INTRODUCTION
Dental pain, dentinal sensitivity, and periodontitis are caused by strnetural damage to the enamel, dentin and periodontal tissues, usually triggered by biofilm on these surfaces. Dental pain associated with microbiological, physical, or chemical stimuli is the most frequent cause for dental consultation, and affects patients’ quality of life. Currently, non-steroidal anti-inflammatory drugs (NSAIDs) and opioids are frequently used for treating pain 1 , but due to the complex pathophysiology of pulpal pain 2 , this traditional approach may not be effective, so it is necessary to find an alternative that could be applied locally.
Odontoblasts and gingival fibroblasts are found in the pulp and gingiva, respectively, and are essential to oral health because they respond to external noxious stimuli and participate in mechanotransduction and tissue repair activities by expressing a variety of ion channels and receptors3,4.
Cannabinoid receptors are ligand-activated G-protein coupled receptors found mainly in the plasma membrane5,6. The binding of the ligand generates allosteric changes in the receptor, enabling the activation or inhibition of multiple signaling pathways. In this regard, the cellular response depends on the type of agonist and the biological context7,8.
The role of CB1 and CB2 in the regulation of pain pathways through the modulation of neurotransmitter release in nerve endings has been widely reported 9 . There is also evidence of the anti-inflammatory effect caused by the activation of both receptors 10 . For these reasons, those receptors have been investigated as therapeutic targets for treating pain and inflammation.
In several pathological conditions, the expression of cannabinoid receptors is regulated to maintain homeostasis and physiological processes, so different cells and tissues express them to mediate antinociceptive and anti-inflammatory effects 11 .
In dental tissues, both receptors could have a crucial role in dental sensory transduction, biomineralization, and tissue repair 12 , which is why extensive research has been performed in recent years. The expression of both receptors has been demonstrated in murine odontoblasts and gingival fibroblasts13-15, and CB1 was found in the human odontoblast primary cultures 12 . Furthermore, the presence of CB2 in gingival fibroblasts seems to be involved in controlling the inflammatory environment, such as during periodontitis 16 .
The aim of this study was to define the presence and activity of cannabinoid receptors in cultured odontoblast-like cells and primary human gingival fibroblasts, and evaluate the change in CB1 and CB2 expression under inflammatory conditions in odontoblasts. Results will provide important information that could be used to develop new therapeutic options using cannabinoid receptors as a pharmacological target for controlling pain and inflammation.
MATERIALS AND METHODS
Cell culture
Odontoblast-like cells (OLC) were differentiated from dental pulp mesenchymal stem cells using 10 ng/mL TGF-p (Abcam, Cambridge, MA, USA) for three weeks, following the previously established protocol 17 . In addition, human gingival fibroblasts (HGF) were harvested from gingival biopsies taken from periodontal plástic surgery. In brief, small tissue fragments were dissociated with a collagenase (100 U/mL) and dispase (1 mg/mL) mix for one hour, then washed with fresh médium and centrifuged. The cell pellet was seeded in 25 cm 2 culture flasks for seven days and maintained at 37 °C in a 5 % of CO2 incubator. In both cases, the cells were cultured in DMEM (Hyclone, Thermo Scientific, Bremen, Germany) supplemented with 10% FBS (Gibco; Thermo Fisher Scientific) and antibiotic (100 U/ml penicillin + 100 μg/ml streptomycin) until 70 % confluence was reached, to be distributed on dark 384-well plates after 0.25% trypsin-versene treatment to harvest cells. In addition, 24-well plates seeded with OLC were treated for 24 hours with 2 μg/ml E. coli LPS (Sigma-Aldrich, St. Louis, MO) or 40 μg/ml Poly-I:C (InvivoGen, San Diego, CA) to simulate bacterial and viral infection inflammatory conditions, respectively. The project received the endorsement of the Ethics Committee of Facultad de Odontología, Universidad Nacional de Colombia (B.CIEFO-008-2021).
CB1 and CB2 transcription by multiplex RT-qPCR in OLC and HGF
A one-step retrotranscription quantitative polymerase chain reaction (RT-qPCR) using Taqman® Multiplex Real-Time solution hydrolysis probes (Thermo Fisher Scientific) was used to determine the transcription of CB1 and CB2 using the CFX96 real-time thermal eyeler detection system (BioRad; Hercules, CA, USA). Amplification conditions were as follows: 10 min retrotranscription at 55 °C, denaturation for 1 min at 95 °C, 40 cycles of amplification with an alignment temperature of 58 °C, using the following primers and probes (Macrogen®) CB1: Forward 5'-ggttagcaagatacactcaagcatga-3' Reverse 3'-CTGGAAAAAGGCCCAACAAG-5', Probe: 6FAM-5 ’-CAGCTGCTGCTTTCTTCTTCTT ACACACCCCGGTCTC-3 ’ -TAMRA; CB2: Forward 5'-gacacacggacccctttttttcttgct-3' Reverse 3'-CCTCGTGGCCCTACCTATCC-5', Probe: ROX-5 ’ -TGGCCTTGCCCACCTGCACACA CAG-3’-TAMRA and P-aetin was used as reference gene: Forward 5'-GGATGCAGAAGGAGATCACT G-3' Reverse 5'-CGATCCACACGGAGTACTTG-3', HEX: 5'-CCCTGGCACCCAGCACAATG-3'.
CB1 and CB2 transcription during inflammatory stimuli in OLC
RT-qPCR was performed to evaluate CB1 and CB2 gene expression of LPS- and Poly-I:C-stimulated odontoblasts using SYBR Green (Luna® Universal One RT-qPCR Kit, New England BioLabs; USA). Amplification conditions were as follows: 10 min retrotranscription at 55 °C, denaturation for 1 min at 95 °C, 40 cycles of amplification with an alignment temperature of 58 °C using the following primers (Macrogen®) CB1: Forward 5'-ggttagcaagatacactcaagcatga-3' Reverse 5' CTGGAAAAAGGCCCAACAAG-3', CB2: Forward 5'-GACACGGACCCCTTTTTGCT-3' Reverse 5' CCTCGTGGCCCTACCTATCC-3' and Cholinergic receptor beta 2 subunit gene (CHRNB2) was validated and used as reference gene: Forward 5'-CAATGCTGACGGCATGTACGA-3' Reverse 5'-CACGAACGGAACTTCATGGTG-3'. PCR effi-ciency was calculated with LinRegPCR sofware (Academic Medical Center, AMC, Amsterdam, Netherlands), and relative quantifieation was performed with the Schefe method 18 using unstimulated cells as a control.
Detection of CB1 and CB2 proteins in OLC and HGF
An indirect immunofluorescence technique was performed for CB1 and CB2 protein detection. Briefly, the cells (5 × 104 cells/well) were seeded on poly-L-lysine-treated glass coverslips for 24 hours, then some cells were treated for 24 hours with 2 μg/ml LPS or 40 μg/ml Poly-I:C, the cells were fixed with 4 % paraformaldehyde (PFA), and permeabilized with Triton X-100, to subsequently block with 10% goat serum. Cells were incubated at 37 °C with anti-CB1 or anti-CB2 polyclonal primary antibody produced in mouse (Invitrogen, Thermo Fisher Scientific), diluted 1:100 in blocking buffer. The samples were then incubated with Alexa 594 conjugated anti-mouse IgG secondary antibody (Thermo Fisher Scientific) diluted 1:200 in PBS at room temperature. The nuclei were counte rstained with DAPI, and the slides were mounted with Prolong®, for observation under the Zeiss Axio Imager A2 microscope (Gottingen, Germany) with the Axio Vision software. The presence or absence of the protein in cells and its location were evaluated in three independent experiments (n = 3).
Agonists, antagonists, and intracellular Ca2+ measurement
In vitro CB1 and CB2 activity in OLC and HGF was determined by calculating changes in intracellular calcium concentrations ([Ca2+]i) after different stimuli. Firstly, cells were loaded with 2 μM of fluo-4 AM solution (Invitrogen, Thermo Fiseher Scientific) at 37 °C for 45 min in the dark. Next, cannabidiol 10 μM (CBD) (Biominerales Pharma, Colombia-Canada) was used for 1 and 10 min cell stimulation, either alone or with a 5-minute pretreatment with different antagonist concentrations (antagonist: agonist combinations, 10μM:10μM; 10μM:1nM; 1nM:1nM; 1nM:10μM), according to a previous viability experiment. A selective CB1 antagonist (AM251, Tocris, Bristol, UK), a selective CB2 inverse agonist (AM630, Tocris, Bristol, UK) and a TRPV1 antagonist (Capsazepine, CZP (Tocris, Bristol, UK) were used. The stock solution at a concentration of 10 mM was prepared in DMSO, and the serial dilutions used were prepared in culture medium.
Ionomycin is a lipophilic molecule that binds to calcium ions and carries them through membranes, so it was used as a positive control. Fluorescence quantitation of the cells was performed at a wavelength of 494/525 nm (excitation/emission) in a spectrofluorimeter (ClarioSTAR, BMG Labteeh). For the analysis of results, the data were normalized >with respect to unstimulated cells (F/FO) and plotted at each evaluation time (1 and 10 min).
Data analysis
Data were organized in excel spreadsheets (Microsoft Office 2010) and described as mean and standard deviation (SD). Figures were prepared using GraphPad Prism 7.0 software (GraphPad Software, San Diego, CA, USA). All experiments were performed in triplicate. One-way analysis of variance ANOVA was used to determine the P-value using an alpha value <0.05.
RESULTS
Odontoblasts and gingival fibroblasts express CB1 and CB2, and are upregulated in the inflammatory environment.
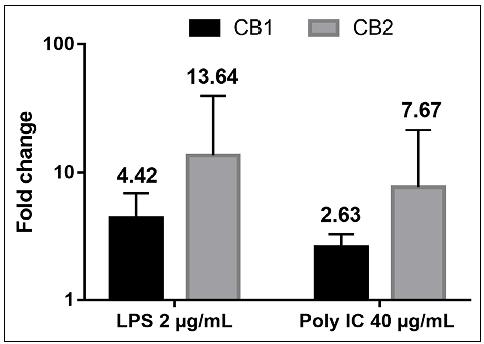
Both OLC and HGF express CB1 and CB2 transcripts. For OLC, the mean Cq values were 31.62 and 32.02 (CB1 and CB2, respectively), while for HGF, they were 29.87 and 31.62, showing a similar pattern of receptor transcription (Fig. 1) . Odontoblasts stimulated with LPS and Poly-I:C showed increases the cannabinoid receptor transcripts. Stimulation with LPS induced a fivefold rise in CB1 expression levels, while CB2 increased more than tenfold regarding unstimulated cells (Fig. 2) . Poly-I:C stimulus caused a significant 4- and 8-fold increase in transcription of CB1 and CB2, respectively, in OLC. Overexpression of CB2 was significantly higher than CB1 transcripts in both LPS and Poly-I:C conditions.
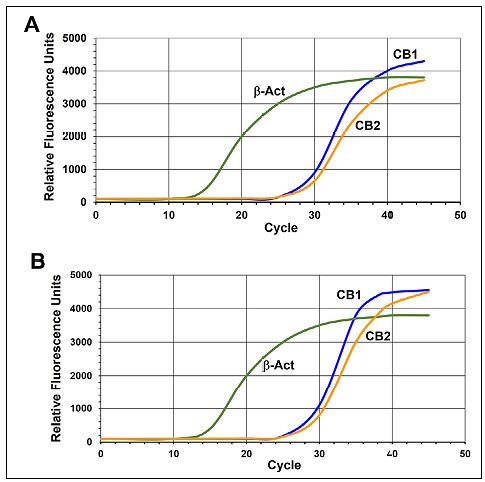
Fig. 1 Multiplex RT-qPCR amplification curves. (A) Human Gingival fibroblasts; (B) Odontoblast-like cells.
Odontoblasts and gingival fibroblasts express CB1 and CB2 protein
Additionally, using irnmunofluorescence, both CB1 and CB2 proteins were identified in HGF ( Fig. 3B ) and OLC ( Fig. 3C ). SH-SY5Y and monocyte-derived macrophage cells were used as positive controls, respectively ( Fig. 3A ). Both receptors were found to be expressed in the cell membrane, nucleus, and cytoplasm, as fluorescence was observed in these areas in both cell types.
CBD increases intracellular calcium concentrations in odontoblasts and gingival fibroblasts
Stimulating gingival odontoblasts and fibroblasts with 10 μM of cannabidiol increased intracellular calcium concentrations compared to unstimulated cells (p<0.001). Pre-treatment with AM251, AM630 and CZP antagonists of CB1, CB2 and TRPV1 respectively, inhibited calcium influxes in both cell types ( Fig. 4 ), demonstrating functional receptors in those cells.
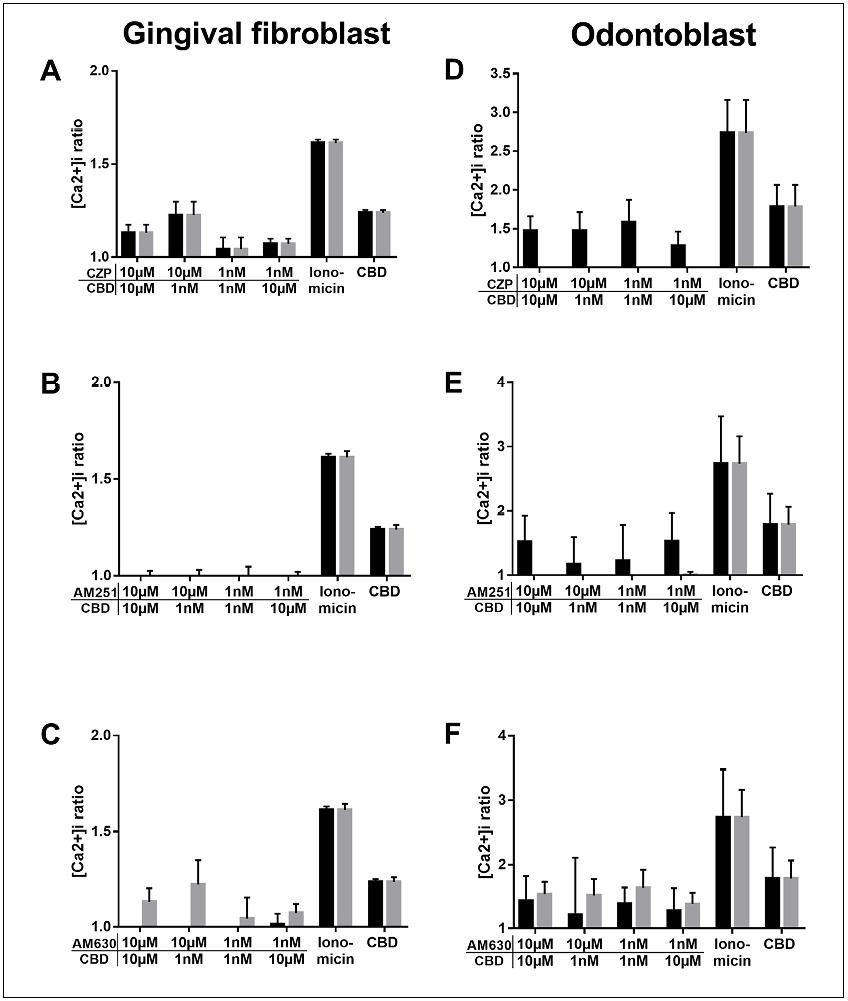
Fig. 4 Change in intracellular calcium concentrations by CBD and antagonists effect. Data are the ratio between specific fluorescence and fluorescence in unstimulated cells (Fi/F0) at one minute (black bar) and 10 minutes (grey bar). A - C: Gingival fibroblasts; D - F: Odontoblasts. P-value = 0.001.
DISCUSSION
This study demonstrated the expression of CB1 and CB2 oannabinoid receptors in human odontoblasts and gingival fibroblasts. An increase in their expression was observed in immune challenges. Additionally, their functionality was evidenced based on observed changes in calcium influx when CB1, CB2 and TRPV1 bind to their respective antagonist or inverse agonist. These cells are involved in response to mechanical, chemical, or infectious challenges in dental or periodontal tissues. Additionally, these receptors were found to be upregulated after inflammatory stimuli.
The expression and function of both receptors in dental tissues have been reported previously. For example, CB1 was found in nerve endings in human dental pulp 19 . Furthermore, immunohistochemistry has found evidence of the presence of CB1 receptors in mouse odontoblasts 20 . Moreover, CB1 and CB2 expression has been reported in periodontal ligament stem cells, where they are involved in differentiation to osteo- and dentinogenic phenotype through p38, MAPK and JNK signaling in an inflammatory environment14,21. The major importance of CB in tissue repair has been highlighted, since they contribute to regulating bone volume and metabolism, bone loss and bone cell function in murine models 15 .
CB1 and CB2 were found to be expressed in mouse periodontal tissue and in human periodontal fibroblasts. The activation of these receptors induces their adhesion and migration through the activation of focal adhesion kinase and MAPK activity 21 , showing that they particípate in the regulation of several signaling pathways and gene expression. Here, under inflammatory conditions, using LPS and Poly-I:C as bacterial or viral challenge surrogates, upregulation of CB1 and CB2 transcripts was found. Higher fluorescence CB receptor reactivity was observed in LPS and Poly I:C treated cells compared to untreated OLC and HFG, which could be interpreted as an increase in the amount of both cannabinoid receptors under inflammatory conditions. Previous research reports that CB2 is expressed in normal conditions but is exceptionally high in pathological inflammation 9 . Maresz, et al, (2005) demonstrated in microglial cells that an inflammatory environment induces cell activation and CB2 overexpression, dependent on IFN-y and GM-CSF cytokines stimuli 22 . Positive labelling for CB1 and CB2 is observed throughout the cell. In addition to membrane activity, functional cannabinoid receptors have been reported to be located intracellularly in the nucleus, vesicles and mitochondria, where they can actívate intracellular >signaling23,24.
Although periodontal ligament cells typically express both cannabinoid receptors, CB1 has higher expression in healthy tissue, even though it is downregulated during bacterial infection, while the CB2 is induced to overexpression. Conversely, sterile inflammation caused significant upregulation of CB1 and CB2 13 . The treatment with CB2 agonists and antagonists was found to regulate the inflammatory response in periodontal ligament fibroblasts. CB2 agonist ligands attenuated p38 and NFkB phosphorylation and simultaneously caused an increase in p-ERK and p-CREB, affecting cAMP concentration and P-arrestin pathways15,25. Nakajima et al. (2006) reported that CB1 and CB2 are upregulated in HGF samples from gingivitis and periodontitis. Stimulation of HGF with anandamide reduced LPS-induced secretion of cytokines, and CB1 or CB2 receptor blocking with specific antagonists attenuated that effect, confirming the involvement of cannabinoid receptors in the anti-inflammatory effect of the endocannabinoid anandamide 26 .
On the other hand, the functionality of the receptors was evaluated by FLUO 4 AM, quantifying intracellular calcium concentrations, as it is known that activation of CB1 and CB2 leads to inhibition of adenylyl cyclase. This results in the blockade of A-type potassium channels (K+A) and Ca2+ channels (L-, N-, P-/Q- and voltage-dependent) and increases the opening of inwardly rectifying potassium channels (Kir), causing cells to hyperpolarize 27 . We found that CBD generates an increase in intracellular calcium concentration. In addition, there is evidence that human odontoblasts express functional CB1, since they respond after stimulation with different agonists and antagonists 12 . CBD is the main non-psychoactive component of cannabis and is reported to possess sedative, anti-inflammatory, and antipsychotic effects 28 , although several of these effects seem to be mediated by mechanisms independent of cannabinoid receptors. Previous evidence has shown that CBD inhibits several receptors such as TRPV1, GPR55, NMDAR, opioid receptors, adenosine A1 receptors and voltage-dependent calcium channels; and interestingly, it also activates PPAR-ɣ, 5HT1A and the glycine receptor29,30. Therefore, changes in intracellular calcium concentrations generated by CBD are due to the activation and blockade of several associated channels. Since calcium channels are involved in the CB1 and CB2 signaling cascade activation, measuring calcium influxes provides indication of whether these receptors are functional.
Thus, the action of CBD through these pathways may be responsible for the suppression of neuronal excitability and pain perception. Still, there is also evidence that CBD inhibits the uptake of dopamine, noradrenaline, GABA, serotonin and anandamide in the synapses 31 , which would explain its antinociceptive and neuroprotective effects. Moreover, the anti-inflammatory effect is also explained by COX-2 inhibition, which prevents the production of arachidonic acid metabolites 32 . Even so, in this work, it was found that when cells were stimulated with CBD and CB1 and CB2 antagonists, intracellular calcium concentrations were reduced, showing that CBD may be acting directly or indirectly through cannabinoid receptors. On the other hand, the selective antagonists used (AM251 and AM630) are also inverse agonists, which means that they have a negative efficacy for a certain signaling pathway, which ends up blocking calcium channels so that when stimulating with CBD, no calcium influx is generated through CB1 and CB2. Crosstalk is a mechanism of interaction between different intracellular substances or signal transduction pathways 12 . Previous reports have shown that there is coupling between cannabinoid receptors and TRPV1. For example, in odontoblast cells, a stimulus inducing an intracellular calcium increase follows the ion extrusion through the sodium-calcium exchanger channels towards the mineralization front 33 , explaining the role of cannabinoid receptors and TRPV1 in calcium apposition in the tertiary dentin and the transduction of external stimuli.
CBD is a TRPV1 agonist and negative allosteric modulator of cannabinoid receptors 34 , so the explanation for the decrease in intracellular calcium when CB1 and CB2 antagonists are applied may be crosstalk between both types of receptors, where signaling via TRPV1 may be decreased when cannabinoid receptors are blocked, even at low concentrations of CB1 and CB2 antagonists and high concentrations of CBD.
Such communication between the two receptors or their signaling pathways causes a physiological balance to be maintained under pathological conditions. For example, previous evidence has demonstrated that cannabinoid receptors mediate anti-inflammatory and protective effects in periodontal tissues, while TRPV1 mediates pro-inflammatory effects, which lead to periodontal injury 15 . Further studies should be performed on odontoblasts and gingival fibroblasts to elucidate the signaling pathways that can be activated by CBD and the mechanism by which crosstalk with TRPV1 is generated, so that CBD can be considered a possible treatment for infectious or inflammatory pathologies in the oral cavity.
CONCLUSIONS
This study demonstrated that human odontoblasts and primary gingival fibroblasts express functional CB1 and CB2 cannabinoid receptors, and that under inflammatory conditions, both receptors are overexpressed, especially CB2. On the other hand, it showed that CBD generates calcium influxes, demonstrating that it has biological activity and functional communication with TRPV1. These results show the importance of the expression of these receptors in the physiological and pathological processes in the dental pulp and periodontal tissues, demonstrating their functional expression in these cells and opening new perspectives for searching for alternatives for more effective treatments against dental pain and inflammation.