INTRODUCTION
Oral mucositis (OM) manifests as inflammation of the oral mucosa with whitish and/or yellowish patches, ulceration, atrophy of the mucosa, erythema, oedema and bleeding 1 . Among the most severe con-sequences of OM are pain, dysphagia, weight loss, malnutrition, difficulty in speaking, and superinfec-tion, leading to serious deterioration in the patient’s quality of life 2 . The lesions may begin 2 to 3 weeks after the start of oncological treatment 3 . When OM is severe, it is a dose-limiting toxic side-effect of ra-diotherapy, chemotherapy, and particle radiation for patients with head and neck cancer, with negative impact on cancer prognosis4,5.
OM can be divided into five stages: initiation, re-sponse to primary damage, amplification of the signal, ulceration, and healing. OM pathogenesis starts with damage to cell genetic material due to intense exposure to reactive oxygen species (ROS), such as free radicals, generated by direct and indi-rect radiation and/or chemotherapy, triggering the clinical effects mentioned above. This exposure leads to the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB), which in turn modulates the expression of the interleuk-ins IL-1, IL-6, and of the tumoral necrosis factor alpha (TNF-α) promoting the production of metal-loproteinases causing cell damage and death 6 . The destruction of the oral mucosa fosters colonization by other microorganisms such as viruses, bacteria or fungi, enhancing their inflammatory activity and thereby increasing lesion severity 7 . OM is diagnosed in 40 - 50% of cancer patients treated with chemotherapy, 80 - 100% of patients treated with stem cell transplants, and 80 - 100% of patients treated with radiotherapy of the head and neck 1 .
Low-level laser treatment has been evaluated in many studies, with controversial results 8-12 . Although its effectiveness in reducing the severity of OM has been demonstrated in several studies, it has not always been found to be superior to placebo or other treatments. Several methods have been used to prevent and treat OM induced by chemotherapy or radiotherapy, but to date, none has proven complete success 13 . Thus, there is a pressing need for strategies to overcome mucositis that are effective, well-tolerated, and easy to use 14 . Therefore, the aim of this systematic review was to update the evidence published during the last five years on the treatment of oral mucositis induced in cancer patients by radiotherapy and/or chemotherapy.
MATERIALS AND METHOD
Search strategy
A systematic search was conducted in the MEDLINE, Scielo and SCOPUS databases using free terms, Mesh terms and the Boolean operators AND and OR, with the search terms: mucositis, stomatitis, therapy, treatment, oral cancer, oral squamous cell carcinoma, head and neck cancer, head and neck carcinoma. The search strategy is summarized in Table 1.
Table 1 Search strategy
| Data base | Search strategy | Results |
|---|---|---|
| PUBMED | (("Mucositis"[Mesh]) OR "Stomatitis"[Mesh]) AND (therapy OR treatment) AND (oral cancer OR oral squamous cell carcinoma OR Head and neck cancer OR head and neck carcinoma) | 175 |
| SCOPUS | (Mucositis OR Stomatitis) AND (therapy OR treatment) AND (oral cancer OR oral squamous cell carcinoma OR Head and neck cancer OR head and neck carcinoma) | 109 |
| SCIELO | (Mucositis OR Stomatitis) AND (therapy OR treatment) AND (oral cancer OR oral squamous cell carcinoma OR Head and neck cancer OR head and neck carcinoma) | 3 |
Review protocol
The systematic review protocol was registered in the International prospective register of systematic reviews (PROSPERO) of the database of the National Institute for Health Research (www.crd.york.ac.uk/prospero), under registration number CRD42020200215. The systematic review was carried out in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Metaanalysis (PRISMA).
Inclusion and exclusion criteria
The inclusion criteria were: randomized clinical trials, in humans, published in English, from 2017 to 2023 and full text available. To ensure that the review is as current as possible, the search was updated to include articles published up to January 2023.
This systematic review followed the PICOS (population, intervention, comparison, outcome, and study design) approach to define the inclusión criteria:
Study design: randomized clinical studies. Population: anyone with cancer and OM induced by radiotherapy and/or chemotherapy.
Intervention: therapy for OM.
Comparison: placebo or no treatment, or another active intervention.
Outcome: OM severity, nutritional status, symptoms and signs, quality of life, oral intake capacity, transition to oral nutrition, duration of severe OM, pain duration and other possible variables.
The exclusion criteria were: non-randomized clinical studies, in vitro studies or studies in animals, articles in languages other than English, patients who did not present OM, and studies which reported only prevention or incidence of OM.
Selection of studies
All the references identified were exported to the Mendeley® Reference Manager to facilitate the management of duplicates. The articles were reviewed by two authors independently (JS, SW), and a third author (LB) resolved disagreements when necessary. An initial selection was based on title and abstract, and followed by full-text analysis using the Cochrane Collaboration Covidence® tool.
Data extraction
In the data extraction process, study details were extracted in two different tables (Tables 2 and 3 ). This was done by two reviewers (JS, GC). All reviewers discussed each article to reach consensus regarding the study details. The information extracted from each study included: author, year of publication, size and age of sample, distribution of the groups, cancer treatment, criteria for OM diagnosis, OM treatment, variables evaluated and results.
Table 2 Characteristics of the selected studies
| Authors | Number of subjects (age) | Sample distribution | Cancer therapy | OM diagnosis criteria according to |
|---|---|---|---|---|
| Lalla 2020 | 108 (>18 y.o) | 55 Dentoxol 53 Control | RT | WHO |
| Soltani 2020 | 46 (18-65 y.o) | 23 Plantago major syrup 23 Control | RT | WHO |
| Chaitanya 2020 | 75 (>18 y.o) | 25 Topical 5% zinc oxide paste 25 Improvised zinc (1%) 25 Control | RT CT | WHO |
| Immonen 2020 | 45 (2-18 y.o) | 24 Caphosol 21 Control | CT | WHO |
| Sun 2019 | 100 (24-67 y.o) | 50 Vitamin B + GeneTime 50 Vitamin B (control). | RT | RTOG |
| Sio 2019 | 275 (>18 y.o) | 78 Doxepin 76 Diphenhydramine- lidocaine-antacid 76 Control | RT | RTOG |
| Huang 2019 | 64 (35-75 y.o) | 31 L- Glutamine 33 Control | RT | CTCAE (Definition OM) |
| Legouté 2019 | 83 (18-75 y.o) | 42 LLL-T/ PBM-T 41 Control | CT | WHO |
| Charambolous 2018 | 72 (>18 y.o) | 36 Thyme honey 36 Control | RT CT CT + surgery | RTOG |
| Huang 2018 | 91 (>20 y.o) | 48 SMR/EP 43 Control | CT RT | WHO |
| Gobbo 2018 | 101 (3-18 y.o) | 51 PBM 50 Control. | CT | WHO |
| Cabrera-Jaime 2017 | 50 (>18 y.o) | 15 Sodium SB 5% + Plantago major 19 SB 5% + CMW 0.12% 16 Control | CT | WHO |
| Medeiros 2017 | 36 (3-16 y.o) | 18 LLL-T 18 LLL-T + PC-T | CT RT | Non mentioned |
| Erden 2017 | 90 (17-66 y.o) | 30 CMW 30 OC-T 30 Control *All received antibiotics for oral ulcerations. | CT | WHO |
| Wong 2017 | 215 (>18 y.o) | 108 Caphosol 107 Control | RT CT | Non mentioned |
| Mohammadi 2021 | 144 (>18 y.o) | 48 Zinc chloride 0.2% 48 SB 5% 48 Control | CT | WHO |
| Yin 2022 | 87 (18-80 y.o) | 44 RADoralex 43 Control | RT CT | WHO |
| Javad Kia 2021 | 50 (>18 y.o) | 25 Curcumin nanomicelle 80 mg 25 Control | RT CT |
WHO |
Table 3 OM Treatment and results of the selected studies
| Authors | OM Treatment | Variables | Results |
|---|---|---|---|
| Lalla 2020 | Dentoxol | Incidence and OM severity Duration of severe OM Pain due to OM | Proportion of severe OM patients at 3-6 weeks after treatment was significantly lower in the dentoxol group vs. control. No significant difference in the other variables evaluated. |
| Soltani 2020 | Plantago major (syrup) | OM severity Pain intensity | Severity of OM and pain was significantly lower in the Plantago major group. |
| Chaitanya 2020 | Zinc oxide Improvised zinc | OM severity | Severity of OM was significantly lower in both zinc group vs. control. No significant difference between improvised zinc group and zinc oxide group. |
| Immonen 2020 | Caphosol | OM severity Patient-reported oral symptoms. | No significant difference between caphosol and control. |
| Sun 2019 | Vitamin B + GeneTime Vitamin B alone | Ulcer area Pain intensity Healing time. | Severity of OM was lower in the Vitamin B+ Genetime group. Significantly more ulcers healed in 1- 2 weeks in Vitamin B+ Genetime group vs. control, but not at 3 weeks. |
| Sio 2019 | Doxepina Diphenhydramine- lidocaine-antacid | Pain reduction | The pain reduction was significantly greater in the doxepin group and lidocaine group vs. control. The stinging and burning were significantly higher in the doxepin group vs. control. |
| Huang 2019 | L- Glutamine | OM severity | L-glutamine significantly decreased the mean maximum severity of OM vs. control. Significantly more patients in control group developed OM. |
| Legouté 2019 | LLL-T/ PBM-T | OM severity Nutritional status Pain intensity | No significant difference in OM severity, nutritional status or pain between LLL-T and control. |
| Charambolous 2018 | Thyme honey | OM severity Symptoms and signs severity Quality of life. | OM severity and weight loss were significantly lower in the thyme honey vs. control. Quality of life was significantly higher in thyme honey vs. control. |
| Huang 2018 | SMR/EP | OM severity Symptoms score Quality of Life | Physical and socio-emotional quality of life was significantly higher in SMR/EP vs. control at 8 weeks. Severity and symptoms of OM were not different between the groups. |
| Gobbo 2018 | PBM | OM severity Pain intensity | OM severity was significantly lower in the PBM vs. control. Pain reduction was significantly higher in the PBM vs. control. |
| Cabrera-Jaime 2017 | SB 5% + Plantago major SB 5% + CMW 0.12% Double SB 5% aqueous solution | Healing time Pain intensity Oral intake capacity Quality of life. | No significant differences in the variables evaluated between the groups. |
| Medeiros 2017 | LLLT LLLT + PCT | OM lip lesion area (cm2) | LLLT + PCT group showed significantly smaller lesion areas vs. LLL-T alone. |
| Erden 2017 | CMW OCT | OM severity Transition time to oral nutrition. | Transition time from oral nutrition in the chlorhexidine group was significantly shorter than in other groups. |
| Wong 2017 | Caphosol | OM severity Duration of OM Incidence and duration of severe dysphagia pain Quality of life. | No significant difference in the variables evaluated between the groups. |
| Mohammadi 2021 | sodium bicarbonate mouthwash zinc chloride mouthwash | OM severity Quality of life |
OM severity was significantly lower in both groups vs. placebo. Quality of life was significantly higher in both groups vs. placebo. |
Risk of bias in individual studies
To evaluate the reliability of the results of the studies selected, the Cochrane criteria for assessing the risk of bias were used by two authors (JS, GC), analyzing the following: sequence generation, allocation concealment, blinding of participants, incomplete outcome data, selective outcome reporting, as well as other possible sources of bias such as conflict of interests, in each of the studies included.
A third author (SW) resolved disagreements when necessary.
RESULTS
A total 287 articles were retrieved, of which 86 were selected by title and abstract and 18 were finally included after full-text analysis 15-32 . The selection process is detailed in Fig. 1. The sample size in the studies selected ranged from 31 to 275 subjects. The sample included adults aged over 18 years in 15 studies 15-17 , 18 , 20-24 , 26 , 28-32 and children/adolescents in 3 studies 19 , 25 , 27 . A meta-analysis was not performed due to the heterogeneity of the studies found.
Risk of bias
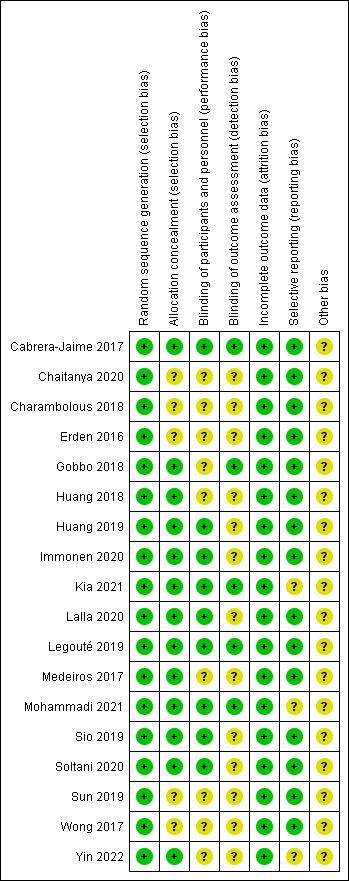
The risk of bias assessment summary is shown in Fig. 2, in which green indicates low risk of bias, yellow indicates unclear risk and red indicates high risk. All studies obtained a low risk of bias in sequence generation and in incomplete outcome data, 72% of the studies had a low risk of bias in allocation concealment, 50% had a low risk of bias in participant blinding, and 83% had a low risk of bias in selective outcome reporting. None of the aspects evaluated showed a high risk of bias.
Type of cancer
Three studies included hematological malignancies 25 , 28 , 30 , three included solid tumours 19 , 26 , 30 , one included hematopoietic stem cell transplantation (HSCT) 13 , nine included head and neck cancer 15 , 17 , 20-23 , 29 , 31 , 32 , two included oral cavity cancer 18 , 24 and one included leukemia, osteosarcoma and/or lymphoma 27 .
Cancer treatment
The cancer treatments were radiotherapy alone in five studies 16 , 17 , 20 , 22 , chemotherapy alone in six studies 15 , 19 , 25 , 28 , 30 and radiotherapy and/or chemotherapy in seven studies 18 , 23 , 24 , 27 , 29 , 31 , 32 .
Oral mucositis diagnosis
In three studies, oral mucositis (OM) was diagnosed according to the criteria established by the Radiation Therapy Oncology Group (RTOG) 20 , 21 , 23 . The RTOG classifies OM into five grades: Grade 0, no change in the mucosa; Grade I, erythema and slight pain; Grade II, patchy mucositis, inflammatory or bloody secretion, and moderate pain; Grade III, confluent fibrous mucositis and severe pain; and Grade IV, ulceration, necrosis, bleeding. Nine studies used the criteria established by the World Health Organization (WHO) 15"18 , 25 , 26 , 28 , 30 , 32 , which also classifies OM into five grades: Grade 0, without OM; Grade 1, painless ulcers, erythema, or mild soreness in the absence of lesions; Grade 2, painful, erythema, oedema or ulcers, but able to eat; Grade 3, painful, erythema, oedema or ulcers, requiring intravenous hydration; and Grade 4, requiring parenteral or enteral nutrition or support. One study used the Oral Mucositis Assessment Scale (OMAS) 31 to grade the severity of ulcers, as follows: Grade 0, no lesion; Grade 1, lesion < 1 cm2; Grade 2, lesion between 1 and 3 cm 2 ; and Grade 3, lesion > 3 cm 2 . In three studies, the diagnostic criteria were not specified, although the selection criteria included patients with 1 and 3 cm 2 ; and Grade 3, lesion > 3 cm 2 . In three
Oral mucositis treatment
The treatments used for OM were Dentoxol mouthwashes 16 , Plantago major extract mouthwashes 17 , 26 , zinc oxide paste 18 , Caphosol mouthwashes 19 , 29 , GeneTime spray with vitamin B 20 , doxepin mouthwashes, diphenhydramine-lidocaine-antacid mouthwashes 21 , low-level laser therapy 15 , 25 , 27 L-glutamine 22 , thyme honey extract mouthwashes 23 , sodium bicarbonate mouthwashes 26 , 30 , 31 , zinc chloride 30 , chlorhexidine mouthwashes 28 and saline solution mouthwashes 24 , Radolex 31 and nanomicelle curcumin capsules 32 .
Variables evaluated
The variables evaluated were: ulcer area 20 , 26 , 31 , pain 15-17 , 20 , 21 , 25 26 , 29 , healing time 20 , 26 , OM severity 15-18 , 22-25 , 28-31 , nutritional status 15 , 31 , symptoms and signs 23 , 24 , quality of life 23 , 24 ’ 26 ’ 29-31 , oral intake capacity 26 , transition to oral nutrition 28 , duration of severe OM 16 , and pain duration 29 . Pain was evaluated in three studies using the visual analogue scale (VAS) 17 , 20 , 26 , in which pain is classified on a scale of 1 to 10, where 1 indicates absence of pain and 10 indicates severe pain. In four studies, pain was evaluated using a Numerical Pain Rating scale (NRS) of 0-10 15 , 21 , 25 , 32 . In one study, appreciation of pain was evaluated by The Oral Mucositis Daily Questionnaire 16 . One study did not specify how pain was evaluated 29 .
DISCUSSION
Oral mucositis (OM) is caused by aggravated tissue damage to the cells of the oral mucosa. Early detection is limited to the symptoms reported by the patient, in most cases resulting in late diagnosis, as the detection parameters are subjective. OM is one of the main limitations in CT and RT dosage, and causes severe deterioration in patients’ quality of life 3 . Plantago major extract is described as having immunomodulatory, anti-inflammatory, antimicrobial, analgesic and antioxidant properties in wounds 30 . Subjects in radiotherapy treated with Plantago major syrup presented less severe OM compared to subjects treated with a sugar-based placebo syrup. Similar results were recorded in pain intensity, as patients treated with Plantago major experienced significantly less pain 17 .
Zinc chloride and sodium bicarbonate mouthwashes have been effective in treating and reducing the severity of OM, and subsequently improving quality of life in patients with cancer under chemotherapy 30 . In another study, the effectiveness of sodium bicarbonate was compared to RADoralex®, a pseudoplastic fluid diluted with pure water that covers the mucosa and forms a thin, sticky coating that acts as a physical barrier, blocking the invasión of pathogenic bacteria. RADoralex® significantly reduced the incidence and severity of radio-chemotherapy-induced oral mucositis in patients with locally advanced nasopharyngeal carcinoma, delayed the progression of OM, promoted the healing of the oral mucosa, and relieved oral and throat pain. In addition, RADoralex® reduced weight loss during treatment and improved patient quality of life 31 .
A mouthwash based on Plantago major combined with 5% sodium bicarbonate, an alkalinizing solution which would prevent the growth of acidophile bacteria, did not prove to be more effective than 5% sodium bicarbonate solution combined with 0.12% chlorhexidine in the evaluation of pain intensity, oral intake capacity or quality of life. Nevertheless, when the healing times to OM grade 0 are compared, it is observed that subjects treated with double solution of 5% sodium bicarbonate alone have a higher probability of early healing than subjects treated with Plantago major extract combined with 5% sodium bicarbonate 26 .
Chlorhexidine would appear to be less effective in OM treatment. Although it has a recognized antiseptic effect, in that it inhibits the enzyme activity of bacteria and prevents colonization 33 , it did not prove effective in diminishing degree of severity or in healing OM lesions, as it was not shown to accelerate cicatrization 26 . Nevertheless, it must be stressed that the mouthwash concentrations in general may result in different degrees of effectiveness of treatment. According to the trials conducted by Erden et al. 28 , there was a significant reduction in the transition time to oral feeding in patients with OM who gargled with chlorhexidine mouthwash, compared to subjects treated with cryotherapy or conventional care routines. However, the concentration of chlorhexidine that produced this effect was not reported by the authors. In trials with Dentoxol mouthwash, which consists of purified water, xylitol, sodium bicarbonate and other excipients, lower severity of OM was observed 3-6 weeks after treatment compared to subjects treated with placebo; however, no significant difference was reported in pain relief 16 . Caphosol mouthwash, a saline ionic solution of phosphate and calcium, has been proposed as an optimal treatment for the relief of OM through stabilization of the pH in the oral cavity 29 . Wong et al. 29 applied this mouthwash to 103 individuals, achieving better results in pain evaluation and dysphagia compared to subjects treated with conventional oral care alone. However, these results were not statistically significant, and furthermore, Caphosol did not reduce the incidence or duration of severe OM during and after radiotherapy. These results agree with those reported by Immonen et al. 19 , who tested Caphosol in pediatric and adolescent patients. Thyme honey mouthwash was shown to produce a significant improvement in the severity of OM and in patient quality of life after 7 weeks’ application 18 . Honey is known to have medicinal properties; it has proven effective in healing burns, surgical wounds and oral infections thanks to its antibacterial and analgesic agents, and its capacity to promote re-epithelialization 34 .
Sio et al. 21 compared two mouthwashes: one based on doxepin and the other on diphenhydramine, lidocaine and an antacid solution. The former is based on a tricyclic antidepressant drug which has been used topically in the treatment of lichen planus and urticaria. The latter contains diphenhydramine (an antihistamine, sedative and hypnotic compound), lidocaine (an anesthetic), and an antacid solution (based on aluminum hydroxide, magnesium hydroxide and simethicone). Both mouthwashes significantly reduced pain from OM during the first 4 hours after administration, compared to placebo 21 . Another plant-based compound is curcumin, which is derived from turmeric and has known anti-inflammatory and antioxidant properties 33 . Low concentrations of curcumin are thought to act as a photosensitizer in the treatment of oral infections, i.e., when curcumin is irradiated by a light source at a wave-length of 450 nm, it produces a photodynamic effect that can destroy microbial pathogens 35-38 . Moreover, capsules of curcumin nanomicelles are effective in preventing and treating radiotherapy and chemo-therapy-induced OM when compared to placebo, and may be an acceptable alternative for the current palliative and local treatments 32 . Other excellent alternative vehicles are pastes, ointments and syrups. Chaitanya et al. 18 tested two topical pastes based on zinc oxide: one at 5% and the other at 1% and in combination with amla, tulsi and curcumin. Zinc has antibacterial properties and may improve the cicatrization of OM-caused ulcers. This study showed lower OM severity with both these zinc compounds compared to the control group, though no difference was found between them 18 .
GeneTime® oral spray combined with a multivitamin B complex, proposed by Sun et al. 20 , acts as a recombinant human growth factor used for cicatrization and wound repair. Subjects treated with this compound presented a significantly lower degree of OM severity, and a larger number of ulcers healed after 2-3 weeks, with evident pain reduction 20 . Following this line of direct pharmacological treatment, Huang et al 22 . proposed the use of L-glutamine (glutamine and maltodextrin) during and after exposure to radiotherapy, achieving considerable improvement in comparison to controls. The groups were instructed to take either glutamine or the placebo orally, dissolved in cold water, 30 min. before a meal, three times per day.
Regarding therapies designed to produce tissue modification, interesting results have been reported for low-level laser therapy (LLLT), similar to those reported for photodynamic therapy (PD-T), photo-biomodulation therapy (PBM-T) and photochemo-therapy (PC-T). In all the therapies described, a co-adjuvant can be used to improve the results obtained; however, the results have been controversial. Photo-biomodulation therapy (PBM-T) has been shown to diminish pain significantly on day 7 after the start of treatment, and significantly reduce the severity of OM induced by chemotherapy in children and adolescents 25 . Similar results have been obtained with LLL-T 27 . This conflicts with the results reported by Legouté et al. 15 , who treated adult subjects with PBM-T and found no significant difference in reduction of OM severity, pain, nutritional state or quality of life in comparison to the controls. Differences in wavelength, power, distance and exposure time of the LLL-T may have affected the results. Clinical and experimental studies suggest that PBM reduces inflammation significantly, prevents fibrosis 35 , 36 and that its biological effects are closely related to dose and exposure time. The action mechanism is based on the predominance of PBM-T over the cytochrome c oxidase (CcO) enzyme in the mitochondrial respiratory chain by facilitating the transport of electrons, resulting in an increase in the transmembrane proton gradient which drives the production of adenosine triphosphate (ATP), enhancing cell metabolism functions 38 , 39 .
When oral cryotherapy is used with short-term intravenous chemotherapy agents, it causes local vasoconstriction which slows the blood flow; consequently, the distribution of the drug to oral epithelial cells diminishes, reducing the risk of OM. However, the reduction in blood flow to the tissues may also slow the OM healing process, as well as recovery of oral intake. This may be related with the findings of Erden et al. 28 , who found no significant result in the time of transition to oral feeding in patients who used cryotherapy for OM, compared to the use of a chlorhexidine mouthwash.
Finally, it is important to mention the role of probiotics in the modulation of oral mucositis processes. Several authors have studied how probiotics play a biomodulatory role in the dysbiosis generated in the body by oral cancer conditions and oral mucositis. In this regard, Xia et al. 40 conducted a phase II of a randomized clinical trial, in addition to testing a new combination of probiotics in patients with the same characteristics as the study described above, to measure the mechanisms of action of probiotics in rats. The probiotic formula used by Xia et al. 40 consisted of: Lactobacillus plantarum MH-301109 CFU, Bifidobacterium animalis subsp. lactis LPL-RH109 CFU, Lactobacillus rhamnosus LGG-18109 CFU, Lactobacillus acidophilus 109 CFU. Clinically, it showed improvement in OM severity, decreasing the inflammatory response. Regarding the mechanisms of action studied in parallel in the rat model, these results indicated inhibition of the peripheral immune response, inflammation and damage pathology, in addition to alleviating the severity of OM induced by radiotherapy and chemotherapy in rats. There was a significant decrease in proinflammatory markers, as well as regulation of the intestinal microbiota and an improvement in OM symptoms in the experimental group of rats vs. control. Both the histopathological and the mucosal analysis found that the strains used improved the dysbiosis generated.
Manifar et al. 41 conducted a randomized double-blind RCT in 64 patients with oral cancer who received radiotherapy, in which a prebiotic-based bioactive mouth rinse was used to alleviate the symptoms of grade 3 OM. The mouth wash was used three times a day for one to three minutes. The degree of mucositis in the case group from the 7th oral examination session was significantly lower than the control (p < 0.05), and this significant difference persisted until the last oral examination session, compared to the group control.
Undoubtedly, bacteria such as the Lactobacillus, Bifidobacterium, Enterococcus and Streptococcus species described by the aforementioned authors play an important role in modifying the symptoms of patients with OM.
CONCLUSIONS
In an extensive review, Worthington et al. 2011 13 conclude that there was some evidence for the effectiveness of cryotherapy, and weaker evidence for a benefit associated with glutamine (intravenous), honey, or laser. There was no evidence of a benefit associated with the use of chlorhexidine.
More than 10 years later, the current review found that the compounds that produced the best results in reducing OM severity were mouthwashes based on Dentoxol, Plantago major extract, and thyme honey; a zinc oxide paste; a vitamin B + GeneTime spray; and the consumption of oral L-glutamine. The results with LLL-T remain controversial and the use of chlorhexidine lacks evidence to support it as being any better than other treatments.
Our suggestion for future studies is to include factors such as discontinuation of treatment, and concentration, dose and frequency of application of therapies, which may have a significant influence on the results. Most of the selected studies used different interventions, which also makes it difficult to draw conclusions. Future systematic reviews comparing studies using a single type of treatment in different populations are suggested.