INTRODUCTION
Adhesive bonding of fiber-reinforced resin posts is a usual procedure in the rehabilitation of endodontically treated teeth 1 .
Bonding to root dentin is complex, and requires close contact between the resin cement and the dentin substrate. Root post space preparation with Gates Glidden and pilot burs produces a large amount of secondary smear layer made up of remains of endodontic sealer, gutta-percha plasticized by frictional heat, and inorganic components 2 . This thick smear layer covering root canal walls jeopardize the effective adhesion of the cement 3 , 4 , even more when self-etching resin-based cements are used with adhesive protocols that do not include rinses and are unable to penetrate it. Neither can acid-etch techniques always remove this smear layer successfully 1 , 2 . The absence of chemical bonding between the polyisoprene component of the guttapercha remaining on root canal walls following deobturation for the post and the methacrylate of the resin cement may compromise bonding to dentin 5 . Certain factors such as endodontic obturation technique and time of de-obturation / post preparation may modify the quantity of endodontic material remaining on the root canal walls. Thermoplastic obturation techniques such as the hybrid technique, systems with carriers or injected gutta-percha fill the irregularities of the root canal better than single-cone and lateral condensation techniques, but make it more difficult to remove material later from inaccessible areas 6 , 7 . Post space preparation can be immediate or delayed until after endodontic treatment. Immediate preparation provides greater benefits regarding control of apical seal and easier removal of remains of yet unhardened endodontic material 6 , 7 , 8 . Dentinal smear layer is often removed by means of a combination of a chelating irrigant solution such as EDTA plus a proteolytic solution such as sodium hypochlorite. However, most studies have shown that these irrigant solutions are unable to remove the smear layer completely, especially when used passively, without mechanical activation 9 . Different methods can be used to activate irrigant solutions and achieve mechanical cleansing of the secondary smear layer and obturation remnants. These include low-speed rotary brushes, ultrasonic endodontic tips, cotton-wrapped files with alcohol, polymer tips on sonic devices such as the EndoActivator (Dentsply Sirona) and more recently, a nickeltitanium instrument designed for final cleansing of endodontic preparation, called XP-endo Finisher (FKG Dentaire, Switzerland).
A conical or cylindrical rotary brush is an inexpensive, simple-to-use instrument. Its use is not limited to endodontics, and it can also be used for cleaning interproximal spaces during dental prophylaxis, mounted on a micromotor and conventional lowspeed contra-angle.
Passive ultrasonic irrigation, described by Weller in 1980 10 , uses a smooth, thin stainless steel file to activate the irrigant inside the root canal. This technique is based on the premise that the energy released by the instrument improves the physicochemical properties of irrigant solutions by cavitation and acoustic flow 11 .
The XP-endo Finisher file (FKG, Dentaire, Switzerland) has recently appeared on the dental market. It is made from a new nickel-titanium alloy that enables it to act inside the root canal by expanding up to 6 mms more than its diameter, thereby reaching all the anatomy. The file is straight in its M-phase at ambient temperature or when cooled. When exposed to body temperature (within the root canal) its shape changes due to its molecular memory, to A-phase, enabling it to access and clean various parts of the root canal such as isthmuses and resorptions that other instruments cannot reach. Comparative studies on the use of the XP-endo Finisher file for post-endodontic instrumentation report promising results 12 , 13 .
The aim of this study was to determine which of the three instruments used (passive ultrasonic irrigation, rotatory brush and XP-endo Finisher) is most efficient in removing the secondary smear layer from the post preparation space, independently of the irrigant solution.
MATERIAL AND METHODS
This was an ex vivo, controlled study. Forty human lower premolars that had been extracted for orthodontic or periodontal reasons were selected. Sampling was probabilistic and random.
Inclusion criteria: Lower premolars with total length 21 mm to 23 mm; apex fully developed; without carious lesions; root canal single, straight, patent to glide path preparation, not excessively oval (canal was considered excessively oval when radiograph showed vestibular-lingual and mesiodistal dimensions in the middle third with a ratio greater than 2:1).
Exclusion criterion: lower premolars with excessively oval anatomy.
Clinical crowns were sectioned 2 mm above the proximal cementoenamel junction. Conventional openings were made, root canal glide paths were prepared, and working length of each specimen was determined as 1mm before the apical foramen, using a #15 K-file. Root canals were instrumented with ProTaper Next system (Dentsply Maillefer, Ballaigues, Switzerland) up to file X3, irrigating each instrument change with 2 ml of 2.5% sodium hypochlorite solution. Final irrigation was done with 17% EDTA for 60 seconds and 2.5% sodium hypochlorite for 30 seconds, irrigating with 3ml distilled water in between. The apical thirds of the root canals were obtured with tapered guttapercha cones, AH Plus endodontic sealer (Dentsply Sirona, Switzerland) and warm vertical compaction technique with Calamus Dual (Dentsply Sirona, Ballaigues, Switzerland). Post spaces were prepared immediately with a Largo #1 bur (Dentsply Maillefer, Ballaigues, Switzerland) and #1, 2 y 3 calibration burs of the Macro-lock XRO (RTD, France) for the fiber posts, depth 12mm. Root canals were irrigated with 3ml of distilled water using a syringe and 1” Terumo 25G needles each time the instrument was changed. Samples were divided randomly into four groups (n=10) and labelled according to the subsequent cleaning protocol to be applied: Group C (control); Group PIU (passive ultrasonic irrigation); Group CEP (conical Rotoprox brush, Hager Werken, Germany) and Group XP (XP-endo Finisher, FKG, Dentaire, Switzerland).
Group C was irrigated with distilled water but no mechanical cleaning system was used after preparation with pilot burs. For the rest of the experimental groups, each selected instrument was used following the manufacturer’s instructions for 30 seconds with distilled water inside the root canal. For Group PIU, a fine stainless steel (white) spreader tip mounted on an adaptor for the Woodpecker ultrasound unit (China) was used at 20% power. For Group CEP, a brush mounted on a micromotor and contra-angle was used at low speed, with in-andout motion, against root canal walls. For Group XP, samples were instrumented in an oven at 37 ºC to simulate body temperature and allow the instrument’s nickel-titanium austenitic phase to be expressed. Then a Teflon roll was placed in each specimen to fill the root canal space, two grooves were carved in the proximal root faces using a diamond disc without invading the endodontic space, and the samples were frozen at -70 ºC for a week, in order to make it easier to section the teeth. The samples were split using a chisel and hammer in order not to create any new smear layer that might alter observations. Half of each sample was selected and a fine-tip marker was used to make marks beside the root canal at 3 mm (coronary third), 6 mm (cervical third) and 9 mm (apical third) from the coronary reference, to enable identification of the areas to be observed under scanning electron microscope. Microphotographs of the center of the root canal were taken of each third at 350X magnification.
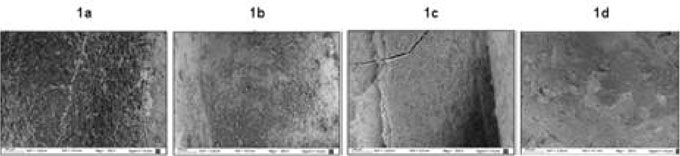
The following scores was applied for evaluation14 (Fig.1):
1: No smear layer.
2: Few areas covered in smear layer, with many open dentinal tubules.
3: Most areas covered in smear layer, with few visible open dentinal tubules.
4: All areas covered in smear layer.
The images were analyzed independently by two calibrated evaluators. If the evaluators disagreed, they analyzed the image together until they reached a consensus. For statistical analysis, values were calculated per root thirds and per group. Kruskall- Wallis and Friedman tests were applied. Significance level was established as p<0.05 for all cases.
RESULTS
Observation by SEM showed presence of dental smear in all samples, thereby corroborating the difficulty in removing secondary smear layer from root canal walls by means of all the systems tested.
In Group C (Fig. 2 a, b, c), there was a thick layer of amorphous smear layer completely covering the observation area in all three root thirds. In Group PIU (Fig. 3 a, b, c), there was smear layer covering the entire dentin surface with presence of scratch marks in different directions on the cervical and middle thirds (Fig. 3 a, b). In Group XP, most areas were covered with smear layer and there were few visible open dentinal tubules (Fig. 4 a, b, c). In Group CEP (Fig. 5 a, b, c) there was a thin, uniform, adhered smear layer covering the observation area, with a few visible open dentinal tubules and some light scratch marks in the apical third (Fig. 5c).

Fig. 2 Scanning electron microphotographs (350X) for observation of secondary smear layer removal in the control group (no mechanical cleaning).a: cervical third, there is a dense, irregular smear layer covering the dentin surface entirely. b: middle third, persistence of a thick, amorphous smear layer with many debris particles of different sizes hiding dentin tubule openings. c: apical third with dense smear layer covering the entire area, with presence of loose debris particles and longitudinally oriented grooves.

Fig. 3 Scanning electron microphotographs (350X) for observation of secondary smear layer removal in Group PIU (passive ultrasonic irrigation).a: cervical third, there is a thick, even smear layer completely covering the dentin surface. b: middle third, scratches on root canal walls. c: apical third, with persistence of a thin smear layer and a few small debris particles, approximately 20μm across, scattered over the surface.

Fig. 4 Scanning electron microphotographs (350X) for observation of secondary smear layer removal in Group XP (XP-endo Finisher).a: cervical third: presence of smear layer covering dentin surface, scattered particles and a few free areas where dentin tubule openings are visible. b: middle third, persistence of smear layer, less loose debris and openings of some dentin tubules visible. c: apical third, many dentin tubules open and some areas covered in smear layer.

Fig. 5 Scanning electron microphotographs (350X) for observation of secondary smear layer removal in the Group CEP (Rotoprox conical brush).a: cervical third, presence of a smear layer of even thickness completely covering the dentin surface with a few small, scattered debris particles. b: middle third, mostly areas with open dentin tubules, with presence of debris particles approximately 25μm across scattered on the surface. c: apical third: thinned smear layer and some slight scratches, close together
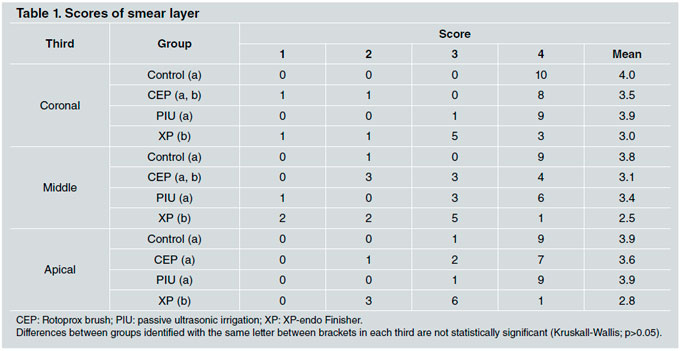
Data and mean values are provided in Table 1.
In Groups C and XP, no statistically significant difference was found between thirds (p>0.05). Within Group CEP, there was a significant difference between the middle third and the apical third. Within Group PUI, there was a significant difference between the middle third and the coronary third.
There were statistically significant differences between Groups XP and C for all thirds. However, the difference was not statistically significant between Groups CEP, PIU and C, although a trend to better removal of smear layer was noted in Group CEP, for all thirds. There was no significant difference between groups CEP and XP for the middle and coronary thirds (p>0.05) (Figs. 2-5).
In some samples, there were light scratch marks on the root canal surface. This was noted in all experimental groups, most frequently in the apical and middle thirds. Scratch marks were found, in descending order, in Groups PIU, CEP, and XP, with the same amount in Groups XP and C (Figs. 2c, 3b, 5c).
DISCUSSION
When luting a fiber post, it is essential to have clean dentin walls free from debris and endodontic filling remnants. It is therefore recommendable to implement a combination of mechanical and chemical cleaning actions prior to cementing 1 , 9 . This is even more important when dealing with oval-shaped canals such as those in single-rooted premolars and canines, which have more pronounced elliptical and irregular shape towards the free faces of the tooth, resulting in areas that retain endodontic sealer and gutta-percha 14 .
The current study used distilled water instead of irrigants in order to evaluate the mechanical action only – without the chemical action of irrigants – for each system tested. In this regard, all the systems evaluated presented difficulty in completely removing the smear layer from all thirds.
In this scenario, the XP-endo Finisher achieved the best cleaning results in post space preparations and was efficient in all root thirds, even in the apical third, which is so difficult to access due to the small diameter of the post space. Thus, our studies agree with Sanabria 11 , who reports that the XP-endo Finisher works better than ultrasound to remove smear layer from endodontic preparations. The XP-endo Finisher is a thin, flexible instrument designed specifically for final cleaning during surgical preparation and it reaches small, constricted areas with diameters smaller than spaces prepared for root posts. In contrast, Azimian 15 reports that the XP-endo Finisher combined with EDTA and sodium hypochlorite used after the final irrigation during surgical preparation is not superior to the same protocol without activation, thereby dismissing the mechanical action of the instrument per se.
In agreement with Coniglio 2 , our study found very similar values between Groups PUI and C in all root thirds when distilled water was used. Consideration should be given to ultrasound intensity, which manifests in the oscillation amplitude of the instrument, application time, volume and concentration of irrigant. Although ultrasound has demonstrated good results in removing debris in combination with EDTA as a final step in surgical preparation of an endodontic treatment 16 , Poletto et al. conclude that ultrasonic activation of irrigant solutions did not contribute to a more effective removal of smear layer after root post preparation 9 . There are different possible explanations for this: Coniglio et al. 2 suggest that ultrasound may compact debris particles laterally against root canal walls, Guo et al. 17 refer to the creation of new smear layer due to the scratch marks left by the instrument on dentin walls, while Chen 18 assumes that the discrepant results among studies are related to the different sizes of the preparations in the models used. Although it was not one of the aims of this study, we noted the presence of scratch marks on some samples in all experimental groups, including Group C, where only passive irrigation of the dental smear was performed after root post space preparation, without activating the distilled water by any mechanized system. This suggests that, at least in Group C, these marks may have been caused by the Largo or pilot bur of the post system during preparation of the root bed. In agreement with Guo et al. 18 , we noted more scratch marks in Group PIU, despite the precautions taken to avoid touching root canal walls upon activating the thin tip of the ultrasound.
In contrast to Goldberg 19 , we found that although there was no significant difference between rotary brush and control groups, the rotary brush tended to provide cleaner results, approaching those of the XP-endo Finisher group in the middle and coronary thirds. This difference between the studies is probably related to differences in the endodontic obturation techniques used. We filled only the apical third using single cone and warm vertical compaction technique, immediately preparing the post space and cleaning unset remains of sealant from root canal walls, which must have facilitated the removal of material. It is accepted that the continuous vertical condensation technique provides better obturation quality in the coronary third because the thermoplasticized gutta-percha provides better filling of the irregularities in the root canal 20 ; nevertheless, in the middle and apical thirds, the single cone and the continuous vertical compaction techniques provide similar sealing 21 .
Despite the difficulties involved in achieving a completely clean dentinal surface after post space preparation, the XP-endo Finisher instrument was the most efficient for removal of secondary smear layer, followed by the Rotoprox conical brush.