INTRODUCTION
Dental caries has multifactorial etiology involving exposure to acid due to bacterial metabolism 1-3 . It is the most prevalent oral health problem throughout the world, causing a considerable economic burden and a negative impact on quality of life 2 , 4 . As human enamel cannot be biologically repaired or replaced 5 , several studies have been conducted in search of means to protect it by improving techniques and products to ensure better prevention and remineralizing capacity 6-9 .
Fluoride is the main factor responsible for the decline in dental caries in recent decades 10 , 11 . Indeed, fluoride dentifrices are widely used and effective at delivering free or soluble fluoride to the oral cavity 12 .
Recent progress has been made in systems for promoting enamel remineralization 6 , 9 . In vitro studies have contributed to the understanding of the chemistry the incorporation of fluoride and its compatibility with the ingredients in dentifrices, and have tested new formulations and mechanisms of caries inhibition 1 , 13 . The anti-cariogenic role of fluoride has been proven 11 , 14 , 15 . Fluoridated hydroxyapatite 2 (mineral crystal of dental tissue) has greater chemical stability and therefore better protects the teeth against acid attacks from cariogenic bacteria and consequent demineralization 1 , 2 .
Thus, local therapies using drug delivery systems have attracted considerable attention as a way to transport and release therapeutic agents or bioactive substances into the oral cavity 16 .
The use of nanotechnology has gained prominence in preventive dentistry as a slow-release system for substances that can control and treat oral problems 17-21 . Involving components less than 100 nanometers in size 22 , this technology may perform on the molecular level with greater affinity and effectiveness compared to micro or macro components 18 , 23 . In preventive dentistry, studies have reported better control of biofilm, prevention of caries, remineralization of initial carious lesions and sub-micrometric dental defects, biomimetic enamel synthesis and repair of microcavities 17-20 , 23 , 24 . The innovative idea of using nano-encapsulated fluorine ions (NanoF) in a controlled release system may be beneficial to enamel, as it would increase its substantivity and enable the substance to remain in contact with the dental surface for a longer time, leading to a greater protective and remineralizing effect. Therefore, the aim of the present study was to evaluate the influence of treatment time on the performance of nano-encapsulated fluoride (NanoF) dentifrices for the remineralization of initial carious lesions using an in vitro pH cycling model. The null hypothesis was that NanoF dentifrices have the same ability to remineralize dental caries as conventional NaF, regardless of treatment time.
MATERIALS AND METHODS
This study received approval from a research ethics committee in Brazil (certificate number: 45917915.6.0000.5188). The donors of the teeth signed a statement of informed consent in accordance with the Declaration of Helsinki and Resolution 466/12 of the Brazilian National Health Board.
Sample size and specimen preparation
Sample size was determined considering remineralizing potential as the primary outcome. Based on a previous study25 and considering a twotailed alpha of 0.01 and 80% power, a minimum of seven samples was required per group. Ninety human third molars with indication for extraction were collected, cleaned, and examined for any enamel alterations.
Enamel blocks (3 x 3 x 2 mm) were obtained from the dental crown (buccal and lingual surfaces) and embedded in self-curing acrylic resin using circular molds (16 x 3 mm). A metallographic polisher was used to flatten the outer enamel (with 400, 600 and 1200 grit sandpaper disks) under constant irrigation. The enamel surfaces were polished with wet felt and a 1 pm diamond suspension (Extec Corporation, Enfield, CT, USA) in a rotating polishing machine (PSK- 2V, Skill-Tec Comercio e Manutenqao Ltda, Sao Paulo, SP, Brazil). All samples were submitted to water sonication for five minutes and stored at -20°C until the day of the experiment.
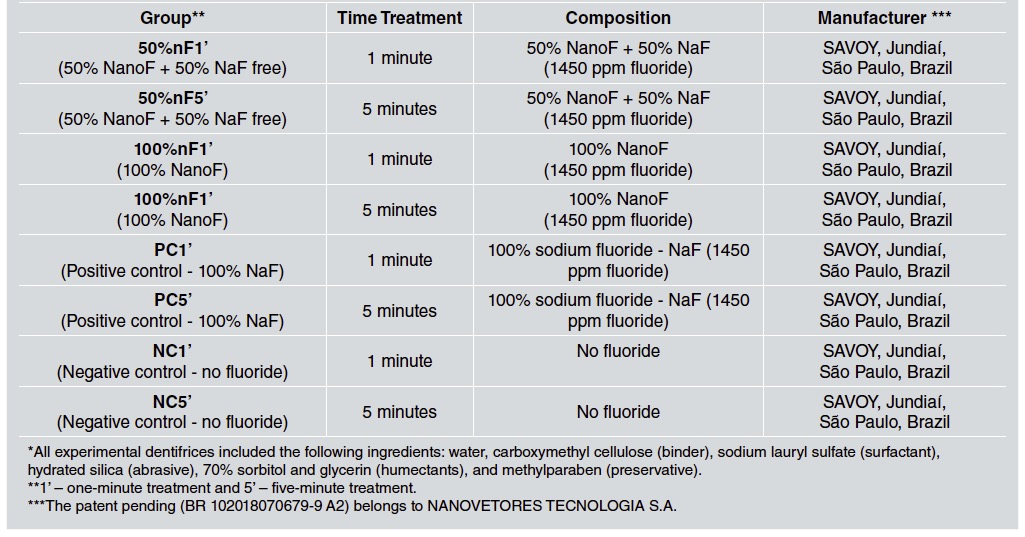
A total 96 enamel specimens were randomly distributed into eight groups (n=12) according to their initial surface microhardness and time treatments: PC1’(dentifrice with 100% NaF fluoride, 1 minute); PC5’(dentifrice with 100% NaF fluoride, 5 minutes); NC1’ (dentifrice without fluoride, 1 minute); NC5’ (dentifrice without fluoride, 5 minutes); 50%nF1’ (dentifrice with 50% NanoF + 50% NaF free, 1 minute); 50%nF5’ (dentifrice with 50% NanoF + 50% NaF free, 5 minutes); 100%nF1’ (dentifrice with 100% NanoF, 1 minute) and 100%nF5’ (dentifrice with 100% NanoF, 5 minutes), coded by an independent researcher, as described in Table 1. Enamel surface microhardness was analyzed with a microhardness tester (Shimadzu HMV - AD Easy Test Version 3.0). Five indentations spaced 100pm from each other were made at the center ofthe enamel surface (Vickers, 100g, 10s). Firstly, a baseline measurement (SH0) was performed, and then new measurements were taken after the formation of the carious lesion (SH1) and remineralizing treatment (SH2), using the same initial parameters. Enamel blocks with initial surface microhardness between 390 and 440 VHN were selected for the study.
Induction of caries lesion
Following the SH0 measurements, the enamel surface of each specimen was divided into three equal parts. One third of the exposed enamel was covered with a double layer of nail varnish (Risque, Niasi, Taboao da Serra, Sao Paulo, Brazil) to preserve a control area in each specimen. Subsurface enamel demineralization was induced in the other two thirds by immersing each enamel specimen in 32 ml of demineralizing solution [1.3 mM/L Ca(NO3)2.4H2O, 0.78 mM/L NaH2PO4H2O in 0.05 M/L acetate buffer, 0.03 √gF/mL (NaF), pH 5.0] for 16 h at 37°C26. Post-demineralization surface hardness (SH1) was determined, and the percentage of surface hardness change (SMHC) was calculated [% SMHC= (SH1 - SH0/ SH0) x 100].
Collection of human saliva
Stimulated human saliva was collected from 12 healthy individuals 18 to 35 years old, of both sexes, without system involvement and resident in a city without fluoridated water. Exclusion criteria were having used drugs that interfere in the salivary flow or fluoride products within the last four weeks; using orthodontic appliances; presence of caries and/or erosion lesions and/or periodontal disease. Stimulated human saliva was collected each day of the study. Each volunteer chewed paraffin wax and spat the stimulated saliva into a plastic cup. The samples were stored in a refrigerator at 5 °C until use27.
Remineralizing pH cycling
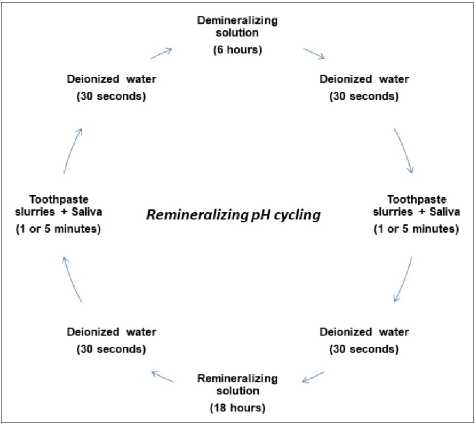
Before the remineralization pH cycling model based on Vieira et al.28, another third of the surface of the enamel specimens was covered with two layers of nail varnish (Risque, Niasi, Taboao da Serra, Sao Paulo, Brazil), to provide a carious lesion reference area (SH1). In each cycling model (Fig 1), the specimens were submitted to a five-day28 pH cycling model at 37°C, and remained in the remineralizing solution for 2 days. The blocks were individually immersed in a remineralization solution (1.5 mmol/L Ca, 0.9 mmol/LP, 150 mmol/L KCl, 0.05 mg F/mL in 0.1 mol/L Tris buffer, pH 7.0) for 18 h. The cariogenic challenge was performed using a demineralizing solution (0.05 mol/L acetate buffer, pH 5.0, containing 1.28 mmol/L Ca, 0.74 mmol/ LP, and 0.03 mg F/mL) for six hours per day. The treatment regime consisted of immersing the enamel samples in dentifrice slurries twice a day (1:3 w/w; 2 mL/enamel specimen) for one or five minutes under agitation. The treatments were performed before and after the demineralizing solution. In order to activate the salivary amylase of the NanoF groups, a volume of approximately 10% of human saliva was added to all the dentifrice slurries. Samples were rinsed with deionized water between steps. The demineralizing and remineralizing solutions were refreshed every day. All eight groups were submitted to the same pH cycling, differing only in treatment times (one or five minutes). At the end of the seventh day of pH cycling, the specimens were washed again with deionized water under sonication for 5 min and stored in a humidity-controlled environment to prevent drying until further analysis.
After the remineralizing pH-cycling, the nail varnish was removed and enamel surface microhardness was again determined (SH2) as baseline parameters. The values were averaged (pm) and the percentage surface microhardness recovery was calculated as following formula: %SMHR = (SH2-SH1/SH0-SH1) x 100.
Statistical analysis
Data were statistically analyzed using the SPSS, version 21.0 (SPSS, Inc., Chicago, IL, USA). Normal Distribution and homogeneity of variances were tested using the Shapiro-Wilk test and
Levine test, respectively. Since data demonstrated homogeneity of variances and Gaussian distribution, no data transformation was necessary. Assumptions of analysis of variance were checked before the use of two-way factorial analysis of variance (ANOVA) model to investigate surface microhardness remineralization in the different groups for the dependent variable (%SMHR), and ANOVA Repeated Measures, followed by Bonferroni, to analyze the variables SH0, SH1, SH2 within the same group at the different analysis times. The level of significance considered was 5% (p<0.05).
RESULTS
Table 2 shows the mean and standard deviation values for the variables SH0, SH1, and SH2 for all test groups. Comparison according to the variables SH0, SH1 and %SMHC showed no difference between groups (p> 0.05). However, for the SH2 variable, differences were observed between groups (p <0.001). The 5-minute PC had the best remineralization value and the 50%nF1’, 50%nF5’ and 100%nF5’ performed similarly to the 1-minute positive control.
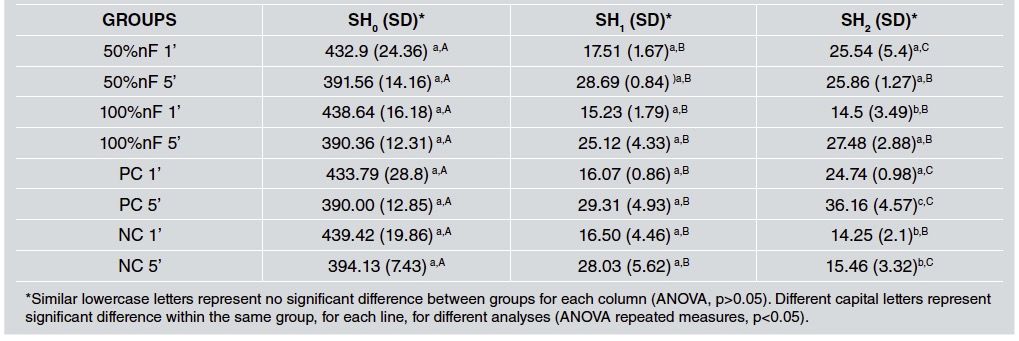
Table 2 Mean and standard deviation (SD) of SH0, SH1 and SH2 variables of all the groups, according to the different treatment times (one and five minutes)
Individual analysis of each dentifrice remineralization showed no significant difference between SH0 and SH1. However, there were differences between the SH1 and SH2 variables, for the 50%nF1’, PC1’, PC5’ and NC5’ groups (ANOVA repeated-measures, p<0.05).
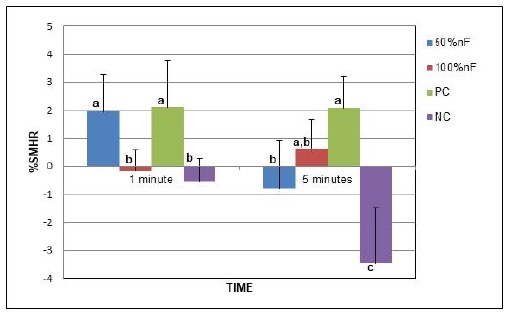
Fig. 2 shows the comparison of the groups using the percentage surface microhardness recovery (%SMHR) variable. Differences were observed between groups (p<0.05). The PC groups presented the best remineralization values, indicating the same pattern observed for the SH2 variable. The 50%nF1’ and 100%nF5’performances were similar to the PC groups (1’ and 5’). No remineralization recovery was detected in the negative control (1 or 5 minutes) or in the 50%nF5’ groups.
DISCUSSION
Studies in the field of cariology have fostered progress in the treatment of dental caries29. Nano-scale drug delivery systems have gained ground in this respect and nanotechnology has been investigated as a means to enhance fluoride substantivity in the oral environment13,30,31, prolonging its effect with a small amount of product32-34. Thus, nano-encapsulated fluoride systems may constitute a promising treatment for carious lesions. In the present study, the nano-encapsulated fluoride (NanoF) system was created as an innovative method to act as a controlled release system to increase fluoride substantivity and bioavailability in the oral cavity. The composition of NanoF is basically sodium fluoride (NaF) encapsulated in a natural biopolymer (hydroxypropyl guar) as the drug carrier. Guar gum is a molecular weight carbohydrate polymer derived from the natural seed of the guar plant (Cyamopsis tetragonoloba (L.) Taub). Its particle size is about 200 nm. Fluoride release is not dependent on pH, but on enzymatic trigger (amylase) following a mechanism of stimuli-controlled release, which is why fresh human saliva was added to the pH cycling model. Also, a previous clinical study35, using the same dentifrices, reported a delay in the fluoride release in saliva within the first 60 minutes after brushing and reported that dental biofilm showed a more prolonged incorporation of fluoride ions when compared to conventional NaF dentifrice35.
It was important to study treatment time (1 or 5 minutes) because the NanoF controlled-release dentifrices were created to deliver fluoride ions more slowly. As expected, there were differences in the potential for remineralizing carious lesions according to treatment time. Also, as the literature lacks studies involving the same NanoF, we suggested some hypotheses to explain the role of these dentifrices in the process of remineralization of the decayed enamel.
Fluoride is known for its anti-caries and remineralizing action in its free form and at appropriate concentrations, with a high salivary dose-response level36. As fluorine is an electronegative element and thus highly reactive, the pH, ionic concentration, and/or the composition of the vehicle may modulate its action36. Based on this concept and the similar remineralization values found in the 50%nF1’ and PC1’ groups, it was evident that the free NaF was responsible for the initial performance before NanoF had had sufficient time to exert an effect. However, the unsatisfactory remineralizing performance observed for the 50% nF group after five minutes of treatment may have been due to the solvation of fluoride. As the release of NanoF occurred gradually, complexation may have occurred between conventional NaF and the gradually released NanoF, preventing the effective performance of the fluoride in the remineralization process. Thus, it can be said that the 50% nF5’ dentifrice behaved similarly to a dentifrice with a low fluoride content.
Analysis of the 100% nF groups showed that the double molecular polymer of the NanoF required a longer treatment time to release fluoride ions more effectively. During the one-minute treatment, the dentifrice resulted in the absence of protective/ remineralizing action. However, an increase of about 75% in %SMHR was found for the five-minute treatment, indicating the importance of the time factor for the release of NanoF. No significant difference was found between the 100%nF5’ and PC groups, despite the lower value for %SMHR.
A higher concentration of fluoride increases its capacity to diffuse through biofilm and exert an effect on the dental surface, promoting a gain in remineralizing capacity37. Nevertheless, a low concentration of fluoride may promote remineralization, provided that it is employed in an acidic vehicle35,38,39. Thus, the poor remineralization found in the 50%nF5’ and 100%nF1’ groups may also be explained by the neutral pH of the dentifrices used. Treatment time, an acidic environment, and sites of greater fluoride concentration are important factors. Promising results were found in a randomized clinical trial using the same experimental dentifrices, which demonstrated an increase in fluoride substantivity, enabling higher concentrations in dental biofilm over time35,38,39. When fluoride is presented only in saliva, dilution and swallowing lead to the return to the initial salivary concentration after 40 to 60 minutes of contact with the dentifrice. Thus, the incorporation of fluoride into the biofilm as a reservoir for subsequent release is more desirable for effective remineralization.
A major problem associated with the delivery of fluoride is the short time of action21. In the present study, salivary amylase is believed to have broken down the polymer, releasing the nanofluoride for fast, lasting action. Based on these findings, NanoF is a promising nano-encapsulated fluoride system that can be adjusted to promote faster fluoride release, achieve optimal salivary levels, and enable adhesion to biofilm with a prolonged performance.