly or indirectly favors adherence. Among the relevant content that should not be omitted, there is the correct diagnosis, guidelines for early recognition of exacerbations and how to respond to them, tools to recognize different types of con trol and rescue pharmacological treatments, the importance of adherence, as well as the relevance of proper usage techniques, of reducing adverse events, seeking medical consultations promptly, and managing comorbidities correctly.1-3,9,22-23,27
Recommendation
The implementation of educational inter ventions is recommended because they have shown to be useful for enhancing adherence, measured by automatic counters on devices (1C).
5. Question. Should pharmacist-led in terventions be used to promote adherence instead of the routine care for adult outpa tients with asthma?
Justification
Problems with adherence to treatment and follow-up are quite common in asthma patients, and this can be attributed to multiple factors, as mentioned earlier. Many studies have inves tigated various types of interventions of phar maceutical personnel to evaluate the beneficial impact on adherence, but yielded inconclusive results. The certainty to improve adherence is very low for pharmacy aerosol refill and low for self-report.28
Recommendation
The use of pharmacist-led interventions is not recommended, since there is no cer tainty regarding their effect on adherence enhancement evaluated through question naires (1C).
6. Question. Should personalized treat ment be used as an intervention to promote adherence instead of the routine care for adult outpatients with asthma?
Justification
Due to contradictory published data regarding the use of mobile personalized action plans versus written action plans and the risk of bias, it is not clear at this time whether one format of action plan is superior to the other for both adolescents and adults.27
Recommendation
The use of personalized treatments is not recommended, as there is no certainty regarding their effect on improving adher ence measured through electronic monitor ing (2B).
About the treatment
7. Question Should pharmacological treat ment + sublingual immunotherapy (SL) be used instead of pharmacological treatment or placebo in adult patients diagnosed with allergic asthma?
Justification
The latest edition of the Global Initiative for Asthma (GINA), states that specific immuno therapy (SIT) for allergens can be a treatment option where allergy plays a prominent role, including asthma with allergic rhinoconjunctivitis.1 There are two forms of SIT: sublingual (SLIT) and subcutaneous (SCIT). While modest effects were identified in a systematic review of SLIT for asthma in adults and children, these effects are predominantly limited to oral and gastrointestinal symptoms.29 As with any treatment, the potential benefits of SLIT for individual patients should be weighed against the risk of adverse effects and the cost for both the patient and the healthcare system.1
On the other hand, in GEMA 2022, reference is made to SCIT with allergenic extracts as an effec tive treatment for well-controlled allergic asthma at low or moderate levels of treatment (therapeutic steps 2 to 4), provided that clinically relevant IgE-mediated sensitization to common aeroallergens has been demonstrated, well-characterized and standardized extracts are used, and the use of complex mixtures is avoided.1-2,30-33 However, many patients with mild intermittent asthma (step 1) concurrently suffer from moderate or severe al lergic rhinitis, which justifies the prescription of immunotherapy.34 SCIT should not be prescribed to patients with severe uncontrolled asthma, as its effectiveness is not well-documented and there is a high risk of suffering severe, even life-threatening adverse reactions.33,35 Therefore, it should be prescribed by specialized physicians with experi ence in this type of treatment and administered in facilities equipped with basic measures for the immediate treatment of a potential severe adverse reaction. The search for safer and more convenient alternatives for the patients has stimulated the study of the effectiveness of SLIT. Most clinical trials that demonstrated clinical efficacy have used well-characterized extracts at doses much higher than those typically used in SCIT. The tolerance profile of sublingual immunotherapy is optimal, with no fatal reactions reported.34,36 When sublin gual immunotherapy (SLIT) in oral lyophilized form for dust mites is added to the controlled maintenance pharmacological treatment, it is ca pable of reducing the number of moderate to severe exacerbations and improving disease control, with a very good safety profile.37 Therefore, its use is recommended in adult patients with moderately controlled or partially controlled asthma.34 If vari ous immunotherapy alternatives are available, pri ority should be given to those that have the status of registered medicines with well-established effi cacy, safety, and quality. At the moment, there are no comparative studies on the cost-effectiveness of immunotherapy versus conventional pharmacotherapy, and sure enough, such studies will not be conducted, as the complexity of their design makes them poorly viable. However, specific im munotherapy (SIT), in addition to controlling the manifestations of the disease, offers several ad ditional advantages over pharmacotherapy. These include maintaining clinical benefits obtained until several years after treatment cessation, reducing the risk of developing asthma in patients with allergic rhinitis, or preventing the development of new sensitizations in mono-sensitive patients 38-41. Furthermore, allergen immunotherapy has a unique immunological justification, as it tailors the approach to an individual’s specific IgE spectrum and modifies the natural course of the disease, with persistent efficacy after treatment completion. From this perspective, allergen immunotherapy (AIT) should currently be considered a prototype of Precision Medicine.42
Recommendation
Sublingual immunotherapy is recommend ed for adult patients with asthma associated with allergic rhinitis who are sensitized to house dust mites and experience persistent asthma symptoms despite low to medium dose inhaled corticosteroid therapy, with a FEV1 (forced expiratory volume in the first second) of more than 70 % of the predicted value (GINA 2), with limited impact on the decrease of exacerbations and the improve ment of quality of life (1B).
8. Question. Should a fast-acting broncho dilator (FABA) + inhaled corticosteroids (ICS) on demand be used instead of a short-acting beta-2 adrenergic bronchodilator (SABA) on demand in adults with GINA Step 2 asthma?
Justification
Based on new clinical information, in 2021 major international guidelines made changes to asthma treatment strategies.1-2 The recommen dation for fixed-dose inhaled corticosteroids in GINA Step 2 is still the treatment of choice, as it allows for better disease control and has consistent accessibility within the healthcare system.1 It’s important to note that the controlled, prospec tive, double-blind studies that have been analyzed were conducted with the fixed-dose combination of budesonide-formoterol.43-48 This treatment strategy has shown a high level of evidence in the decrease of exacerbations that require the use of corticosteroids (CS). However, it does not have the same level of evidence for other variables such as emergency department visits, improved quality of life, asthma control, and improved lung function.49
While the analyzed information may not strictly apply to patients included in GINA Step 1, it is understood that this strategy could be applied to such patients, with a lower level of evidence. As a strong recommendation in favor of using the budesonide-formoterol combination on demand, which is available in our country, the goal is to reduce the frequency of exacerbations that re quire systemic corticosteroids, likely leading to a decrease in the number of visits to the emergency department.
Recommendation
The use of a fast-acting bronchodilator + inhaled corticosteroids (FABA + ICS) on demand is recommended instead of a short-acting beta-2 adrenergic bronchodilator (SABA) on demand in adults with GINA Step 2 asthma not receiving any other treatment, because it reduces the frequency of exacer bations requiring systemic corticosteroids (1A), and could reduce the number of visits to the emergency department (1C).
9. Question. Should long-acting anticho linergic bronchodilators (LAACs) + ICS + long-acting beta-2 adrenergic bronchodila tors (LABA) be used instead of LABA + ICS for adult patients with severe uncontrolled asthma?
Justification
Anticholinergic bronchodilators were among the first pharmacological groups used to treat asthma, as a natural component of belladonna. Beyond their bronchodilator action, long-acting anticho linergics have anti-inflammatory effects through both neuronal and non-neuronal route, acting on inflammatory cells and molecules.50 Tiotropium is the LAAC with the largest amount of clinical information and has been studied in children, ado lescents, and adults.51 Two other LAACs (glycopyr ronium and umeclidinium) have been investigated in the CAPTAIN, IRIDIUM, TRIMERAN, TRIG GER, and ARGON studies, evaluating the clinical impact of the triple therapy in a single inhaler containing three pharmacological groups (LAAC, LABA, and inhaled corticosteroids) in patients with moderate and severe asthma not controlled with ICS/LABA.52-55 There are some differences in the evidence related to the duration of action of each drug, as well as the quantity and quality of available studies and the specific combinations of LABA+LAAC+ICS.
In an extensive review that included some studies with all three LAACs (11,894 children and adults; mean age: 52 years [range, 9-71 years]; 57.7 % women), the main objectives assessed were severe exacerbations, asthma control (measured by the Asthma Control Questionnaire, ACQ-7), quality of life (measured using the Asthma Quality of Life Questionnaire, AQLQ), mortality, and ad verse events.56 The results obtained demonstrated (with high certainty) that triple therapy versus dual therapy (LABA+ICS) in a single device once-daily was significantly associated with improved lung function (high certainty) and reduced risk of exacerbations requiring systemic corticosteroids (SCS) (moderate certainty), or hospitalization (low certainty).56 There were no significant differences regarding the quality of life (high certainty of the evidence) or mortality (high certainty of the evi dence) between dual and triple therapy.56
Triple therapy was significantly associated with an increase in dry mouth and dysphonia, and for serious adverse events, there was no difference between the groups, including cardiovascular events (moderate certainty of the evidence).52-55,57-61
One of the benefits of a fixed triple therapy ver sus an open one could be better treatment adher ence, as it would reduce the number of inhalers a patient needs to use, as well as the number of doses. Despite these results and other previous studies with another fixed triple therapy (TRI MARAN and TRIGGER), more studies are needed to confirm these improvements, especially with regard to exacerbations.53-55
Most safety studies were conducted in patients with COPD (higher mean age, and higher number of concomitant diseases of greater severity). In asthma, only one study on cardiovascular effects has moderate certainty of the evidence.61-62
According to all asthma management guide lines, patients with severe asthma should be phe notyped.63-64 Regarding the therapeutic approach in this stage, the efficacy of the LAACs is independent of the asthma phenotype, irrespective of the eo sinophilia degree and the fraction of exhaled nitric oxide (FeNO).65-66 The addition of LAACs could be considered for patients with persistent bronchial obstruction, symptomatic patients, and patients who are not frequent exacerbators (low certainty of the evidence).52-55
The GINA guideline recommends their use in patients who continue to have exacerbations despite intensive treatment with two controllers (inhaled corticosteroids and LABA), at step 4 or 5.1 The GEMA (Guía Española para el Manejo del Asma) guideline suggests the use of LAACs from step 4 and 5 in combination with ICS and LABA.2 The ATS/ERS (American Thoracic Society/Euro pean Respiratory Society) guideline recommends them in children, adolescents, and adults with severe uncontrolled asthma regardless of the GINA step 4/5 controller treatment.63
Recommendation
The addition of a LAAC to LABA+ICS is recommended in patients with severe uncon trolled asthma, as it provides a benefit in the improvement of lung function and has an effect on the decrease of exacerbations re quiring corticosteroids, as well as in asthma control. No benefit was found in the decrease of exacerbations requiring hospitalization or the improvement of quality of life (1B).
10. Question Is it safe to use LAAC+ICS+LABA in adult patients with severe uncontrolled asthma?
Justification
The rationale for this question is based on question 9.
Recommendation
The addition of a LAAC to LABA+ICS is recommended in patients with severe uncontrolled asthma due to its safety, since it doesn’t increase the cardiovascular risk (1B).
11. Question Should LAAC+LABA+ICS be used according to the phenotype for adult patients with severe uncontrolled asthma?
Justification
The rationale for this question is based on question 9.
Recommendation
The addition of a LAAC to LABA+ICS is recommended in patients with uncontrolled severe asthma regardless of the phenotype (2B).
Biologics
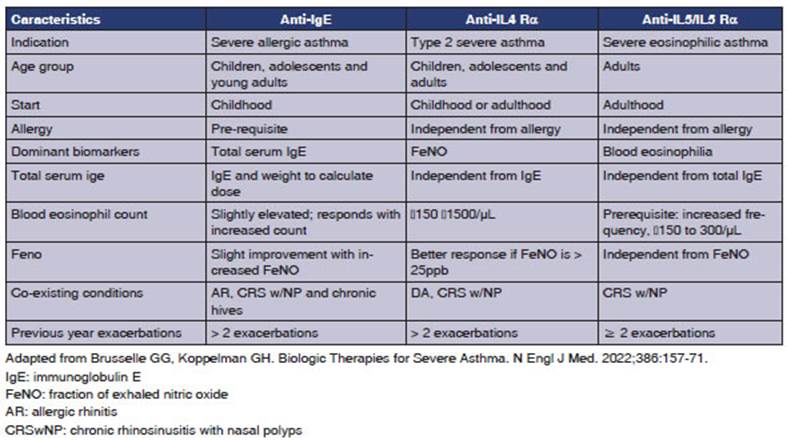
Severe asthma constitutes 3 to 5 % of the population with asthma. It is characterized by the persistence of symptoms, higher number of visits to emergency rooms or unscheduled outpatient consultations, more hospitalizations, an increased use of rescue medication, systemic cor ticosteroids, antibiotics, and the resulting impact on the increased use of healthcare resources and increased mortality.63-64 Severe asthma represents a heterogeneous syndrome with multiple clinical variants. Over the past two decades, it has been intensely studied, and different phenotypes have been defined.67-69 Establishing the asthma pheno type in patients with severe uncontrolled asthma is part of the diagnosis and evaluation of these individuals, since it can lead to differential treat ment and have prognostic implications. 63-64,67-69 Two inflammatory phenotypic patterns have been defined: T2-high (present in allergic and eosino philic asthma) and non-T2, also called T2-low. Both T2-high phenotypes often show some degree of overlapping. The fraction of exhaled nitric oxide, eosinophilia, and IgE are good biomarkers for the T2-high phenotype. Allergic T2 asthma represents 40-50 % of severe asthma and has an atopic basis orchestrated by the activation of T helper type 2 cells (Th2), the production of interleukins IL-4, IL-5, and IL-13, and isotype switching in B lym phocytes towards IgE production. Eosinophilic T2 asthma represents more than 25 % of severe asthma and is characterized by the presence of eosinophils in bronchial biopsies and sputum, even in patients receiving high doses of glucocorticoids. It may be associated with chronic rhinosinusitis and nasal polyps.63-64,67-69
The following questions and their respective recommendations are related to the use of biolog ics in severe asthma.
12. Question. Should omalizumab be used instead of placebo in patients with severe uncontrolled asthma with a T2 phenotype?
Justification
Omalizumab is a humanized monoclonal anti- IgE antibody (Mab) that binds to free IgE, pre venting its attachment to mast cell and basophil receptors, and it can also reduce IgE receptors on effector cells.63-64
Omalizumab is indicated for type 2 allergic asthma with total IgE values between 30-1,500 IU. The dosage varies depending on the IgE level and body weight.63-64
The quality of the evidence is high, the mag nitude of beneficial effects is moderate, and the magnitude of adverse events (AEs) is low. Therefore, the benefit-risk ratio favors the use of omalizumab.63-64,71-76
The evidence is moderate regarding the decrease of exacerbations requiring SCS and the improve ment of FEV1.63-64,71-76 The evidence is low for the decrease of exacerbations requiring emergency care and hospitalization and for the improvement of the ACQ.63-64,71-76
There are no cost-effectiveness studies in our country, but despite the high cost, most probably the benefit-risk ratio favors the use of the drug.
Recommendation
The use of omalizumab is recommended as additional maintenance treatment for severe uncontrolled asthma with an allergic T2 phenotype in children over 6 years and adults sensitized to perennial allergens, in order to reduce the rate of exacerbations, improve quality of life, and decrease the use of SCS, with no evidence of an increase in severe adverse effects (1B).
13. Question. Should mepolizumab be used instead of placebo in adult patients with severe uncontrolled asthma with a T2 phenotype?
Justification
Mepolizumab and reslizumab are both IL-5 inhibitors, but only mepolizumab is commercially available in our country.70
The quality of the evidence is high, the magnitude of beneficial effects is moderate, and the magnitude of adverse events is low. Therefore, the benefit-risk ratio favors the use of mepolizumab.70,77-81
The evidence is of high quality regarding the decrease of exacerbations requiring SCS and those requiring emergency care or hospitaliza tion.70,77-81 The evidence is moderate regarding the improvement of the ACQ and the improvement of FEV1.70,77-81
There are no cost-effectiveness studies in our country, but despite the high cost, most probably the benefit-risk ratio favors the use of the drug.
Recommendation
The use of mepolizumab is recommended in patients with severe uncontrolled asth ma aged 12 years or older as additional maintenance treatment for severe asthma with eosinophilic T2 phenotype, in order to reduce the rate of exacerbations, improve quality of life, decrease the use of SCS, and improve lung function, with no evidence of an increase in severe adverse effects (1A).
14. Question. Should benralizumab be used instead of placebo in adult patients with severe uncontrolled asthma with a T2 phenotype?
Justification
Benralizumab is an inhibitor of the IL-5 re ceptor α.70 The quality of the evidence is high, the magnitude of beneficial effects is moderate, and the magnitude of adverse events is low.82-87 Therefore, the benefit-risk ratio favors the use of benralizumab.82-87
The evidence is high regarding the decrease of exacerbations requiring SCS and the improvement of FEV1.82-87 The evidence is of high quality regard ing the improvement of quality of life (ACQ) and the decrease of exacerbations requiring emergency care or hospitalization.82-87
There are no cost-effectiveness studies in our country, but despite the high cost, most probably the benefit-risk ratio favors the use of the drug.
Recommendation
The use of benralizumab is recommended in adult patients with severe uncontrolled asthma as additional maintenance treat ment for severe asthma with eosinophilic T2 phenotype, in order to reduce the rate of exacerbations, improve quality of life, decrease the use of SCS, and improve lung function, with no evidence of an increase in severe adverse effects (1A).
15. Question. Should dupilumab be used instead of placebo in patients with severe uncontrolled asthma with a T2 phenotype?
Justification
Dupilumab is an inhibitor of the IL-4 receptor α subunit, which interferes with the action of both IL-4 and IL-13.88
The quality of the evidence is high, the mag nitude of beneficial effects is moderate, and the magnitude of adverse events is low.88-93 Therefore, the benefit-risk ratio favors the use of dupil umab.88-93
The evidence is of high quality regarding the decrease of exacerbations requiring SCS and those requiring emergency care or hospitalization, and also regarding the improvement in ACQ and the FEV1.88-93
There are no cost-effectiveness studies in our country, but despite the high cost, most probably the benefit-risk ratio favors the use of the drug.
Recommendation
The use of dupilumab is recommended as additional maintenance treatment for severe uncontrolled asthma with a T2 pheno type in children aged 6 and older and adults, or associated with other T2 comorbidities (atopic dermatitis and chronic rhinosinus itis with nasal polyps, CRSw/NP), to reduce the exacerbation rate, improve quality of life, decrease the use of SCS, and improve lung function, with no evidence of an in crease in serious adverse effects (1A).
GENERAL CONCLUSION REGARDING BIOLOGICS (TABLE 3)
Severe uncontrolled asthma is associated with a reduced quality of life, increased exacerbations, hospital admissions with frequent use of systemic corticosteroids, and elevated death risk.63-64 Pheno typing patients with severe uncontrolled asthma (SUA) is necessary to prescribe the precise biologic therapy for each phenotype.63-64
Biologics targeting type 2 inflammation have shown improvement in disease control when used as additional therapy alongside maintenance treat ment in patients with SUA (Step 5 according to GINA, Step 6 according to GEMA).1-2,63-64,94
FINAL CONCEPTS
The modified Delphi methodology is a well-defined technique for reaching a consensus among special ists in areas of uncertainty, and it is particularly useful for making decisions in medical situations where scientific evidence is scarce or nonexis tent.13-14 One of the strengths of this document is that it achieved 100 % agreement among the participants within two rounds. Another strength is that the specialists were selected by the two scientific societies for their expertise in the topics being discussed. The Delphi technique suggests that the participation of up to twelve specialists is sufficient and recommended.13-14 All authors had the opportunity to vote freely and express their opinions during discussion moments. This manuscript also has some limitations. The absence of participation from clinical physicians may, to some extent, limit the perspective regarding adherence or asthma management, especially in milder forms of the disease. Another limitation is that the specialists’ opinions, as reflected in the selection of the supporting literature or their own experience in the field, may not encompass all the published evidence in the area. Furthermore, it has limited temporal validity and may change with the emergence of new scientific information. It should be interpreted rationally and complemented in the future with further research, especially within the context of areas of greater uncertainty.
In conclusion, this document provides recom mendations based on expert opinion and grounded in scientific evidence with regard to the importance of enhancing adherence to treatment and follow-up through different asthma management strate gies, especially given the frequent poor control of asthma in our country.7-8 It also provides updated recommendations on the critical aspects of the treatment of mild to severe asthma.
KEY POINTS
Current knowledge
Despite the advances in asthma management and pre ventive treatment that improve quality of life and reduce morbidity and mortality, our country still has poor asthma control and an unacceptable rate of hospitalizations and mortality.
Contributions of the article to current knowledge:
Specialists from two medical societies committed to ta king actions to improve asthma control in our country have made locally adapted recommendations in various critical aspects of asthma management and treatment.