INTRODUCTION
Hymenochaetaceae can be distinguished among the poroid Basidiomycota by the rusty brown coloration of the basidiomata and positive xanthochroic reaction when in contact with potassium hydroxide (KOH) (Ryvarden & Johansen, 1980; Ryvarden, 2004). The basidioma can be stipitate, pileated or resupinate, the hymenophore hydnoid, smooth or more usually poroid, the hyphal system monomitic or dimitic, frequently presenting setae, and the spores usually smooth, rarely ornamented (Ryvarden, 2004). The family has around 55 genera (Wu et al., 2016, 2022; Salvador-Montoya et al., 2020; Catalogue of Life, 2022; Xavier de Lima et al., 2022), Fomitiporia and Fuscoporia among them. These genera have similar characteristics, but can be differentiated by morphological analysis. Fomitiporia presents basidiomata with pileal surface velutinate to smooth, generative hyphae hyaline to pale yellowish, thinto fairly thick walled, spores subglobose, hyaline, thick-walled, strongly dextrinoid and very strongly cyanophilous. Fuscoporia, in turn, presents basidiomata mostly with pileal surface tomentose to velutinate, non-crusted, generative hyphae at dissepiment edge or hymenium usually covered by crystals, which dissolve in KOH, spores cylindric, oblong-ellipsoid, broadly ellipsoid or subglobose, hyaline, thin-walled, smooth (Dai, 2010). In Brazil, both genera have an increasing number of occurrences, with records for all regions. Fomitiporia has so far 26 registered species, while Fuscoporia has 13, being, however, the real number probably much higher due to the frequent updates on the diversity of these genera (Flora and Funga do Brasil, 2022; Species Link, 2022).
The Atlantic Forest in Brazil originally covered 15% (MMA, 2010), mostly along the entire coastal zone of the country, suffering the impacts of the various economic cycles in Brazil and being today considered the most devastated and threatened biome on the entire planet (Cardoso, 2016). Nowadays, the remaining vegetation of the Atlantic Forest is around 28% (Rezende et al., 2018), holding from 1 to 8% of the world’s biodiversity, often distributed in areas still little known (Silva & Casteleti, 2005; MMA, 2010).
In the Atlantic Forest of Northeastern Brazil, two species of Fomitiporia and four of Fuscoporia are reported: Fo. conyana, Fo. maxonii (Flora e Funga do Brasil, 2022), Fu. formosana, Fu. licnoides, Fu. marquesiana, and Fu. scruposa (Yuan et al., 2020). In the current study, materials collected in Atlantic Forests areas in Northeastern Brazil were incorporated into the previous phylogeny of Fomitiporia and Fuscoporia, and improved the knowledge about the distribution of some other species of these genera, with new records for areas of Atlantic Forest in Brazil as well as for the states of Alagoas, Bahia and Pernambuco.
MATERIALS AND METHODS
The collections were carried out in the Parque National e Histórico (PARNAH) de Monte Pascoal, located in the extreme southern Bahia, in the municipality of Porto Seguro; in the Reserva Biológica (REBIO) de Pedra Talhada, located on the border between the states of Pernambuco and Alagoas and in the 7th Grupo de Artilharia de Campanha (GAC), municipality of Olinda, also in the state of Pernambuco. All areas belong to the Atlantic Rain Forest domain.
Morphological analyses
The specimens collected were dried in an oven and initially analyzed macroscopically. A drop of 3% KOH was poured over the basidioma to observe the presence or absence of color change (xanthochroic reaction). The basidiomata were analyzed according to the length, width, thickness, insertion in the substrate, consistency, color and characteristics of the abhymenial and hymenial surfaces, the context, and the margin of the basidioma (Kornerup & Wanscher, 1978; Fidalgo & Bononi, 1989). For microscopic analyses of hyphae, spores, basidia and setae, slides were prepared in 3% KOH in order to observe the microstructures, such as hyphae, basidia, basidiospore and hymenal setae. Melzer’s reagent was used to observe dextrinoid reactions of these structures.
Molecular and phylogenetic analyses
DNA extraction was performed according to the protocol of Góes-Neto et al. (2005) and the DNA regions of interest were amplified by polymerase chain reactions (PCR). The internal transcribed spacer regions (ITS) and the partial nuclear large subunit rDNA (nLSU) were amplified with primer pairs ITS4 and ITS5 for the ITS region, and LR0R and LR5 for the nLSU region (White et al., 1990). For Fuscoporia, the sequences from the ITS region presented problems and could not be included in the phylogenetic analysis. The electropherograms were analyzed and edited in the Staden Package 2.0 software (Staden et al., 1998) and the sequences obtained were compared with those deposited in GenBank using the BLASTn tool. Phellinus uncisetus Robledo, Urcelay & Rajchenberg was designated as an outgroup in the analyses of the genus Fomitiporia Murrill following Decock et al. (2007), while Coniferiporia sulphurascens (Pilát) L.W. Zhou & Y.C. Dai and Phellinidium fragrans (M.J. Larsen & Lombard) Nuss were used for Fuscoporia Murrill following Chen et al. (2019).
The phylogenetic trees were constructed using the Maximum Likelihood (ML) and Maximum Parsimony (MP) method with 1,000 bootstrap resamplings (Fig. 1 and 2). The phylogenetic reconstruction by ML and MP was performed using the MEGA X program (Kumar et al., 2018).
RESULTS AND DISCUSSION
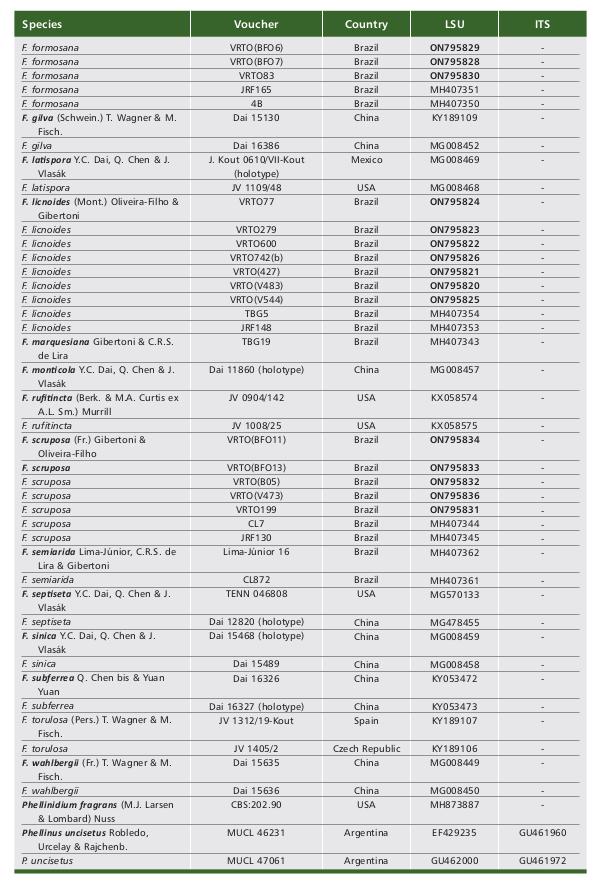
The LSU and ITS region datasets included 27 new sequences generated in this study (Table 1). The best evolutionary models estimated for the alignments were T92+G+1 for the Fomitiporia dataset and K2+G+I for Fuscoporia dataset.
Taxonomy
Fomitiporia conyanaAlves-Silva & Drechsler-Santos, Mycological Progress 19 (8): 781 (2020).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°15’02.3’’ S, 36°25’37.8’’ W, 753 m asl, V.R.T. Oliveira, (VRTO8, URM 94049).
Notes.- Fomitiporia conyana has a perennial, pileate, sessile, solitary or gregarious basidiomata, then emerging in groups, semicircular in outline, rarely pendant, triquetrous, obtriquetrous to ungulate in section, occasionally with a basal umbo, pileus glabrous, slightly convex, concentrically zonate with multiple narrow bands, sometimes interleaved with broad bands and moderated sulcus, radially cracked when dried and old, pores round to angular, 6-8(9)/mm, hyphal system dimitic in all parts, hymenial setae absent, cystidioles fusoid, lanceolate, hyaline, basidiospores subglobose to globose (4.5)5-5.5(6) × 4-5(6) μm (Alves-Silva et al. 2020). Fomitiporia conyana is reported from Brazil, Ecuador and French Guiana, being in Brazil previously collected in the Bahia, Rio Grande do Sul and Santa Catarina states (Alves-Silva et al. 2020; Flora e Funga do Brasil, 2022; Species Link, 2022). Here, it is reported as the second record for the Northeastern Brazil, being a new record for the Alagoas state.
Fomitiporia maxonii Murrill ENT#091;as ‘maxoni’ENT#093;, North American Flora 9 (1): 11 (1907).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°15’23.0’’ S, 36°24’47.0’’ W, 542 m asl, 14-V-2019, V.R.T. Oliveira,
(VRTO463, URM 93748); Pernambuco, Recife, Universidade Federal de Pernambuco, 8°3’6.239” S, 34°57’2.578” W, 15-V-2018, I. Oliveira-Júnior, (VRTO438, URM 94982).
Notes.- Fomitiporia maxonii has a perennial and resupinate basidiomata with a sterile margin, corky to woody, hymenophore with circular to ellipsoid or oblique pores, 7-9 per mm, hyphal system dimitic with hymenial setae absent, basidiospores subglobose to globose, (4.5-)5.3-6.5(-7) × (4-)4.8-6(-6.7) μm, and hyaline rhomboid crystals abundant in the hymenium as an important feature (Raymundo et al., 2012). Fomitiporia maxonii is reported from Argentina (Raymundo et al., 2012), Belize (Ryvarden, 2004; Raymundo et al., 2012), Brazil(Raymundo et al., 2012), Costa Rica (Ryvarden, 2004; Decock et al., 2007), Cuba (Decock et al., 2007; Raymundo et al., 2012), Ecuador (Ryvarden, 2004; Raymundo et al., 2012), Jamaica (Decock et al., 2007; Raymundo et al., 2012), Mexico (Raymundo et al., 2012), USA (Vlasák et al., 2011), and Venezuela (Decock et al., 2007; Raymundo et al., 2012), being in Brazil reported in the North, Northeast, Southeast and South regions, as well as for the states studied here (Flora e Funga do Brasil, 2022; Species Link, 2022).
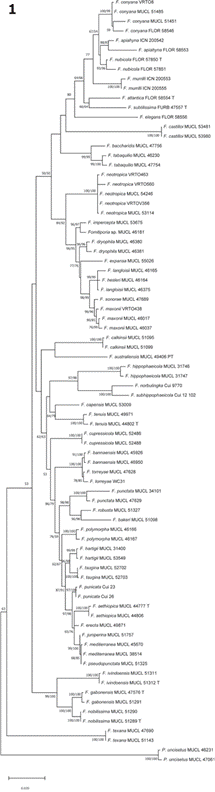
Fig 1 Fig. 1 (left). Maximum likelihood (ML) tree of Fomitiporia from dataset of combined sequences LSU and ITS. Bootstrap values above 50 % are shown. (esquerda). Árvore de máxima verossimilhança (ML) de Fomitiporia a partir do conjunto de dados de sequências LSU. Valores de bootstrap acima de 50% são mostrados.

Fig. 2 Fig. 2 (above). Maximum likelihood (ML) tree of Fuscoporia from dataset of LSU sequences. Bootstrap values above 50 % are shown. (acima). Árvore de máxima verossimilhança (ML) de Fuscoporia a partir do conjunto de dados de sequências LSU. Valores de bootstrap acima de 50% são mostrados.
Table 1 Tabela 1 Table 1 Tabela 1 Table 1 Tabela 1 (1/3). Data of sequences included in the phylogenetic molecular analyses. New sequences generated in this study are in boldface. (1/3). Dados de sequências incluídas nas análises moleculares filogenéticas. Novas sequências geradas neste estudo estão em negrito. (2/3). (2/3). (3/3). (3/3).


Fomitiporia neotropica Camp.-Sant., Amalfi, R.M. Silveira, Robledo & Decock, Mycological Progress 13: 610 (2014).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°15’23.0’’ S, 36°24’47.0’’ W, 542 m asl, 15-V-2019, V.R.T. Oliveira,
(VRTO560, URM 94056); Bahia, Itamaraju, Parque Nacional e Histórico do Monte Pascoal, 09°15’02.3’’ S, 36°25’37.8’’ W, 753 m asl, 8-VIII-2018, V. Xavier de Lima, ENT#091;VRTO(V356), URM 93714ENT#093;.
Notes.- The species has seasonal to at least bi-seasonal basidiomata, resupinate, effusive and adnate, cork consistency when fresh and hard cork when dry, with a densely fibrous texture, dense and velvety margin, small, round to ellipsoid pores in inclined parts, 6-9 per mm, dimitic hyphae system with hymenial setae ranging from absent to abundant and hyaline basidiospores subglobose to largely obovoid, 5-7 (−7.5) × 4.5-7 μm (CamposSantana et al., 2014). Fomitiporia neotropica is reported from Argentina, Brazil and French Guiana, being in Brazil previously collected in the North, Midwest, Southeast and South regions (CamposSantana et al., 2014; Flora e Funga do Brasil, 2022; GBIF, 2022; Species Link, 2022). Here, it is reported as the first record for the Northeastern Brazil, being a new record for the states Alagoas and Bahia.
Fuscoporia atlantica Motato-Vásq., R.M. Pires & Gugliotta, Mycotaxon 130 (3): 848 (2015).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°15’00.7’’ S, 36°25’38.3’’ W, 758 m asl, 16-VII-2018, V.R.T. Oliveira, (VRTO24, URM 94980).
Notes.- The species is characterized by having an annual, hairy, sessile to effuse-reflex basidioma, flexible when fresh to leathery when dry, solitary to imbricate, semicircular and flattened cap with a concentrically zoned upper surface, radially wrinkled, glabrous to tomentose, 7-9 pores per mm, hyphal system dimitic with abundant hymenial setae, spores from broadly ellipsoid to ellipsoid, hyaline to pale yellow, smooth, thin-walled and inamyloid, 4-4.5 × (2-)3-3.5 μm (Pires et al., 2015). Our specimen differs from the original description by a leatherier to papyraceous basidioma when dry. It was known only from the type locality in the Atlantic Forest in the state of São Paulo. Here, we report the second record of the species and the first for the Atlantic Forest of Northeastern Brazil, more specifically for the state of Alagoas (Pires et al., 2015; Species Link, 2022).
Fuscoporia formosana (T.T. Chang & W.N. Chou) T. Wagner & M. Fisch., Mycologia 94 (6): 1013 (2002).
Basionym: Inonotus formosanus T.T. Chang & W.N. Chou, Mycological Research 102 (7): 789 (1998).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°14’44.8’’ S, 36°25’14.7 W’’, 688 m asl, 16-VII-2018, V.R.T. Oliveira,
(VRTO83, URM 93763); Pernambuco, Olinda, 7th Grupo de Artilharia de Campanha, 8°0’10.728’’ S, 34°51’25.427’’ W, 16-VI-2018, V.R.T. Oliveira, ENT#091;VRTO(BFO7), URM 94503ENT#093;.
Notes.- Fuscoporia formosana has an annual, sessile, solitary or overlapping basidiomata, of woody consistency, flattened, convex, fan-shaped cap with sharp edges, gray to black on the surface, rounded or subangular pores, 6-8 per mm, hyphae system dimitic and with hymenial setae, ellipsoid to ovoid basidiospores, smooth, yellow or brown, 4-5 × 3-4 μm (Chang & Chou, 1999; Yuan et al., 2020). Fuscoporia formosana was previously reported for Maranhão and Pernambuco, being those the first for the Americas (Yuan et al., 2020). Here, the species is reported for the first time for the Alagoas state, but it may occur in other states identified as Phellinus gilvus (Schwein.) Pat. or P. gilvus var. scruposus (Fr.) S. Ahmad (Yuan et al., 2020).
Fuscoporia licnoides (Mont.) Oliveira-Filho & Gibertoni, Fungal Diversity 104: 129 (2020).
Basionym: Polyporus licnoides Mont., Annales des Sciences Naturelles Botanique 13: 204 (1840).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°14’47.0’’ S, 36°25’15.0’’ W, 695 m asl, 17-VII-2018, V.R.T. Oliveira, (VRTO77, URM 93747); 09°15’25.0’’ S, 36°24’47.0’’ W, 540 m asl, 4-II-2019, V.R.T.
Oliveira, (VRTO279, URM 93769); 09°15’00.7’’ S, 36°25’38.3’’ W, 758 m asl, 14-V2019, V.R.T. Oliveira, (VRTO600, URM 93816); ENT#091;VRTO742(b), URM 94976ENT#093;; Bahia,
Itamaraju, Parque Nacional e Histórico do Monte Pascoal, 16°51’55.6’’ S, 39°24’54.7’’ W 131 m asl, 9-VIII-2018, V. Xavier de Lima, ENT#091;VRTO(V427), URM 93739ENT#093;; 16°53’33.8’’ S, 39°24’37.8’’ W, 408 m asl, 7-VIII-2018, V. Xavier de Lima, ENT#091;VRTO(V483), URM 93742ENT#093;; 16°51’01.7’’ S, 39°24’14.7’’ W, 36 m asl, 5-VIII-2018, V. Xavier de Lima, ENT#091;VRTO(V544), URM 93740ENT#093;.
Notes.- The species is characterized by hard corky to slightly flexible pileus up to 1 cm thick and a hymenial surface concentrically and narrowly zonate and sulcate, grayish shade and often showing at the middle portion of the surface zones with a purplish-brown shade (Fidalgo & Fidalgo, 1968; Yuan et al., 2020). Fuscoporia licnoides may be confused to F. semiarida Lima-Júnior, C.R.S. de Lira & Gibertoni or F. atlantica or may have been previously identified as P. gilvus. Thus, specimens identified as such should be reexamined (Yuan et al., 2020). In Brazil, it is reported in Pará, Paraíba, Pernambuco, and Rondônia (Yuan et al., 2020) and it is here reported for the first time for the states of Alagoas and Bahia.
Fuscoporia scruposa (Fr.) Gibertoni & Oliveira-Filho, Fungal Diversity 104: 130 (2020).
Basionym: Polyporus scruposus Fr., Epicrisis Systematis Mycologici: 473 (1838).
Material examined.- BRAZIL. Alagoas, Quebrangulo, Reserva Biológica de Pedra Talhada, 09°15’31.0’’ S, 36°25’10.3’’W, 599 m asl, 8-II-2019, V.R.T. Oliveira,
(VRTO199, URM 93756); Bahia, Itamaraju, Parque Nacional e Histórico do Monte Pascoal, 16°51’01.4’’ S, 39°24’12.9’’ W, 41 m asl, 12-II-2019, D. P. B. Monte, ENT#091;VR-
TO(B05), URM 93727ENT#093;; Pernambuco, Olinda, 7th Grupo de Artilharia de Campanha, 8°0’10.728’’ S , 34°51’25.427’’ W, 16-VI-2018, V.R.T. Oliveira, ENT#091;VRTO(BFO13), URM
94662)ENT#093;; ENT#091;VRTO(BFO11), URM 94981ENT#093;.
Notes.- Fuscoporia scruposa has a hard to corky, but more often flexible pileus up 0.5 cm thick, hymenial surface concolorous, strigose and fibrillose with radial furrows, being ochraceous to rusty-brown and becoming glabrous with age (Fidalgo & Fidalgo, 1968; Yuan et al., 2020). Fuscoporia scruposa may be confused with F. formosana or have been previously identified as P. gilvus or P. gilvus var. scruposus, thus specimens worldwide should be reexamined (Yuan et al., 2020). In Brazil, there are reports in Alagoas, Maranhão and Piauí (Yuan et al., 2020), being here reported for the first time to Bahia and Pernambuco.
In the current study, the collected and analyzed materials were placed in Fomitiporia and Fuscoporia with good support (Fig. 1 and 2) and represent Fomitiporia conyana, Fo. maxonii, Fo. neotropica, Fuscoporia atlantica, Fu. formosana, Fu. licnoides and Fu. scruposa. So far, 12 species of Fomitiporia and six of Fuscoporia are phylogenetically confirmed to Brazil (Table 2 and 3).
Among the species in Fomitiporia, VRTO8 formed a clade with F. conyana, indicating that it is the same species (bootstrap = 100/99) (Fig. 1). The sequences grouped in the clade and are from Ecuador and Brazil (Tab. 1). Fomitiporia conyana was situated close to F. apiahyna and F. nubicola (Fig. 1). The similarity between the species has already been observed by Alves-Silva et al. (2020), which reports that
F. apiahyna has basidiospores up to 7 μm wide and not more than 8 pores/mm and that F. nubicola has basidiospores up to 6.5 μm wide and up to 9 pores/mm, while in
F. conyana the basidiospores are, in average, 4.9 × 4.4 μm.
The clade formed by VRTO463, VRTO560 and VRTO(V356) and F. neotropica (MUCL 53114 from French Guiana and MUCL 54246 from Brazil) (bootstrap = 100/100) confirms that they belong to the same species (Fig. 1). The type of F. neotropica is from French Guiana and the species is common in the Neotropics (Campos-Santana et al., 2014). Fomitiporia neotropica is in a clade with F. dryophila, F. expansa, F. impercepta, F. langloisii/hesleri, F. maxonii, and F. sonorae (bootstrap = 84/92) (Fig. 1). Fomitiporia dryophila differs from F. neotropica in the cushion-shaped to pseudopileate basidiomata and larger basidiospores (5.5-)6.2-8(−8.5) × (5-)5.7-7.3(−7.5) μm (Decock et al., 2007; Campos-Santana et al., 2014). Fomitiporia neotropica is rather similar morphologically, but differs by F. expansa in having much less extended basidiomata, with a distinctly brown pore surface and smaller pores, mostly 6-9/ mm (Amalfi & Decock, 2014). Fomitiporia impercepta differs by smaller basidiospores (4.0)5.0-6.0(7.0) × 4.0-6.0(7.0) μm (Campos-Santana et al., 2014; Morera et al., 2017; Rajchenberg et al. 2019). Fomitiporia langloisii has a paler pore surface, grayish and honey-colored, sometimes with a slight pinkish tinge in young specimens, while F. neotropica commonly has a pore surface yellowish brown, greyish brown, dark yellow to dark brown or greyish chocolate brown (Decock et al., 2007; Raymundo et al., 2012; Campos-Santana et al., 2014). Decock et al. (2007) suggested that F. langlosi is an older available name for F. hesleri. Fomitiporia neotropica and F. maxonii share similar characteristics, except for the presence of setae, which are not reported in F.
Table 2 Tabela 2 Morphological characters of species of Fomitiporia species onfirmed for Northeast Brazil through molecular analyses. Características morfológicas de espécies das espécies de Fomitiporia confirmadas para o Nordeste do Brasil por meio de análises moleculares.
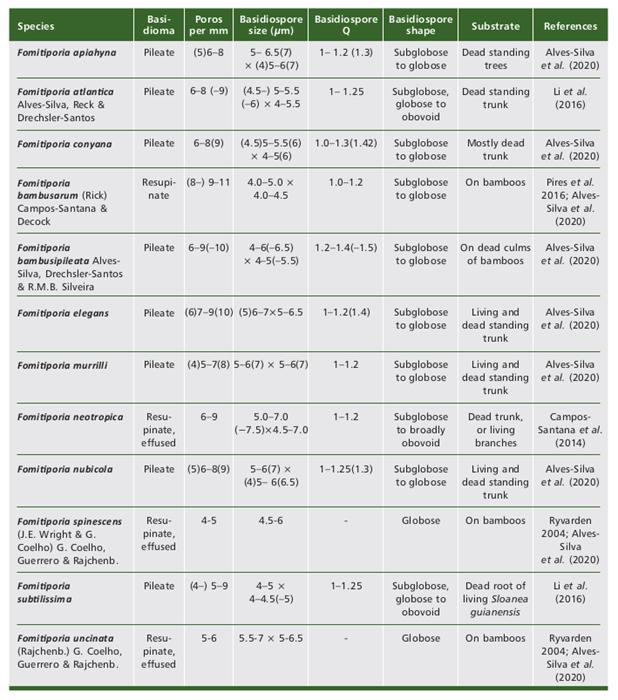
Table 3 Tabela 3 Morphological characters of species of the genus Fuscoporia confirmed for Northeast Brazil through molecular analyses. Características morfológicas de espécies do gênero Fuscoporia confirmadas para o Nordeste do Brasil por meio de análises moleculares.

VRTO438 formed a well-supported clade with F. maxonii and F. sonorae (bootstrap = 96/96) (Fig. 1). This clade indicates that there is a strong proximity between both species, as already demonstrated in previous works (Amalfi et al., 2012; Amalfi & Decock, 2013; Ota et al., 2014). Fuscoporia sonorae has been collected so far only in southern USA and northern Mexico (Gilbertson & Ryvarden, 1987; Raymundo et al., 2012), while F. maxonii is a more distinctly tropical species, with records from southern Florida to Argentina, with records from Brazil (Decock et al., 2007; Vlasák et al., 2011; Raymundo et al., 2012; Species Link, 2022). We consider that our specimen represents F. maxonii and suggest that, for a good delimitation between these two species, it is necessary to include other regions of the DNA in the phylogenetic analyses. The F. maxonii/F. sonorae clade is very close to F. langloisii (bootstrap = 98/99) (Fig. 1). Fomitiporia maxonii and F. langloisii are morphologically similar, but they can be differentiated by the firm adherence to the substrate and the resupinate to effused basidioma in F. langloisii, while in F. maxonni the basidioma is only resupinate and can be easily detached from the substrate (Raymundo et al., 2012).
Among the species of Fuscoporia, VRTO24 formed a clade with F. atlantica (bootstrap = 99/99), indicating that it is a representative of this species (Fig. 2). Fuscoporia atlantica formed a clade without support with F. scruposa (Fig. 2), from which it can be distinguished by the concentrically zonate pileus and the setae subulate to ventricose, mostly uncinate or hooked (Pires et al., 2015; Yuan et al., 2020).
VRTO(BFO3), VRTO(BFO6), VRTO(BFO7) and VRTO83 formed a clade with F. formosana (bootstrap = 91/99), while VRTO(BFO11), VRTO(BFO13), VRTO(B05), VRTO(V473) and VRTO199 with F. scruposa (bootstrap = 94/99) (Fig. 2). Fuscoporia formosana and F. scruposa are macro and microscopically very similar, both belonging to the “Phellinus scruposus” group within the “P. gilvus” complex (Yuan et al., 2020), requiring, therefore, molecular analyses for a better distinction. Phylogenetically, F. formosana formed a clade in common with two sequences identified as F. gilva from China (bootstrap = 85/91). The type of this species, however, was originally collected in the USA (Pennsylvania) and these samples should be reanalyzed (Yuan et al., 2020). Fuscoporia scruposa, in turn, is close to the clade formed between the species F. atlantica, as previously discussed.
CONCLUSION
The results indicate the importance of continuous investigation in the Atlantic Forest, which continues to reveal novelties about Hymenochaetaceae. Furthermore, the results obtained in this study improved the data on the geographic distribution of species and to elucidate the existing complexes, helping to identify species that are well distinguished only by molecular analysis.














